Chem 171 Review - Exam 1
... atoms and atomic structure early experiments and scientists that established our modern understanding of the atom and its structure protons, electrons, neutrons - know the characteristics of these particles including relative masses and electrical charge atomic structure - where do the various subat ...
... atoms and atomic structure early experiments and scientists that established our modern understanding of the atom and its structure protons, electrons, neutrons - know the characteristics of these particles including relative masses and electrical charge atomic structure - where do the various subat ...
Chapter 3 Atoms and the Periodic Table
... – electrons are going in the s orbital – Group 1 • H down to Fr • only one electron in the outer most energy level • 1 electron in the s orbital • these elements have 1 valence electron – Group 2 • Be down to Ra • 2 electrons in the outer most energy level • 2 electrons in the s orbital and it is no ...
... – electrons are going in the s orbital – Group 1 • H down to Fr • only one electron in the outer most energy level • 1 electron in the s orbital • these elements have 1 valence electron – Group 2 • Be down to Ra • 2 electrons in the outer most energy level • 2 electrons in the s orbital and it is no ...
National 4/5 Chemistry Homework
... o A chemical reaction always results in a new substance being formed o A chemical reaction can be shown by a change in appearance of a substance o A chemical reaction can be shown by a detectable energy change o A chemical reaction can be shown by precipitation (a solid forming in a solution) o A ch ...
... o A chemical reaction always results in a new substance being formed o A chemical reaction can be shown by a change in appearance of a substance o A chemical reaction can be shown by a detectable energy change o A chemical reaction can be shown by precipitation (a solid forming in a solution) o A ch ...
Chapter 2. Atoms, Molecules, and Ions
... The radiation is passed between two electrically charged plates and detected. Three spots are observed on the detector: 1. a spot deflected in the direction of the positive plate, 2. a spot that is not affected by the electric field, and 3. a spot deflected in the direction of the negative plate. A ...
... The radiation is passed between two electrically charged plates and detected. Three spots are observed on the detector: 1. a spot deflected in the direction of the positive plate, 2. a spot that is not affected by the electric field, and 3. a spot deflected in the direction of the negative plate. A ...
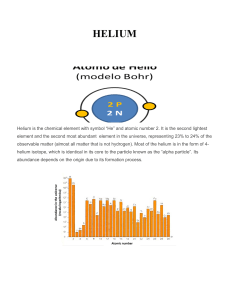
HELIUM - IDC
... Helium is the chemical element with symbol “He” and atomic number 2. It is the second lightest element and the second most abundant element in the universe, representing 23% to 24% of the observable matter (almost all matter that is not hydrogen). Most of the helium is in the form of 4helium isotope ...
... Helium is the chemical element with symbol “He” and atomic number 2. It is the second lightest element and the second most abundant element in the universe, representing 23% to 24% of the observable matter (almost all matter that is not hydrogen). Most of the helium is in the form of 4helium isotope ...
Chemistry(Class-IX)- Atoms and Molecules
... Several elements are named after the place where they discovered, such as ‘Copper’ which was taken from Cyprus. Some elements are named after their colour, such as ‘Gold’ which means yellow. Atoms and Molecules ...
... Several elements are named after the place where they discovered, such as ‘Copper’ which was taken from Cyprus. Some elements are named after their colour, such as ‘Gold’ which means yellow. Atoms and Molecules ...
CHAPTER 8: CHEMICAL COMPOSITION
... then the (weighted average) atomic mass of carbon is calculated as follows: (12.000000 amu)(0.9893) + (13.003354 amu)(0.0107) = 12.01 amu So how many carbon atoms are present in 12.01 grams of carbon? This number was determined experimentally to be 6.022×1023. – It was named Avogadro’s number, to ho ...
... then the (weighted average) atomic mass of carbon is calculated as follows: (12.000000 amu)(0.9893) + (13.003354 amu)(0.0107) = 12.01 amu So how many carbon atoms are present in 12.01 grams of carbon? This number was determined experimentally to be 6.022×1023. – It was named Avogadro’s number, to ho ...
Chapter 4 Elements and the Periodic Table
... neutrons are about equal in mass. An electron has about 1/2,000 the mass of a proton or neutron. ...
... neutrons are about equal in mass. An electron has about 1/2,000 the mass of a proton or neutron. ...
Chapter 4 Elements and the Periodic Table
... neutrons are about equal in mass. An electron has about 1/2,000 the mass of a proton or neutron. ...
... neutrons are about equal in mass. An electron has about 1/2,000 the mass of a proton or neutron. ...
2.2 the observations that led to an atomic view of matter
... Table 2.1 shows a striking example: soft, silvery sodium metal and yellowgreen, poisonous chlorine gas are very different from the compound they form—white, crystalline sodium chloride, or common table salt! Figure 2.1 Elements, compounds, and mixtures on the atomic scale. The samples depicted here ...
