Week 1 -- Schedule
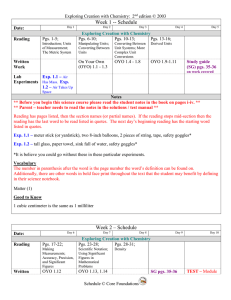
... ** Before you begin this science course please read the student notes in the book on pages i-iv. ** ** Parent – teacher needs to read the notes in the solutions / test manual ** Reading has pages listed, then the section names (or partial names). If the reading stops mid-section then the reading has ...
... ** Before you begin this science course please read the student notes in the book on pages i-iv. ** ** Parent – teacher needs to read the notes in the solutions / test manual ** Reading has pages listed, then the section names (or partial names). If the reading stops mid-section then the reading has ...
Question Bank - Edudel.nic.in
... Which of the following is a dimensionless quantity; molarity, molality or mole fraction? [Ans. : mole fraction] ...
... Which of the following is a dimensionless quantity; molarity, molality or mole fraction? [Ans. : mole fraction] ...
4420 Natural History of the Vertebrates. (3-3) Environmental
... 4422 Mammalogy. (3-3) The taxonomy, distribution, ecology, behavior and evolution of mammals with particular emphasis on wild animals of the southwest. Laboratory will emphasize anatomy, identification, preparation of specimens and field exercises in the methods of population analysis. Prerequisites ...
... 4422 Mammalogy. (3-3) The taxonomy, distribution, ecology, behavior and evolution of mammals with particular emphasis on wild animals of the southwest. Laboratory will emphasize anatomy, identification, preparation of specimens and field exercises in the methods of population analysis. Prerequisites ...
Conformational studies of aliphatic secondary ozonides
... conformations of such a ring can be classified into an envelope (E) form in which one atom lies out of the plane of the other atoms, and twist (T) form in which two consecutive atoms lie on opposite faces of the plane of the other three atoms. Theoretically the five membered ring of the secondary ozon ...
... conformations of such a ring can be classified into an envelope (E) form in which one atom lies out of the plane of the other atoms, and twist (T) form in which two consecutive atoms lie on opposite faces of the plane of the other three atoms. Theoretically the five membered ring of the secondary ozon ...
Chemistry
... – for his development of nuclear magnetic resonance spectroscopy for determining the three-dimensional structure of biological macromolecules in solution ...
... – for his development of nuclear magnetic resonance spectroscopy for determining the three-dimensional structure of biological macromolecules in solution ...
Chapter 6
... plexation reactions, and oxidation–reduction reactions. In this section we review these reactions and their equilibrium constant expressions. 6D.1 Precipitation Reactions In a precipitation reaction, two or more soluble species combine to form an insoluble precipitate. The most common precipitation ...
... plexation reactions, and oxidation–reduction reactions. In this section we review these reactions and their equilibrium constant expressions. 6D.1 Precipitation Reactions In a precipitation reaction, two or more soluble species combine to form an insoluble precipitate. The most common precipitation ...
CHM203 - National Open University of Nigeria
... The melting point of a substance can be defined as the temperature at which it undergoes the transition from the solid to the liquid state. Pure crystalline solids have sharp melting points. Thus, melting point is used as an important physical property both for the identification of organic compound ...
... The melting point of a substance can be defined as the temperature at which it undergoes the transition from the solid to the liquid state. Pure crystalline solids have sharp melting points. Thus, melting point is used as an important physical property both for the identification of organic compound ...
Document
... • To measure the reaction rate you need to be able to measure the concentration of at least one component in the mixture at many points in time • There are two ways of approaching this problem 1. for reactions that are complete in less than 1 hour, it is best to use continuous monitoring of the conc ...
... • To measure the reaction rate you need to be able to measure the concentration of at least one component in the mixture at many points in time • There are two ways of approaching this problem 1. for reactions that are complete in less than 1 hour, it is best to use continuous monitoring of the conc ...
Chapter
... • To measure the reaction rate you need to be able to measure the concentration of at least one component in the mixture at many points in time • There are two ways of approaching this problem 1. for reactions that are complete in less than 1 hour, it is best to use continuous monitoring of the conc ...
... • To measure the reaction rate you need to be able to measure the concentration of at least one component in the mixture at many points in time • There are two ways of approaching this problem 1. for reactions that are complete in less than 1 hour, it is best to use continuous monitoring of the conc ...
