Helium bubbling in a Molten Salt Fast Reactor
... experimental method included the making of a new set-up with a newly fabricated piece of equipment and developing a method to work with this set-up. The result of the set-up is a modified version of a Hallimond tube. The set-up is built to use the methods to examine a flotation process, which are de ...
... experimental method included the making of a new set-up with a newly fabricated piece of equipment and developing a method to work with this set-up. The result of the set-up is a modified version of a Hallimond tube. The set-up is built to use the methods to examine a flotation process, which are de ...
Chemistry - Plymouth Public Schools
... Central Concept: Physical and chemical properties reflect the nature of the interactions between molecules or atoms, and can be used to classify and describe matter. MA CHM 1.1 Identify and explain physical properties (e.g., density, melting point, boiling point, conductivity, malleability) and chem ...
... Central Concept: Physical and chemical properties reflect the nature of the interactions between molecules or atoms, and can be used to classify and describe matter. MA CHM 1.1 Identify and explain physical properties (e.g., density, melting point, boiling point, conductivity, malleability) and chem ...
Nuclear Glossary as PDF-file
... accounting relates to a defined, limited, walled-in space the contents of which results from the difference of all continuously measured nuclear material additions and withdrawals. At the end of the accounting period the plant inventory is determined by an independent direct measurement. →MUF Activa ...
... accounting relates to a defined, limited, walled-in space the contents of which results from the difference of all continuously measured nuclear material additions and withdrawals. At the end of the accounting period the plant inventory is determined by an independent direct measurement. →MUF Activa ...
The Role of Initial Conditions in the Decay of Spatially Periodic
... This is obvious when inspecting their q−dependence plotted as a function of the dimensionless wavenumber square q 02 = (qd/π)2 in Figs. 3a and 3b for k = 1, ..., 8. The wk have been determined with the help of Eq. (3), where for simplicity ϑEC (q, z, 0) has been approximated by its leading term sin( ...
... This is obvious when inspecting their q−dependence plotted as a function of the dimensionless wavenumber square q 02 = (qd/π)2 in Figs. 3a and 3b for k = 1, ..., 8. The wk have been determined with the help of Eq. (3), where for simplicity ϑEC (q, z, 0) has been approximated by its leading term sin( ...
Science Focus 9 Matter and Chemical Change Class Notes Topic 1
... Scientists began using heating, burning, mixing, and cooling to take matter down until it could not be broken down any further, to determine if a substance was a pure substance or a mixture. Antoine Lavoisier defined elements as pure substances that could not be decomposed into simpler substances by ...
... Scientists began using heating, burning, mixing, and cooling to take matter down until it could not be broken down any further, to determine if a substance was a pure substance or a mixture. Antoine Lavoisier defined elements as pure substances that could not be decomposed into simpler substances by ...
As a result of activities in grades 9
... Solids, liquids, and gases differ in the distances and angles between molecules or atoms and therefore the energy that binds them together. In solids the structure is nearly rigid; in liquids molecules or atoms move around each other but do not move apart; and in gases molecules or atoms move almost ...
... Solids, liquids, and gases differ in the distances and angles between molecules or atoms and therefore the energy that binds them together. In solids the structure is nearly rigid; in liquids molecules or atoms move around each other but do not move apart; and in gases molecules or atoms move almost ...
서울대학교 일반화학실험
... lead to ions of different charge; however, identity of the element remains unchanged. Changes in the number of protons deep in the atomic nucleus results in the creation of entirely different elements with new chemical and physical properties. Hydrogen made in the big bang universe and heavy element ...
... lead to ions of different charge; however, identity of the element remains unchanged. Changes in the number of protons deep in the atomic nucleus results in the creation of entirely different elements with new chemical and physical properties. Hydrogen made in the big bang universe and heavy element ...
atomic mass
... • Consists of two types of particles • Proton: Positively charged subatomic particle – Number defines the element • Neutron: Electrically neutral subatomic particle © 2014 W. W. Norton Co., Inc. ...
... • Consists of two types of particles • Proton: Positively charged subatomic particle – Number defines the element • Neutron: Electrically neutral subatomic particle © 2014 W. W. Norton Co., Inc. ...