... Table 2.1 shows a striking example: soft, silvery sodium metal and yellowgreen, poisonous chlorine gas are very different from the compound they form—white, crystalline sodium chloride, or common table salt! Figure 2.1 Elements, compounds, and mixtures on the atomic scale. The samples depicted here ...
Matter is anything that has mass and occupies space. Three
... When gases combine or are produced in a chemical reaction, they do so in a simple ratio by volume, provided all the gases are at the same temperature and pressure. Avogadro Law: Equal volumes of gases at the same temperature and pressure should contain equal number of molecules. Dalton’s Atomic Theo ...
... When gases combine or are produced in a chemical reaction, they do so in a simple ratio by volume, provided all the gases are at the same temperature and pressure. Avogadro Law: Equal volumes of gases at the same temperature and pressure should contain equal number of molecules. Dalton’s Atomic Theo ...
UNIT 3 - ATOMS 1 HISTORY OF ATOMIC THEORY NOTES I
... When we want to know how many atoms of a substance are in a sample of the substance that we can see, counting the atoms individually is not practical. First of all, atoms are so small that we cannot see them individually with our eyes. Second, we would be counting for a very, very, very long time. H ...
... When we want to know how many atoms of a substance are in a sample of the substance that we can see, counting the atoms individually is not practical. First of all, atoms are so small that we cannot see them individually with our eyes. Second, we would be counting for a very, very, very long time. H ...
Chapter 4 Notes
... Atomic Number – number of p+ in the nucleus of an atom (always equal to number of e-) Mass Number – number of p+ and n0 in the nucleus of an atom Atomic Weight (mass) – the average mass of the isotopes The mass number is just the atomic weight rounded off to a whole number!! Atomic Weight ...
... Atomic Number – number of p+ in the nucleus of an atom (always equal to number of e-) Mass Number – number of p+ and n0 in the nucleus of an atom Atomic Weight (mass) – the average mass of the isotopes The mass number is just the atomic weight rounded off to a whole number!! Atomic Weight ...
Dalton`s Atomic Theory
... 2. All atoms of the same element are exactly alike; in particular, they all have the same mass. 3. Atoms of different elements are different; in particular, they have different masses. 4. Compounds are formed by the joining of atoms of two or more elements. They are joined in a definite whole-number ...
... 2. All atoms of the same element are exactly alike; in particular, they all have the same mass. 3. Atoms of different elements are different; in particular, they have different masses. 4. Compounds are formed by the joining of atoms of two or more elements. They are joined in a definite whole-number ...
5 - Particles in an atom
... Substances that contain only one kind of atom are called elements. Some familiar elements are oxygen, gold, silver, and helium. An atom is the smallest part of an element that can be broken down and still have the characteristics of that element. All atoms are basically the same. All atoms of the sa ...
... Substances that contain only one kind of atom are called elements. Some familiar elements are oxygen, gold, silver, and helium. An atom is the smallest part of an element that can be broken down and still have the characteristics of that element. All atoms are basically the same. All atoms of the sa ...
2 The Atom - Mr. Krohn 8th grade science
... Neutrons are particles in the nucleus of an atom that do not have an electric charge. A neutron has a little more mass than a proton does. However, the difference in mass is so small that the mass of a neutron can be thought of ...
... Neutrons are particles in the nucleus of an atom that do not have an electric charge. A neutron has a little more mass than a proton does. However, the difference in mass is so small that the mass of a neutron can be thought of ...
Lesson 4: Atomic Structure
... compounds. His work led him to discover that the amount of electricity applied to a sample compound is related to the amount of an element that is isolated. He concluded that the structure of the atom must play a role in this. Building on Faraday's experiments, English physicist J. J. Thompson disco ...
... compounds. His work led him to discover that the amount of electricity applied to a sample compound is related to the amount of an element that is isolated. He concluded that the structure of the atom must play a role in this. Building on Faraday's experiments, English physicist J. J. Thompson disco ...
Ch 01 notes
... • A second common notation for isotopes is the chemical symbol (or chemical name) followed by a dash and the mass number of the isotope. ...
... • A second common notation for isotopes is the chemical symbol (or chemical name) followed by a dash and the mass number of the isotope. ...
Atomic Theory Quiz A
... 4. What isotope is the most common isotope of Cadmium? On the periodic table, cadmium’s average weighted atomic mass is 112.41 amu, which rounds to 114. That’s the most common isotope, Cd-114 5. Is Sr-87 the most common isotope of strontium? Is Sr-88 the most common isotope of strontium? How can you ...
