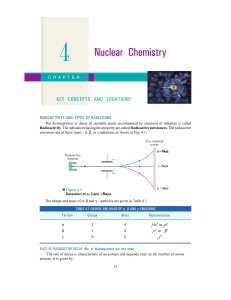
types of reactions
... state where 2 exactly opposite chemical reactions are occurring at the same place, same time, and same rate (speed) where reactions continuously occur •Two opposing forces are being exerted but they are in a state of balance •Amounts of all chemical entities are constant but do not have to be the sa ...
... state where 2 exactly opposite chemical reactions are occurring at the same place, same time, and same rate (speed) where reactions continuously occur •Two opposing forces are being exerted but they are in a state of balance •Amounts of all chemical entities are constant but do not have to be the sa ...
Nuclear Chemistry
... Mass defect = Experimental mass of the nucleus – (mass of protons + electrons + neutrons). It is equal to the total nucleon mass minus the nuclear mass. This loss of mass is converted into energy which stabilizes the nucleus. This energy is known as binding energy. It is also equal to the energy nee ...
... Mass defect = Experimental mass of the nucleus – (mass of protons + electrons + neutrons). It is equal to the total nucleon mass minus the nuclear mass. This loss of mass is converted into energy which stabilizes the nucleus. This energy is known as binding energy. It is also equal to the energy nee ...
Study of Neutron and Gamma Radiation Protective
... Concrete is one of the best and most widely used materials for manufacture of Gamma and neutron radiation shield, because in addition to having the proper Structural properties of the material, there are variety choice of the materials used to build it, that lead to manufacture of concrete with diff ...
... Concrete is one of the best and most widely used materials for manufacture of Gamma and neutron radiation shield, because in addition to having the proper Structural properties of the material, there are variety choice of the materials used to build it, that lead to manufacture of concrete with diff ...
General, Organic, and Biological Chemistry: An Integrated Approach
... 10) A 100-mg technetium-99m sample is used in a medical study. How much of the Technetium99m sample remains after 24 hours? The half-life of Tc-99m is 6 hours. Answer: 24 hours/6 hours = 4 half lives; 100 mg → 50 mg → 25 mg → 12.5 mg → 6.25 mg Section: 2-8 11) Krypton-81m is used for lung ventilatio ...
... 10) A 100-mg technetium-99m sample is used in a medical study. How much of the Technetium99m sample remains after 24 hours? The half-life of Tc-99m is 6 hours. Answer: 24 hours/6 hours = 4 half lives; 100 mg → 50 mg → 25 mg → 12.5 mg → 6.25 mg Section: 2-8 11) Krypton-81m is used for lung ventilatio ...
Nuclear medicine physics - The Canadian Organization of Medical
... 9. Material X has an initial number of nuclei and is placed in a neutron field with fluence rate ...
... 9. Material X has an initial number of nuclei and is placed in a neutron field with fluence rate ...
Sub Unit Plan 1 Chem Periodic Table
... II.1 The placement or location of elements on the Periodic Table gives an indication of physical and chemical properties of that element. The elements on the Periodic Table are arranged in order of increasing atomic number. (3.1y) II.2 The number of protons in an atom (atomic number) identifies the ...
... II.1 The placement or location of elements on the Periodic Table gives an indication of physical and chemical properties of that element. The elements on the Periodic Table are arranged in order of increasing atomic number. (3.1y) II.2 The number of protons in an atom (atomic number) identifies the ...
3 - Study Hungary
... A: the atomic number decreases by 2 and the mass number by 4. B: the atomic number decreases by 4 and the mass number by 2. C: the atomic number increases by 1 and the mass number doesn’t change. D: the loss of a neutron decreases the mass number by 1 and the charge by 1. E: the loss of a proton dec ...
... A: the atomic number decreases by 2 and the mass number by 4. B: the atomic number decreases by 4 and the mass number by 2. C: the atomic number increases by 1 and the mass number doesn’t change. D: the loss of a neutron decreases the mass number by 1 and the charge by 1. E: the loss of a proton dec ...
