Higher Chemistry Resources Guide - Glow Blogs
... attraction that can operate between all atoms and molecules. These forces are much weaker than all other types of bonding. They are formed as a result of electrostatic attraction between temporary dipoles and induced dipoles caused by movement of electrons in atoms and molecules. The strength of Lon ...
... attraction that can operate between all atoms and molecules. These forces are much weaker than all other types of bonding. They are formed as a result of electrostatic attraction between temporary dipoles and induced dipoles caused by movement of electrons in atoms and molecules. The strength of Lon ...
Chapter 6 Ionic and Molecular Compounds
... • What do they have in common that could be the cause of this lack of reactivity? • Examination of their electron configurations reveals that the noble gases either have 1. an outermost electron energy level that is completely filled with electrons (He = 2 e- in the 1st energy level, Ne = 8 e- in th ...
... • What do they have in common that could be the cause of this lack of reactivity? • Examination of their electron configurations reveals that the noble gases either have 1. an outermost electron energy level that is completely filled with electrons (He = 2 e- in the 1st energy level, Ne = 8 e- in th ...
EVS - RSC - Developments in Microwave Chemistry
... Initially, microwave chemistry was primarily used to carry out analytical processes such as ashing, digestion, extraction, fat analysis and protein hydrolysis. As microwave chemical synthesis has advanced, its applications have been extended to include the synthesis of fine chemicals, organometallic ...
... Initially, microwave chemistry was primarily used to carry out analytical processes such as ashing, digestion, extraction, fat analysis and protein hydrolysis. As microwave chemical synthesis has advanced, its applications have been extended to include the synthesis of fine chemicals, organometallic ...
Toward Greener Chemistry Methods for Preparation of
... In polymer chemistry, microwave-assisted reactions present a dramatic increasing in reac‐ tion speed and significant improvements in yield compared with conventional heating. These advantages are attributed to instantaneous and direct heating of the reactants, which lead to reduction in reaction tim ...
... In polymer chemistry, microwave-assisted reactions present a dramatic increasing in reac‐ tion speed and significant improvements in yield compared with conventional heating. These advantages are attributed to instantaneous and direct heating of the reactants, which lead to reduction in reaction tim ...
Higher Chemistry Resources Guide - Glow Blogs
... In a covalent bond, atoms share pairs of electrons. The covalent bond is a result of two positive nuclei being held together by their common attraction for the shared pair of electrons. Polar covalent bonds are formed when the attraction of the atoms for the pair of bonding electrons is different. D ...
... In a covalent bond, atoms share pairs of electrons. The covalent bond is a result of two positive nuclei being held together by their common attraction for the shared pair of electrons. Polar covalent bonds are formed when the attraction of the atoms for the pair of bonding electrons is different. D ...
FREE Sample Here
... OBJ: Describe the classifications of matter: elements, compounds, and mixtures (heterogeneous and homogeneous). TOP: general concepts | matter KEY: states of matter MSC: general chemistry 16. A solution is a A) pure element. B) pure mixture. C) heterogeneous mixture. D) homogeneous mixture. E) pure ...
... OBJ: Describe the classifications of matter: elements, compounds, and mixtures (heterogeneous and homogeneous). TOP: general concepts | matter KEY: states of matter MSC: general chemistry 16. A solution is a A) pure element. B) pure mixture. C) heterogeneous mixture. D) homogeneous mixture. E) pure ...
Chemical Dynamics at Surfaces
... the third of four sons of Charles Langmuir and Sadie, neé Comings. His early education was obtained in various schools and institutes in the USA, and in Paris (1892-1895). He graduated as a metallurgical engineer from the School of Mines at Columbia University in 1903. Postgraduate work in Physical ...
... the third of four sons of Charles Langmuir and Sadie, neé Comings. His early education was obtained in various schools and institutes in the USA, and in Paris (1892-1895). He graduated as a metallurgical engineer from the School of Mines at Columbia University in 1903. Postgraduate work in Physical ...
GCSE Chemistry Sample Assessment Materials
... Write your name, centre number and candidate number in the spaces at the top of this page. Answer all questions. Write your answers in the spaces provided in this booklet. ...
... Write your name, centre number and candidate number in the spaces at the top of this page. Answer all questions. Write your answers in the spaces provided in this booklet. ...
Grossmont College Chemistry 120 Laboratory Manual 6th Edition
... with the eye, the most effective first aid is the immediate flushing of the eye with copious amounts of tap water. You are seldom more than a few seconds from a faucet. Continue flushing for at least fifteen minutes and then consult a physician at once. If your laboratory is equipped with eye founta ...
... with the eye, the most effective first aid is the immediate flushing of the eye with copious amounts of tap water. You are seldom more than a few seconds from a faucet. Continue flushing for at least fifteen minutes and then consult a physician at once. If your laboratory is equipped with eye founta ...
