CHEM110P1_06_2015_Y_P1
... A student used a titration to determine whether an unknown sample was malonic acid (CH2(COOH)2, molar mass = 104.1 g mol–1). The student weighed 1.08 g of the unknown acid and transferred it to a 250.0 mL volumetric flask and prepared a standard solution. The burette was filled with 0.09970 M NaOH s ...
... A student used a titration to determine whether an unknown sample was malonic acid (CH2(COOH)2, molar mass = 104.1 g mol–1). The student weighed 1.08 g of the unknown acid and transferred it to a 250.0 mL volumetric flask and prepared a standard solution. The burette was filled with 0.09970 M NaOH s ...
21:3 Classifying Chemical Reactions
... Yeast is a microscopic, one-celled organism belonging to the group of organisms called fungi. There are many kinds of yeasts, some of them of great importance to humans. Yeast is necessary to make leavened bread, beer, and cheese. It is rich in B vitamins; a form of yeast called brewer's yeast is us ...
... Yeast is a microscopic, one-celled organism belonging to the group of organisms called fungi. There are many kinds of yeasts, some of them of great importance to humans. Yeast is necessary to make leavened bread, beer, and cheese. It is rich in B vitamins; a form of yeast called brewer's yeast is us ...
Science Focus 9 Matter and Chemical Change Class Notes Topic 1
... Scientists began using heating, burning, mixing, and cooling to take matter down until it could not be broken down any further, to determine if a substance was a pure substance or a mixture. Antoine Lavoisier defined elements as pure substances that could not be decomposed into simpler substances by ...
... Scientists began using heating, burning, mixing, and cooling to take matter down until it could not be broken down any further, to determine if a substance was a pure substance or a mixture. Antoine Lavoisier defined elements as pure substances that could not be decomposed into simpler substances by ...
eBook AQA GCSE Chemistry Unit C2 Part 1
... Today, scientists are excited about nanoscience – the study of structures measuring around one billionth of a metre. Will discoveries at the nanoscale lead to new cancer cures, greener energy technologies, and better computers? ...
... Today, scientists are excited about nanoscience – the study of structures measuring around one billionth of a metre. Will discoveries at the nanoscale lead to new cancer cures, greener energy technologies, and better computers? ...
Chapter 3 Molecules, Compounds, and Chemical Equations How
... release hydrogen ions (H+) when dissolved in water. • Acids are composed of hydrogen, usually written first in their formula, and one or more nonmetals, written second. – HCl is a molecular compound that, when dissolved in water, forms H+(aq) and Cl–(aq) ions, where aqueous (aq) means dissolved in w ...
... release hydrogen ions (H+) when dissolved in water. • Acids are composed of hydrogen, usually written first in their formula, and one or more nonmetals, written second. – HCl is a molecular compound that, when dissolved in water, forms H+(aq) and Cl–(aq) ions, where aqueous (aq) means dissolved in w ...
CHEMISTRY Academic Standards Statement
... v. Chemical bonds form through the sharing or transfer of electrons between atoms. The nature and quantity of chemical bonds in a chemical species give rise to the shape, structure and microscopic properties of that species. 2.1.2 Methods of structure determination i. A variety of experimental (e.g. ...
... v. Chemical bonds form through the sharing or transfer of electrons between atoms. The nature and quantity of chemical bonds in a chemical species give rise to the shape, structure and microscopic properties of that species. 2.1.2 Methods of structure determination i. A variety of experimental (e.g. ...
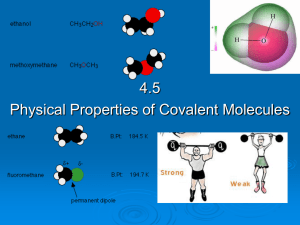
4.5 Physical properties of molecular covalent
... Covalent molecules are not charged because they are overall neutral and therefore do not conduct electricity. • Some covalent molecules can react with water and produce free ions which can carry an electrical current. • E.g. ammonia, NH3 NH3 (l) + H2O (l) ↔ NH4+ (aq) + OH- (aq) ...
... Covalent molecules are not charged because they are overall neutral and therefore do not conduct electricity. • Some covalent molecules can react with water and produce free ions which can carry an electrical current. • E.g. ammonia, NH3 NH3 (l) + H2O (l) ↔ NH4+ (aq) + OH- (aq) ...
Syllabus of Medical / Dental Colleges Entrance Test 2016
... e) Explain the shape of, and bond angles in ethane, ethene and benzene molecules in terms of σ and Π bonds ...