DOE FUNDAMENTALS HANDBOOK NUCLEAR PHYSICS AND REACTOR THEORY Volume 1 of 2
... Science; Chemistry; Engineering Symbology, Prints, and Drawings; and Nuclear Physics and Reactor Theory. The handbooks are provided as an aid to DOE nuclear facility contractors. These handbooks were first published as Reactor Operator Fundamentals Manuals in 1985 for use by DOE category A reactors. ...
... Science; Chemistry; Engineering Symbology, Prints, and Drawings; and Nuclear Physics and Reactor Theory. The handbooks are provided as an aid to DOE nuclear facility contractors. These handbooks were first published as Reactor Operator Fundamentals Manuals in 1985 for use by DOE category A reactors. ...
Chapter 4 and 5
... 6.022*1023 atoms of any element will have a mass equal to the amu mass but in grams! The mole concept is based on the 12C isotope because it was available in high purity at the time. Mole – the quantity of a substance which contains the same number of units as there are atoms in exactly 12g of 12C. ...
... 6.022*1023 atoms of any element will have a mass equal to the amu mass but in grams! The mole concept is based on the 12C isotope because it was available in high purity at the time. Mole – the quantity of a substance which contains the same number of units as there are atoms in exactly 12g of 12C. ...
FREE Sample Here
... D. There are no other factors related to the 11 protons and 12 neutrons. Section 2.4 Difficulty Level: Easy 17. Atoms of elements belonging to the same group have an identical number of A. total electrons. B. energy levels. C. inner electrons. D. valence electrons. Section 2.4 Difficulty Level: Easy ...
... D. There are no other factors related to the 11 protons and 12 neutrons. Section 2.4 Difficulty Level: Easy 17. Atoms of elements belonging to the same group have an identical number of A. total electrons. B. energy levels. C. inner electrons. D. valence electrons. Section 2.4 Difficulty Level: Easy ...
FREE Sample Here
... into simpler substances. Examples of a metal, a nonmetal, and a semimetal will vary. ...
... into simpler substances. Examples of a metal, a nonmetal, and a semimetal will vary. ...
general-organic-and-biological-chemistry-3rd-edition
... Difficulty Level: Easy 1. Which statement is incorrect? A. According to the atomic theory, all matter is composed of atoms. B. Protons, neutrons, and electrons are subatomic particles. C. Electrons have greater mass than protons. D. Neutrons are found in the nucleus of the atom. Section 2.1 Difficul ...
... Difficulty Level: Easy 1. Which statement is incorrect? A. According to the atomic theory, all matter is composed of atoms. B. Protons, neutrons, and electrons are subatomic particles. C. Electrons have greater mass than protons. D. Neutrons are found in the nucleus of the atom. Section 2.1 Difficul ...
Nuclear Physics 1 NWNC
... Science; Chemistry; Engineering Symbology, Prints, and Drawings; and Nuclear Physics and Reactor Theory. The handbooks are provided as an aid to DOE nuclear facility contractors. These handbooks were first published as Reactor Operator Fundamentals Manuals in 1985 for use by DOE category A reactors. ...
... Science; Chemistry; Engineering Symbology, Prints, and Drawings; and Nuclear Physics and Reactor Theory. The handbooks are provided as an aid to DOE nuclear facility contractors. These handbooks were first published as Reactor Operator Fundamentals Manuals in 1985 for use by DOE category A reactors. ...
Physical Earth Daily Learning Guide DRAFT - Burlington
... 9-11 PS1B Average acceleration 9-11 PS1C Newton’s First Law of Motion, the Law of Inertia 9-11 PS1D Newton’s Second Law of Motion, F=ma 9-11 PS1E Newton’s Third Law of Motion 9-11 PS1F Newton’s Law of Universal Gravitation 9-11 PS1G Electrical force 9-11 PS1H Electricity and magnetism are two aspect ...
... 9-11 PS1B Average acceleration 9-11 PS1C Newton’s First Law of Motion, the Law of Inertia 9-11 PS1D Newton’s Second Law of Motion, F=ma 9-11 PS1E Newton’s Third Law of Motion 9-11 PS1F Newton’s Law of Universal Gravitation 9-11 PS1G Electrical force 9-11 PS1H Electricity and magnetism are two aspect ...
IN-BEAM SPECTROSCOPY OF EXTREMELY NEUTRON
... The nucleus can be regarded as being made of neutrons and protons interacting predominantly via the strong force. However, the forces between the nucleons in a nucleus are not due to the bare interactions between the constituent quarks, but rather effective forces that can be schematically described, ...