GCE Chemistry SAMs 2009 onwards pdf
... Here are his results Mass of ethanol before experiment Mass of ethanol after experiment Mass of water Temperature of water before experiment Temperature of water after experiment ...
... Here are his results Mass of ethanol before experiment Mass of ethanol after experiment Mass of water Temperature of water before experiment Temperature of water after experiment ...
PART 3-ICHO 11-15
... individual compound of metal A obtained as a precipitate, was separated, thoroughly washed, dried and calcinated. The mass of the precipitate after the calcination to constant mass, was 0.3265 g. An aqueous ammonia solution was added in excess to the solution obtained after separation of the precipi ...
... individual compound of metal A obtained as a precipitate, was separated, thoroughly washed, dried and calcinated. The mass of the precipitate after the calcination to constant mass, was 0.3265 g. An aqueous ammonia solution was added in excess to the solution obtained after separation of the precipi ...
Chapter - Imperial Valley College
... Molecular Models • Models show the 3-dimensional structure along with all the other information given in structural formula • Ball-and-Stick Models use balls to represent the atoms and sticks to represent the attachments between them ...
... Molecular Models • Models show the 3-dimensional structure along with all the other information given in structural formula • Ball-and-Stick Models use balls to represent the atoms and sticks to represent the attachments between them ...
Soft X-Ray-Induced Decomposition of Amino Acids: An XPS, Mass
... using only Gaussian line shapes and thus neglecting the small Lorentzian contribution was sufficient. Intensities of the shake-up satellites were not taken into account. Standard atomic sensitivity factors (40) were used, which were empirically corrected for the analyzer transmission so as to achiev ...
... using only Gaussian line shapes and thus neglecting the small Lorentzian contribution was sufficient. Intensities of the shake-up satellites were not taken into account. Standard atomic sensitivity factors (40) were used, which were empirically corrected for the analyzer transmission so as to achiev ...
Worked solutions to the problems
... materials that your students use and produce. Students should of course also make themselves aware of any hazards associated with the chemicals that they will be using in any exercise and we encourage you to bring these to their attention. For our junior colleagues who will spend many hours over the ...
... materials that your students use and produce. Students should of course also make themselves aware of any hazards associated with the chemicals that they will be using in any exercise and we encourage you to bring these to their attention. For our junior colleagues who will spend many hours over the ...
Examiners` Report November 2012 GCSE Chemistry
... This was the second 5CH2H paper to be offered; the first being set in June 2012. This question paper assessed the specification items to be in Unit 2 Discovering Chemistry which forms part of the Additional Science course along with the corresponding biology and physics units, and also as part of th ...
... This was the second 5CH2H paper to be offered; the first being set in June 2012. This question paper assessed the specification items to be in Unit 2 Discovering Chemistry which forms part of the Additional Science course along with the corresponding biology and physics units, and also as part of th ...
2.6 M - Thierry Karsenti
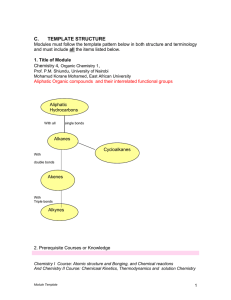
... atoms in the cyclic structure is other than carbon. Heterocyclic componds may be aliphatic or aromatic 15. Isomers: These are different compounds that have the same molecular formula. Isomers are further subdivided into: (a) structural isomers, (b) geometrical isomers and (c) stereoisomers(optical i ...
... atoms in the cyclic structure is other than carbon. Heterocyclic componds may be aliphatic or aromatic 15. Isomers: These are different compounds that have the same molecular formula. Isomers are further subdivided into: (a) structural isomers, (b) geometrical isomers and (c) stereoisomers(optical i ...
wiley_ch6_Chemical_Equilibrium
... Effect of Temperature Changes T shifts reaction in direction that produces endothermic (heat absorbing) change T shifts reaction in direction that produces exothermic (heat releasing) change Changes in T change value of mass action expression at equilibrium, so K changed K depends on T ...
... Effect of Temperature Changes T shifts reaction in direction that produces endothermic (heat absorbing) change T shifts reaction in direction that produces exothermic (heat releasing) change Changes in T change value of mass action expression at equilibrium, so K changed K depends on T ...
AP Chemistry - Notes
... A. Stoichiometry - the study of quantities of substances consumed and produced in chemical reactions. 1. Atomic mass - the average mass of an atom of an element in atomic mass units (amu) a. Atomic masses are based on 12C ("carbon twelve"), which is assigned a value of exactly 12 atomic mass units. ...
... A. Stoichiometry - the study of quantities of substances consumed and produced in chemical reactions. 1. Atomic mass - the average mass of an atom of an element in atomic mass units (amu) a. Atomic masses are based on 12C ("carbon twelve"), which is assigned a value of exactly 12 atomic mass units. ...