... e) Explain the shape of, and bond angles in ethane, ethene and benzene molecules in terms of σ and Π bonds ...
a. Matter First Day of Class
... 4. Describe the activities that are part of the scientific method. 5. Describe how you tell call whether you have a pure element or a compound. Major Goals of Sections 3.1 - 3.3 1. The organization of matter concept map. 2. Classify matter as pure substances or mixtures. 3. Homogeneous versus hetero ...
... 4. Describe the activities that are part of the scientific method. 5. Describe how you tell call whether you have a pure element or a compound. Major Goals of Sections 3.1 - 3.3 1. The organization of matter concept map. 2. Classify matter as pure substances or mixtures. 3. Homogeneous versus hetero ...
ordinary level chemistry syllabus
... not have been successful without the participation of a range of different education stakeholders and the financial support from different donors. For this I would like to express my deep gratitude. My thanks firstly goes to the Rwanda Education leadership who supervised the curriculum review proces ...
... not have been successful without the participation of a range of different education stakeholders and the financial support from different donors. For this I would like to express my deep gratitude. My thanks firstly goes to the Rwanda Education leadership who supervised the curriculum review proces ...
InChI keys as standard global identifiers in chemistry web services
... would be a non-proprietary identifier for chemical substances that could be used in printed and electronic data sources thus enabling easier linking of diverse data compilations. The initial work focused on the development of algorithms for converting an input organic chemical structure to a unique ...
... would be a non-proprietary identifier for chemical substances that could be used in printed and electronic data sources thus enabling easier linking of diverse data compilations. The initial work focused on the development of algorithms for converting an input organic chemical structure to a unique ...
Practice Problem Set #6
... 3. Complete and balance the equations for the following reactions. a. Na(s) + Br2(l) → b. Mg(s) + O2(g) → c. Al(s) + F2(g) → d. C(s) + O2(g) → (assume an excess of oxygen has been added) 4. Calcium oxide, CaO, is used to remove SO2 from power plant exhaust. These two compounds react to give so ...
... 3. Complete and balance the equations for the following reactions. a. Na(s) + Br2(l) → b. Mg(s) + O2(g) → c. Al(s) + F2(g) → d. C(s) + O2(g) → (assume an excess of oxygen has been added) 4. Calcium oxide, CaO, is used to remove SO2 from power plant exhaust. These two compounds react to give so ...
Unit 2 Summary - A
... State the two main applications of fossil fuels:............................................ and .................................... State two significant disadvantages of being over-reliant on fossil fuels: ................................................................... and ................... ...
... State the two main applications of fossil fuels:............................................ and .................................... State two significant disadvantages of being over-reliant on fossil fuels: ................................................................... and ................... ...
Kinetics in the Study of Organic Reaction Mechanisms
... establishing the correct mechanism, but in ruling out all of the possible mechanisms which are ineonswtent with the kineticdata. I n the examples used above, it mas assumed, as is common practice, that concentrations and activities of reactant species are the same. This is not usually the case, exce ...
... establishing the correct mechanism, but in ruling out all of the possible mechanisms which are ineonswtent with the kineticdata. I n the examples used above, it mas assumed, as is common practice, that concentrations and activities of reactant species are the same. This is not usually the case, exce ...
Chapter 8
... decomposition, single-displacement, and doubledisplacement reactions. • Classify a reaction as a synthesis, decomposition, single-displacement, double-displacement, or combustion reaction. • List three kinds of synthesis reactions and six kinds of decomposition reactions. ...
... decomposition, single-displacement, and doubledisplacement reactions. • Classify a reaction as a synthesis, decomposition, single-displacement, double-displacement, or combustion reaction. • List three kinds of synthesis reactions and six kinds of decomposition reactions. ...
7.1 Describing Reactions
... 3. Ethylene, C2H4, burns in the presence of oxygen to produce carbon dioxide and water vapor. Write a balanced equation for this reaction. Answer: C2H4 + 3O2 2CO2 + 2 H2O ...
... 3. Ethylene, C2H4, burns in the presence of oxygen to produce carbon dioxide and water vapor. Write a balanced equation for this reaction. Answer: C2H4 + 3O2 2CO2 + 2 H2O ...
7.1 Describing Reactions
... 3. Ethylene, C2H4, burns in the presence of oxygen to produce carbon dioxide and water vapor. Write a balanced equation for this reaction. Answer: C2H4 + 3O2 2CO2 + 2 H2O ...
... 3. Ethylene, C2H4, burns in the presence of oxygen to produce carbon dioxide and water vapor. Write a balanced equation for this reaction. Answer: C2H4 + 3O2 2CO2 + 2 H2O ...
Slide 1
... 3. Ethylene, C2H4, burns in the presence of oxygen to produce carbon dioxide and water vapor. Write a balanced equation for this reaction. Answer: C2H4 + 3O2 2CO2 + 2 H2O ...