Chapter 14 (Kinetics) – Slides and Practice
... • To measure the reaction rate you need to be able to measure the concentration of at least one component in the mixture at many points in time • There are two ways of approaching this problem 1. for reactions that are complete in less than 1 hour, it is best to use continuous monitoring of the conc ...
... • To measure the reaction rate you need to be able to measure the concentration of at least one component in the mixture at many points in time • There are two ways of approaching this problem 1. for reactions that are complete in less than 1 hour, it is best to use continuous monitoring of the conc ...
Unit 8: Reactions
... 3. Double Replacement: A solution reaction in which the positive ion of one compound combines with the negative ion of the other compound to form a precipitate, and the other ions remain dissolved in solution. 4. Law of Conservation of Charge: Charge may not be created or destroyed by physical or ch ...
... 3. Double Replacement: A solution reaction in which the positive ion of one compound combines with the negative ion of the other compound to form a precipitate, and the other ions remain dissolved in solution. 4. Law of Conservation of Charge: Charge may not be created or destroyed by physical or ch ...
Biosignature storage in sulfate minerals
... The discovery of jarosite on Mars in 2004 generated increased interest in the properties of the mineral related to the search for life on other planets. Several studies indicate that the formation of jarosite can be linked to biological activity on Earth and biomolecules such as amino acids have bee ...
... The discovery of jarosite on Mars in 2004 generated increased interest in the properties of the mineral related to the search for life on other planets. Several studies indicate that the formation of jarosite can be linked to biological activity on Earth and biomolecules such as amino acids have bee ...
Mead Chemistry Lap 11: Stoichiometry Chapter 12 12.1 Balanced
... • Number of moles ▫ Use coefficients to find # of moles ▫ __ mole of N2 reacts with __ moles of H2 to form __ moles of NH3 ▫ Ratio of 1 N2: 3 H2 :2 NH3 • Mass in grams ▫ All reactions must follow law of conservation of mass ▫ Atoms and mass will always follow conservation ▫ Use mole ratios and molar ...
... • Number of moles ▫ Use coefficients to find # of moles ▫ __ mole of N2 reacts with __ moles of H2 to form __ moles of NH3 ▫ Ratio of 1 N2: 3 H2 :2 NH3 • Mass in grams ▫ All reactions must follow law of conservation of mass ▫ Atoms and mass will always follow conservation ▫ Use mole ratios and molar ...
Experimental Study of Closed System in the Chlorine Dioxide
... 2.1. Materials. Chlorine dioxide aqueous solution was prepared from sodium chlorite and diluted sulfuric acid and was purified by bubbling through 10% sodium chlorite aqueous solution to remove trace Cl2 , then absorbed in distilled water. Stock solutions of ClO2 were stored in darkness at 5◦ C. The ...
... 2.1. Materials. Chlorine dioxide aqueous solution was prepared from sodium chlorite and diluted sulfuric acid and was purified by bubbling through 10% sodium chlorite aqueous solution to remove trace Cl2 , then absorbed in distilled water. Stock solutions of ClO2 were stored in darkness at 5◦ C. The ...
Chapter Four - Salina USD 305
... • Write formulas for the reactants and predicted products for the chemical reactions that follow: 1. Solid calcium carbonate is strongly heated in a test tube. ...
... • Write formulas for the reactants and predicted products for the chemical reactions that follow: 1. Solid calcium carbonate is strongly heated in a test tube. ...
Document
... • so to describe solutions accurately, we must describe how much of each component is present we saw that with pure substances, we can describe them with a single name because all samples identical Tro, Chemistry: A Molecular Approach ...
... • so to describe solutions accurately, we must describe how much of each component is present we saw that with pure substances, we can describe them with a single name because all samples identical Tro, Chemistry: A Molecular Approach ...
Chapter 4 Chemical Quantities and Aqueous Reactions
... • so to describe solutions accurately, we must describe how much of each component is present we saw that with pure substances, we can describe them with a single name because all samples identical Tro, Chemistry: A Molecular Approach ...
... • so to describe solutions accurately, we must describe how much of each component is present we saw that with pure substances, we can describe them with a single name because all samples identical Tro, Chemistry: A Molecular Approach ...
TRO Chapter 4
... • so to describe solutions accurately, we must describe how much of each component is present we saw that with pure substances, we can describe them with a single name because all samples identical Tro, Chemistry: A Molecular Approach ...
