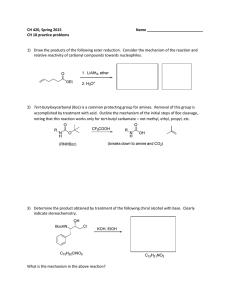
CH 420, Spring 2015 Name ___________________________ CH 18 practice problems
... 7) Rank the following compounds according to their relative acidity: cyclohexanol, phenol, pmethoxyphenol, p-nitrophenol. ...
... 7) Rank the following compounds according to their relative acidity: cyclohexanol, phenol, pmethoxyphenol, p-nitrophenol. ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034
... Write the Woodward Hoffmann rules for cycloaddition reactions. What is group transfer reaction? Give an example. Give the (1,5)-sigmatropic rearrangement reactions. What is photoisomerisation? Draw Jablonskii diagram and mention the photophysical processes. Mention the importance of retrosynthetic a ...
... Write the Woodward Hoffmann rules for cycloaddition reactions. What is group transfer reaction? Give an example. Give the (1,5)-sigmatropic rearrangement reactions. What is photoisomerisation? Draw Jablonskii diagram and mention the photophysical processes. Mention the importance of retrosynthetic a ...
Chapter Nine: Alcohols, Ethers and Epoxides
... common reagents. Predict the likelihood of carbon skeleton rearrangement under a given set of conditions. Predict the relative acidity of alcohols within a functional group class and compared to other functional groups. ...
... common reagents. Predict the likelihood of carbon skeleton rearrangement under a given set of conditions. Predict the relative acidity of alcohols within a functional group class and compared to other functional groups. ...
Fundamentals Of Organic Chemistry
... Mechanism of the dehydration of alcohol is discussed below, and also in elimination reactions, later in this book. Mechanism Protonation of the hydroxyl group of the alcohol followed by the loss of water molecule to give a 1° carbocation. The 1° carbocation undergo rearrangement to form more stable ...
... Mechanism of the dehydration of alcohol is discussed below, and also in elimination reactions, later in this book. Mechanism Protonation of the hydroxyl group of the alcohol followed by the loss of water molecule to give a 1° carbocation. The 1° carbocation undergo rearrangement to form more stable ...
Here is the Original File - University of New Hampshire
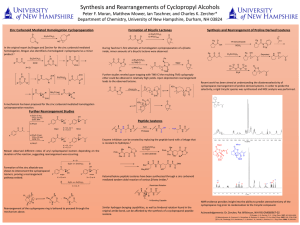
... Recent work has been aimed at understanding the diastereoselectivity of cyclopropane rearrangement of proline-derived systems. In order to probe the selectivity, a rigid tricyclic species was synthesized and NOE analysis was performed. ...
... Recent work has been aimed at understanding the diastereoselectivity of cyclopropane rearrangement of proline-derived systems. In order to probe the selectivity, a rigid tricyclic species was synthesized and NOE analysis was performed. ...
Vinylcyclopropane rearrangement

The vinylcyclopropane rearrangement or vinylcyclopropane-cyclopentene rearrangement is a ring expansion reaction, converting a vinyl-substituted cyclopropane ring into a cyclopentene ring.Intense experimental as well as computational investigations have revealed that mechanistically, the vinylcyclopropane rearrangement can be thought of as either a diradical-mediated two-step and/or orbital-symmetry-controlled pericyclic process. The amount by which each of the two mechanisms is operative is highly dependent on the substrate.Due to its ability to form cyclopentene rings the vinylcyclopropane rearrangement has served several times as a key reaction in complex natural product synthesis.