... 3. Ethylene, C2H4, burns in the presence of oxygen to produce carbon dioxide and water vapor. Write a balanced equation for this reaction. Answer: C2H4 + 3O2 2CO2 + 2 H2O ...
VCE Chemistry Study Design
... Chemistry is a key science in explaining the workings of our universe through an understanding of the properties and interaction of substances that make up matter. Most processes, from the formation of molecules in outer space to the complex biological interactions occurring in cells, can be describ ...
... Chemistry is a key science in explaining the workings of our universe through an understanding of the properties and interaction of substances that make up matter. Most processes, from the formation of molecules in outer space to the complex biological interactions occurring in cells, can be describ ...
7.1 Describing Reactions
... 3. Ethylene, C2H4, burns in the presence of oxygen to produce carbon dioxide and water vapor. Write a balanced equation for this reaction. Answer: C2H4 + 3O2 2CO2 + 2 H2O ...
... 3. Ethylene, C2H4, burns in the presence of oxygen to produce carbon dioxide and water vapor. Write a balanced equation for this reaction. Answer: C2H4 + 3O2 2CO2 + 2 H2O ...
States of Matter
... Many liquids are composed of molecules that exert specific and often highly directional forces on each other. The most notable example of such a liquid is water, in which a hydrogen atom of one H2 O molecule is attracted to the oxygen atom of another molecule. This effect known as hydrogen bonding; hy ...
... Many liquids are composed of molecules that exert specific and often highly directional forces on each other. The most notable example of such a liquid is water, in which a hydrogen atom of one H2 O molecule is attracted to the oxygen atom of another molecule. This effect known as hydrogen bonding; hy ...
Chapter 2: Atoms, Molecules and Ions
... 1. Each element is composed of extremely small, indivisible particles called atoms. 2. All atoms of a given element are identical to one another in mass and other properties, but the atoms of one element are different from the atoms of all other elements. 3. Atoms of an element are not changed into ...
... 1. Each element is composed of extremely small, indivisible particles called atoms. 2. All atoms of a given element are identical to one another in mass and other properties, but the atoms of one element are different from the atoms of all other elements. 3. Atoms of an element are not changed into ...
406 K (English version)
... conceived: moving from a transmission method of teaching (expository teaching model) to a method of knowledge development. It involves prioritising the learner-centred approach: Increasing the number of secondary school teachers and improving their performance through ICT. Integrating ICT tools ...
... conceived: moving from a transmission method of teaching (expository teaching model) to a method of knowledge development. It involves prioritising the learner-centred approach: Increasing the number of secondary school teachers and improving their performance through ICT. Integrating ICT tools ...
Organic chemistry

Organic chemistry is a chemistry subdiscipline involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms. Study of structure includes many physical and chemical methods to determine the chemical composition and the chemical constitution of organic compounds and materials. Study of properties includes both physical properties and chemical properties, and uses similar methods as well as methods to evaluate chemical reactivity, with the aim to understand the behavior of the organic matter in its pure form (when possible), but also in solutions, mixtures, and fabricated forms. The study of organic reactions includes probing their scope through use in preparation of target compounds (e.g., natural products, drugs, polymers, etc.) by chemical synthesis, as well as the focused study of the reactivities of individual organic molecules, both in the laboratory and via theoretical (in silico) study.The range of chemicals studied in organic chemistry include hydrocarbons (compounds containing only carbon and hydrogen), as well as myriad compositions based always on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (these, included in many organic chemicals in biology) and the radiostable elements of the halogens.In the modern era, the range extends further into the periodic table, with main group elements, including:Group 1 and 2 organometallic compounds, i.e., involving alkali (e.g., lithium, sodium, and potassium) or alkaline earth metals (e.g., magnesium)Metalloids (e.g., boron and silicon) or other metals (e.g., aluminium and tin)In addition, much modern research focuses on organic chemistry involving further organometallics, including the lanthanides, but especially the transition metals; (e.g., zinc, copper, palladium, nickel, cobalt, titanium and chromium)Finally, organic compounds form the basis of all earthly life and constitute a significant part of human endeavors in chemistry. The bonding patterns open to carbon, with its valence of four—formal single, double, and triple bonds, as well as various structures with delocalized electrons—make the array of organic compounds structurally diverse, and their range of applications enormous. They either form the basis of, or are important constituents of, many commercial products including pharmaceuticals; petrochemicals and products made from them (including lubricants, solvents, etc.); plastics; fuels and explosives; etc. As indicated, the study of organic chemistry overlaps with organometallic chemistry and biochemistry, but also with medicinal chemistry, polymer chemistry, as well as many aspects of materials science.