... • so to describe solutions accurately, we must describe how much of each component is present we saw that with pure substances, we can describe them with a single name because all samples identical Tro, Chemistry: A Molecular Approach ...
TOPIC 11 Further equilibrium 11.1 Chemical equilibrium
... If the pressure is increased then the volume of the reaction mixture will decrease. This will result in an instantaneous equal increase in the concentration of each component. However, the increase in the magnitude of [CH3OH(g)][H2(g)]2 will be greater than the increase in the magnitude of [CH3OH(g) ...
... If the pressure is increased then the volume of the reaction mixture will decrease. This will result in an instantaneous equal increase in the concentration of each component. However, the increase in the magnitude of [CH3OH(g)][H2(g)]2 will be greater than the increase in the magnitude of [CH3OH(g) ...
Chemistry
... – for his development of nuclear magnetic resonance spectroscopy for determining the three-dimensional structure of biological macromolecules in solution ...
... – for his development of nuclear magnetic resonance spectroscopy for determining the three-dimensional structure of biological macromolecules in solution ...
Chemistry 11 - Correspondence Studies
... much product will be formed? This unit will answer these questions and other questions related to amount of matter. The word stoichiometry comes from the Greek words, stoicheion (meaning any first thing or principle) and metron (meaning measure). Stoichiometry deals with the mass-mass or molemole re ...
... much product will be formed? This unit will answer these questions and other questions related to amount of matter. The word stoichiometry comes from the Greek words, stoicheion (meaning any first thing or principle) and metron (meaning measure). Stoichiometry deals with the mass-mass or molemole re ...
- Academy Test Bank
... 16. Monodisperse polyacrylonitrile contains molecules with the general formula -(CH2CHCN)n-, where n is typically greater than 10,000. Given that a sample of monodisperse polyacrilonitrile weighs 197.4 g and contains 1.046 10 20 molecules of -(CH2CHCN)n-, calculate n. A) 2.141 10 4 B) 6.026 1 ...
... 16. Monodisperse polyacrylonitrile contains molecules with the general formula -(CH2CHCN)n-, where n is typically greater than 10,000. Given that a sample of monodisperse polyacrilonitrile weighs 197.4 g and contains 1.046 10 20 molecules of -(CH2CHCN)n-, calculate n. A) 2.141 10 4 B) 6.026 1 ...
Support Material
... Doping is the process of increasing the conductivity of intrinsic semiconductors by adding an appropriate amount of suitable impurity in Si or Ge. * n-type semiconductors : Silicon or Germinium (group-14) doped with electron rich impurity (group-15 element like P or As), Here conductivity is due to ...
... Doping is the process of increasing the conductivity of intrinsic semiconductors by adding an appropriate amount of suitable impurity in Si or Ge. * n-type semiconductors : Silicon or Germinium (group-14) doped with electron rich impurity (group-15 element like P or As), Here conductivity is due to ...
Document
... Any property that only depends on object’s current state or condition Independence from method, path or mechanism by which change occurs is important feature of all state functions Some State functions, E, P, t, and V : ...
... Any property that only depends on object’s current state or condition Independence from method, path or mechanism by which change occurs is important feature of all state functions Some State functions, E, P, t, and V : ...
Document
... disappear, or become a liquid – the mixture is homogeneous the salt is still there, as you can tell from the taste, or simply boiling away the water ...
... disappear, or become a liquid – the mixture is homogeneous the salt is still there, as you can tell from the taste, or simply boiling away the water ...
Analytical chemistry

Analytical chemistry is the study of the separation, identification, and quantification of the chemical components of natural and artificial materials. Qualitative analysis gives an indication of the identity of the chemical species in the sample, and quantitative analysis determines the amount of certain components in the substance. The separation of components is often performed prior to analysis.Analytical methods can be separated into classical and instrumental. Classical methods (also known as wet chemistry methods) use separations such as precipitation, extraction, and distillation and qualitative analysis by color, odor, or melting point. Classical quantitative analysis is achieved by measurement of weight or volume. Instrumental methods use an apparatus to measure physical quantities of the analyte such as light absorption, fluorescence, or conductivity. The separation of materials is accomplished using chromatography, electrophoresis or field flow fractionation methods.Analytical chemistry is also focused on improvements in experimental design, chemometrics, and the creation of new measurement tools to provide better chemical information. Analytical chemistry has applications in forensics, bioanalysis, clinical analysis, environmental analysis, and materials analysis.