Grossmont College Chemistry 141 Laboratory Manual 6th Edition
... weigh approximately 1.5 grams. If you try to measure the length of a board and the end of the board falls between two of the marks on your measuring tape; you would have to estimate the last fractional length. The only measurement that can be determined with certainty is counting a small set of obje ...
... weigh approximately 1.5 grams. If you try to measure the length of a board and the end of the board falls between two of the marks on your measuring tape; you would have to estimate the last fractional length. The only measurement that can be determined with certainty is counting a small set of obje ...
PDF of this page - Miami bulletin
... phenomena, collecting and sharing data and evaluating results critically. IVB, LAB. CAS-D/LAB. Co-requisite: CHM 141 required. CHM 144H. College Chemistry Laboratory. (2) (MPF) Presents laboratory exercises to illustrate the fundamental principles of chemistry. An emphasis will be placed on safety, ...
... phenomena, collecting and sharing data and evaluating results critically. IVB, LAB. CAS-D/LAB. Co-requisite: CHM 141 required. CHM 144H. College Chemistry Laboratory. (2) (MPF) Presents laboratory exercises to illustrate the fundamental principles of chemistry. An emphasis will be placed on safety, ...
17 - Wiley
... 14.47 Use arrows of different sizes to show differences in electron density shifts. ...
... 14.47 Use arrows of different sizes to show differences in electron density shifts. ...
Unit 8: Reactions
... 3. Double Replacement: A solution reaction in which the positive ion of one compound combines with the negative ion of the other compound to form a precipitate, and the other ions remain dissolved in solution. 4. Law of Conservation of Charge: Charge may not be created or destroyed by physical or ch ...
... 3. Double Replacement: A solution reaction in which the positive ion of one compound combines with the negative ion of the other compound to form a precipitate, and the other ions remain dissolved in solution. 4. Law of Conservation of Charge: Charge may not be created or destroyed by physical or ch ...
PDF of this page
... B. S. in Biochemistry in collaboration with the Department of Biological Sciences. For students in the College of Engineering and Applied Sciences we offer the B. S. in Chemistry. In the College of Arts and Sciences, the traditional degree certified by the American Chemical Society is offered; the B ...
... B. S. in Biochemistry in collaboration with the Department of Biological Sciences. For students in the College of Engineering and Applied Sciences we offer the B. S. in Chemistry. In the College of Arts and Sciences, the traditional degree certified by the American Chemical Society is offered; the B ...
Unit V The Mole
... ________________________________________________________________________ During the time that Dalton’s mass scale was just being introduced, the French chemist ______________________________ began to study how gases reacted. When Gay– Lussac reacted pairs of gases at the same temperature and pressur ...
... ________________________________________________________________________ During the time that Dalton’s mass scale was just being introduced, the French chemist ______________________________ began to study how gases reacted. When Gay– Lussac reacted pairs of gases at the same temperature and pressur ...
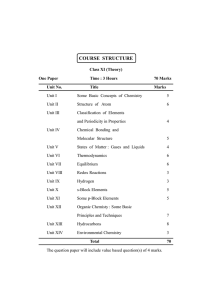
COURSE STRUCTURE
... where h is Planck’s constant and p is momentum of the particle. The wave nature of electron has been confirmed byDavisson and Germer’s experiment whereas the particle nature is confirmed byscintillation method as well as by the photoelectric effect. Heisenberg’s uncertainty Principle states that ‘‘I ...
... where h is Planck’s constant and p is momentum of the particle. The wave nature of electron has been confirmed byDavisson and Germer’s experiment whereas the particle nature is confirmed byscintillation method as well as by the photoelectric effect. Heisenberg’s uncertainty Principle states that ‘‘I ...
Analytical chemistry

Analytical chemistry is the study of the separation, identification, and quantification of the chemical components of natural and artificial materials. Qualitative analysis gives an indication of the identity of the chemical species in the sample, and quantitative analysis determines the amount of certain components in the substance. The separation of components is often performed prior to analysis.Analytical methods can be separated into classical and instrumental. Classical methods (also known as wet chemistry methods) use separations such as precipitation, extraction, and distillation and qualitative analysis by color, odor, or melting point. Classical quantitative analysis is achieved by measurement of weight or volume. Instrumental methods use an apparatus to measure physical quantities of the analyte such as light absorption, fluorescence, or conductivity. The separation of materials is accomplished using chromatography, electrophoresis or field flow fractionation methods.Analytical chemistry is also focused on improvements in experimental design, chemometrics, and the creation of new measurement tools to provide better chemical information. Analytical chemistry has applications in forensics, bioanalysis, clinical analysis, environmental analysis, and materials analysis.