Structure - Bhoj University
... or end point energy, as it is called, is different for different nuclides. 3. The emission of -particles may not be associated with the emission -rays e.g. Na24 emits - rays whereas N13, P32, In114 do not emit -rays. In some cases of -emission a single continuous spectrum is found and ther ...
... or end point energy, as it is called, is different for different nuclides. 3. The emission of -particles may not be associated with the emission -rays e.g. Na24 emits - rays whereas N13, P32, In114 do not emit -rays. In some cases of -emission a single continuous spectrum is found and ther ...
AP Chemistry Summer Assignment - Belle Vernon Area School District
... The summer assignment for AP Chemistry has two parts. 1. Complete the practice problems in this packet. You may use the resources listed below as well as any notes/worksheets from Accel. Chem that you may have. 2. You need to master the formulas, charges, and names of the common ions. On the first w ...
... The summer assignment for AP Chemistry has two parts. 1. Complete the practice problems in this packet. You may use the resources listed below as well as any notes/worksheets from Accel. Chem that you may have. 2. You need to master the formulas, charges, and names of the common ions. On the first w ...
chem pre ap atom and nuclear practice test
... c. of each element are identical in size, mass, and other properties. d. of different elements cannot combine. 2. Which of the following statements is true? a. Atoms of the same element may have different masses. b. Atoms may be divided in ordinary chemical reactions. c. Atoms can never combine with ...
... c. of each element are identical in size, mass, and other properties. d. of different elements cannot combine. 2. Which of the following statements is true? a. Atoms of the same element may have different masses. b. Atoms may be divided in ordinary chemical reactions. c. Atoms can never combine with ...
PPT format - Columbia University
... Element: An element is a substance which cannot be decomposed into simpler substances by chemical processes. Examples: hydrogen, carbon, oxygen. Atomic interpretation: An element is a substance that contains only one kind of atom. Hydrogen (H) atoms, carbon atoms (C), oxygen atoms (O). Compound: A c ...
... Element: An element is a substance which cannot be decomposed into simpler substances by chemical processes. Examples: hydrogen, carbon, oxygen. Atomic interpretation: An element is a substance that contains only one kind of atom. Hydrogen (H) atoms, carbon atoms (C), oxygen atoms (O). Compound: A c ...
Elements, Compounds, and Chemical Equations
... 1. Counting Subatomic Particles: Use the periodic table • The atomic number is the same as the number of protons. • The number of protons is the same as the number of electrons. • The mass number is the protons added to the neutrons. ...
... 1. Counting Subatomic Particles: Use the periodic table • The atomic number is the same as the number of protons. • The number of protons is the same as the number of electrons. • The mass number is the protons added to the neutrons. ...
Directed Reading
... a. Helium does not react with other substances but does form new substances. b. Helium reacts with other substances but does not form new substances. c. Helium reacts with other substances to form new substances. d. Helium does not react with other substances to form new substances. ______ 9. A subs ...
... a. Helium does not react with other substances but does form new substances. b. Helium reacts with other substances but does not form new substances. c. Helium reacts with other substances to form new substances. d. Helium does not react with other substances to form new substances. ______ 9. A subs ...
Atomic/Nuclear Models
... combination of atoms of individual elements in simple proportions (e.g., 1:1, 1:2, etc.). This atomic hypothesis explained chemical reactions and the distinct ratios in which elements combined to form compounds. However, Dalton made no statement about the structure of an atom. At the beginning of th ...
... combination of atoms of individual elements in simple proportions (e.g., 1:1, 1:2, etc.). This atomic hypothesis explained chemical reactions and the distinct ratios in which elements combined to form compounds. However, Dalton made no statement about the structure of an atom. At the beginning of th ...
preface The given educational edition on professional English
... 0.002 centimetre) thick would make an impression with blurry edges. For some particles, the blurring corresponded to a two-degree deflection. Remembering those results, Rutherford had his postdoctoral fellow, Hans Geiger, and an undergraduate student, Ernest Marsden, refine the experiment. The young ...
... 0.002 centimetre) thick would make an impression with blurry edges. For some particles, the blurring corresponded to a two-degree deflection. Remembering those results, Rutherford had his postdoctoral fellow, Hans Geiger, and an undergraduate student, Ernest Marsden, refine the experiment. The young ...