... The nucleus can be regarded as being made of neutrons and protons interacting predominantly via the strong force. However, the forces between the nucleons in a nucleus are not due to the bare interactions between the constituent quarks, but rather effective forces that can be schematically described, ...
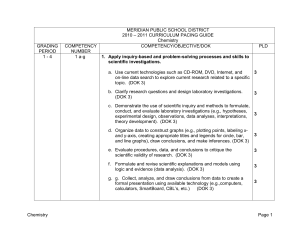
MERIDIAN PUBLIC SCHOOL DISTRICT
... of Dalton, Thomson, Rutherford, Bohr, de Broglie, and Schrődinger and describe how each discovery contributed to the current model of atomic and nuclear structure. (DOK 2) dWrite appropriate equations for nuclear decay reactions, describe how the nucleus changes during these reactions, and compare t ...
... of Dalton, Thomson, Rutherford, Bohr, de Broglie, and Schrődinger and describe how each discovery contributed to the current model of atomic and nuclear structure. (DOK 2) dWrite appropriate equations for nuclear decay reactions, describe how the nucleus changes during these reactions, and compare t ...
M-2 Slide Show File
... Helium’s melting and boiling points are lower than those of any other known substance and is the only element that cannot be solidified by cooling under standard conditions. All of the Noble Gases have very low melting and boiling points, and are all very close together, meaning that they are liquid ...
... Helium’s melting and boiling points are lower than those of any other known substance and is the only element that cannot be solidified by cooling under standard conditions. All of the Noble Gases have very low melting and boiling points, and are all very close together, meaning that they are liquid ...
Sample Final Questions Key/FS12
... f. Johannes Diderik van der Waals (Gas Laws) revised the ideal gas law equation so that it can be used for real gases. g. Henri Becquerel (Nuclear) while studying fluorescence determined that some glowing rocks actually have particles coming off of them – the advent of radioactivity. h. Pierre and M ...
... f. Johannes Diderik van der Waals (Gas Laws) revised the ideal gas law equation so that it can be used for real gases. g. Henri Becquerel (Nuclear) while studying fluorescence determined that some glowing rocks actually have particles coming off of them – the advent of radioactivity. h. Pierre and M ...
Ground state properties of neutron-rich Mg isotopes – the “island of
... what is known about the properties of stable and long-lived, near-stable nuclei. Between these nuclei and the drip lines, where nuclear binding comes to an end, lies an unexplored landscape containing more than 90 percent of all expected bound nuclear systems, a region where many new nuclear phenome ...
... what is known about the properties of stable and long-lived, near-stable nuclei. Between these nuclei and the drip lines, where nuclear binding comes to an end, lies an unexplored landscape containing more than 90 percent of all expected bound nuclear systems, a region where many new nuclear phenome ...
Pearson Physics Level 30 Unit VIII Atomic Physics: Chapter 16
... The quantity of mass that is converted to 5.00 GJ of energy is 5.56 108 kg . 5. Isotopes are atoms that have the same atomic number but different neutron numbers. They are chemically very similar but do not have the same atomic mass. 6. A stable nucleus is bound together by the strong nuclear for ...
... The quantity of mass that is converted to 5.00 GJ of energy is 5.56 108 kg . 5. Isotopes are atoms that have the same atomic number but different neutron numbers. They are chemically very similar but do not have the same atomic mass. 6. A stable nucleus is bound together by the strong nuclear for ...
Untitled
... The chemistry we have talked about so far has dealt with what happens _________ the nucleus. Remember that there are 3 particles that make up an atom. The _________ and ___________ reside inside the nucleus while the _________ (which are responsible for ions, bonding, reactions, electricity, light, ...
... The chemistry we have talked about so far has dealt with what happens _________ the nucleus. Remember that there are 3 particles that make up an atom. The _________ and ___________ reside inside the nucleus while the _________ (which are responsible for ions, bonding, reactions, electricity, light, ...
19-Oct
... the mole proportions of chemical reactions. Stoichiometric ratio: The ratio of any two species (reactants or products) in a balanced chemical reaction. ...
... the mole proportions of chemical reactions. Stoichiometric ratio: The ratio of any two species (reactants or products) in a balanced chemical reaction. ...