... 4. What isotope is the most common isotope of Cadmium? On the periodic table, cadmium’s average weighted atomic mass is 112.41 amu, which rounds to 114. That’s the most common isotope, Cd-114 5. Is Sr-87 the most common isotope of strontium? Is Sr-88 the most common isotope of strontium? How can you ...
LESSON PLAN School : State Senior High School ……………… The
... b. From high energy to lower level is called de-excitation, and it release energy. Proton is positively charged particle which was discovered by Eugene Goldstein by continuation rays experiment. ...
... b. From high energy to lower level is called de-excitation, and it release energy. Proton is positively charged particle which was discovered by Eugene Goldstein by continuation rays experiment. ...
atom - Social Circle City Schools
... determine the charge-to-mass ratio of a charged particle, then compared it to known values. • The mass of the charged particle was much less than a hydrogen atom, then the lightest known atom. • Thomson received the Nobel Prize in 1906 for identifying the first subatomic particle—the electron ...
... determine the charge-to-mass ratio of a charged particle, then compared it to known values. • The mass of the charged particle was much less than a hydrogen atom, then the lightest known atom. • Thomson received the Nobel Prize in 1906 for identifying the first subatomic particle—the electron ...
atom
... • Nuclear reactions can change one element into another element. • In the late 1890s, scientists noticed some substances spontaneously emitted radiation, a process they called radioactivity. • The rays and particles emitted are called radiation. • A reaction that involves a change in an atom's nucle ...
... • Nuclear reactions can change one element into another element. • In the late 1890s, scientists noticed some substances spontaneously emitted radiation, a process they called radioactivity. • The rays and particles emitted are called radiation. • A reaction that involves a change in an atom's nucle ...
Unit 2: Atomic Concepts and Periodic Table (Level 1)
... o Some elements exist as two or more forms in the same phase. These forms differ in their molecular or crystal structure and therefore in their properties. The word to describe this phenomenon is ALLOTROPE. o Ozone and oxygen gases are allotropes of each other. Ozone is O 3 and it is very dangerous ...
... o Some elements exist as two or more forms in the same phase. These forms differ in their molecular or crystal structure and therefore in their properties. The word to describe this phenomenon is ALLOTROPE. o Ozone and oxygen gases are allotropes of each other. Ozone is O 3 and it is very dangerous ...
Chemical element
A chemical element (or element) is a chemical substance consisting of atoms having the same number of protons in their atomic nuclei (i.e. the same atomic number, Z). There are 118 elements that have been identified, of which the first 94 occur naturally on Earth with the remaining 24 being synthetic elements. There are 80 elements that have at least one stable isotope and 38 that have exclusively radioactive isotopes, which decay over time into other elements. Iron is the most abundant element (by mass) making up the Earth, while oxygen is the most common element in the crust of the earth.Chemical elements constitute approximately 15% of the matter in the universe: the remainder is dark matter, the composition of it is unknown, but it is not composed of chemical elements.The two lightest elements, hydrogen and helium were mostly formed in the Big Bang and are the most common elements in the universe. The next three elements (lithium, beryllium and boron) were formed mostly by cosmic ray spallation, and are thus more rare than those that follow. Formation of elements with from six to twenty six protons occurred and continues to occur in main sequence stars via stellar nucleosynthesis. The high abundance of oxygen, silicon, and iron on Earth reflects their common production in such stars. Elements with greater than twenty six protons are formed by supernova nucleosynthesis in supernovae, which, when they explode, blast these elements far into space as planetary nebulae, where they may become incorporated into planets when they are formed.When different elements are chemically combined, with the atoms held together by chemical bonds, they form chemical compounds. Only a minority of elements are found uncombined as relatively pure minerals. Among the more common of such ""native elements"" are copper, silver, gold, carbon (as coal, graphite, or diamonds), and sulfur. All but a few of the most inert elements, such as noble gases and noble metals, are usually found on Earth in chemically combined form, as chemical compounds. While about 32 of the chemical elements occur on Earth in native uncombined forms, most of these occur as mixtures. For example, atmospheric air is primarily a mixture of nitrogen, oxygen, and argon, and native solid elements occur in alloys, such as that of iron and nickel.The history of the discovery and use of the elements began with primitive human societies that found native elements like carbon, sulfur, copper and gold. Later civilizations extracted elemental copper, tin, lead and iron from their ores by smelting, using charcoal. Alchemists and chemists subsequently identified many more, with almost all of the naturally-occurring elements becoming known by 1900. The properties of the chemical elements are summarized on the periodic table, which organizes the elements by increasing atomic number into rows (""periods"") in which the columns (""groups"") share recurring (""periodic"") physical and chemical properties. Save for unstable radioactive elements with short half-lives, all of the elements are available industrially, most of them in high degrees of purity.