IPC – First Semester Exam Review Be able to classify an example
... Understand how to read chemical formulas o Chemical formulas represent substances – they usually represent compounds o Know how to count the number of elements in a chemical formula Count how many types of elements there are (count the number of atomic symbols) 5H2O = 2 elements: Hydrogen and Ox ...
... Understand how to read chemical formulas o Chemical formulas represent substances – they usually represent compounds o Know how to count the number of elements in a chemical formula Count how many types of elements there are (count the number of atomic symbols) 5H2O = 2 elements: Hydrogen and Ox ...
Chemistry General v. 2016
... Chemical Properties- A property of matter that describes a substance’s ability to participate in a chemical reaction; Endothermic Reaction- Describe a process in which heat is absorbed from the environment; Exothermic Reaction- Describe a process in which a system releases heat into the environment; ...
... Chemical Properties- A property of matter that describes a substance’s ability to participate in a chemical reaction; Endothermic Reaction- Describe a process in which heat is absorbed from the environment; Exothermic Reaction- Describe a process in which a system releases heat into the environment; ...
39 The Atomic Nucleus and Radioactivity
... • When two nucleons are just a few nucleon diameters apart, the nuclear force they exert on each other is nearly zero (small force vectors). • This means that if nucleons are to be held together by the strong force, they must be held in a very small volume. • Nuclei are tiny because the nuclear forc ...
... • When two nucleons are just a few nucleon diameters apart, the nuclear force they exert on each other is nearly zero (small force vectors). • This means that if nucleons are to be held together by the strong force, they must be held in a very small volume. • Nuclei are tiny because the nuclear forc ...
39 The Atomic Nucleus and Radioactivity
... • When two nucleons are just a few nucleon diameters apart, the nuclear force they exert on each other is nearly zero (small force vectors). • This means that if nucleons are to be held together by the strong force, they must be held in a very small volume. • Nuclei are tiny because the nuclear forc ...
... • When two nucleons are just a few nucleon diameters apart, the nuclear force they exert on each other is nearly zero (small force vectors). • This means that if nucleons are to be held together by the strong force, they must be held in a very small volume. • Nuclei are tiny because the nuclear forc ...
Midterm Review Teacher Answer Key December 21, 2011 `see
... eliminate static electricity in machinery. It is also used in brushes to remove dust from camera lenses. Polonium-210 can be created in the laboratory by bombarding bismuth-209 with neutrons to create bismuth210. The bismuth-210 undergoes beta decay to produce polonium-210. Polonium-210 has a halfli ...
... eliminate static electricity in machinery. It is also used in brushes to remove dust from camera lenses. Polonium-210 can be created in the laboratory by bombarding bismuth-209 with neutrons to create bismuth210. The bismuth-210 undergoes beta decay to produce polonium-210. Polonium-210 has a halfli ...
transmutation of nuclides
... The half-life is specific for a nuclide, unaffected by the chemical or physical state of the element. It can be used for the radioactive nuclide identifications. Half-life measurements are often done in order to identify the nuclide present. As pointed out in the previous two sections, sometimes the ...
... The half-life is specific for a nuclide, unaffected by the chemical or physical state of the element. It can be used for the radioactive nuclide identifications. Half-life measurements are often done in order to identify the nuclide present. As pointed out in the previous two sections, sometimes the ...
A. Alpha particle. - MDC Faculty Web Pages
... measuring the rate of decay of a known quantity using a radiation detector. Copyright © 2008 Pearson Education, Inc., publishing as Pearson Addison-Wesley ...
... measuring the rate of decay of a known quantity using a radiation detector. Copyright © 2008 Pearson Education, Inc., publishing as Pearson Addison-Wesley ...
Radioactive Decays – transmutations of nuclides
... The half-life is specific for a nuclide, unaffected by the chemical or physical state of the element. It can be used for the radioactive nuclide identifications. Half-life measurements are often done in order to identify the nuclide present. As pointed out in the previous two sections, sometimes the ...
... The half-life is specific for a nuclide, unaffected by the chemical or physical state of the element. It can be used for the radioactive nuclide identifications. Half-life measurements are often done in order to identify the nuclide present. As pointed out in the previous two sections, sometimes the ...