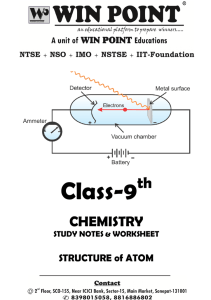
Review Study Guide for the Final
... To what volume, in liters, must you dilute a solution containing 4 liters of 0.100 M of Ca(OH)2 to obtain a 0.00100 M solution as calcium hydroxide (Ca(OH)2)? ...
... To what volume, in liters, must you dilute a solution containing 4 liters of 0.100 M of Ca(OH)2 to obtain a 0.00100 M solution as calcium hydroxide (Ca(OH)2)? ...
inorganic chemistry
... compared to chlorine is due to very small size of the fluorine atom. As a result, there are strong inter-electronic repulsions in the relatively small 2p subshell of fluorine and thus the incoming electron does not feel much attraction. Therefore, its electron affinity is small. Thus, electron affin ...
... compared to chlorine is due to very small size of the fluorine atom. As a result, there are strong inter-electronic repulsions in the relatively small 2p subshell of fluorine and thus the incoming electron does not feel much attraction. Therefore, its electron affinity is small. Thus, electron affin ...
Atoms and bonds in molecules and chemical
... In a second approach, no more than interpretative postulates is required and no approximation is considered in the principles. The key is then to import a mathematical method of analysis to perform the partition. Theories derived accordingly should (Aslangul et al., 1972) “provide the mathematical b ...
... In a second approach, no more than interpretative postulates is required and no approximation is considered in the principles. The key is then to import a mathematical method of analysis to perform the partition. Theories derived accordingly should (Aslangul et al., 1972) “provide the mathematical b ...
Chapter 2 – Atoms, Ions, and the Periodic Table
... chemical reactions, mass must be conserved. If elements could be changed into other elements during chemical reactions (as the alchemists were trying to do), then masses of atoms would change during reactions and mass would not be conserved. ...
... chemical reactions, mass must be conserved. If elements could be changed into other elements during chemical reactions (as the alchemists were trying to do), then masses of atoms would change during reactions and mass would not be conserved. ...
FREE Sample Here
... Full file at http://testbank360.eu/test-bank-chemistry-atoms-first-2nd-edition-burdge ...
... Full file at http://testbank360.eu/test-bank-chemistry-atoms-first-2nd-edition-burdge ...
Introduction to Atoms - Mrs Gillum`s Web Page!
... An atom is the smallest particle into which an element can be divided and still be the same substance. Atoms make up elements; elements combine to form compounds. Because all matter is made of elements or compounds, atoms are often called the building blocks of matter. Before the scanning tunneling ...
... An atom is the smallest particle into which an element can be divided and still be the same substance. Atoms make up elements; elements combine to form compounds. Because all matter is made of elements or compounds, atoms are often called the building blocks of matter. Before the scanning tunneling ...
File
... (a) An atom is made up of three particles: electrons, protons and neutrons. Electrons have negative charge, protons have positive charge whereas neutrons have no charge, they are neutral. Due to the presence of equal number of negative electrons and positive protons, the atom on the whole is electri ...
... (a) An atom is made up of three particles: electrons, protons and neutrons. Electrons have negative charge, protons have positive charge whereas neutrons have no charge, they are neutral. Due to the presence of equal number of negative electrons and positive protons, the atom on the whole is electri ...
Chemistry - Summative Practice and Review for Chapter 4 and 5
... c. relative abundance of protons in that element d. mass and relative abundance of each isotope of that element ____ 33. How are the frequency and wavelength of light related? a. They are inversely proportional to each other. b. Frequency equals wavelength divided by the speed of light. c. Waveleng ...
... c. relative abundance of protons in that element d. mass and relative abundance of each isotope of that element ____ 33. How are the frequency and wavelength of light related? a. They are inversely proportional to each other. b. Frequency equals wavelength divided by the speed of light. c. Waveleng ...
1 Atomic Orbital Theory
... The detailed study of the structure of atoms (as distinguished from molecules) is largely the domain of the physicist. With respect to atomic structure, the interest of the chemist is usually confined to the behavior and properties of the three fundamental particles of atoms, namely the electron, th ...
... The detailed study of the structure of atoms (as distinguished from molecules) is largely the domain of the physicist. With respect to atomic structure, the interest of the chemist is usually confined to the behavior and properties of the three fundamental particles of atoms, namely the electron, th ...
Review Unit: Chemistry Review
... simplest way possible. Scientists refine the descriptions of the natural world so that these descriptions are as precise and complete as possible. In science, reliable and accurate descriptions of phenomena become scientific laws. In scientific problem solving, descriptions, predictions, and explana ...
... simplest way possible. Scientists refine the descriptions of the natural world so that these descriptions are as precise and complete as possible. In science, reliable and accurate descriptions of phenomena become scientific laws. In scientific problem solving, descriptions, predictions, and explana ...
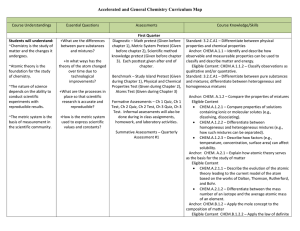
Chemistry Curriculum Map - Belle Vernon Area School District
... for the first twenty elements of the periodic table. Anchor: CHEM.A.2.2 – Describe the behavior of electrons in atoms. Eligible Content: CHEM.A.2.2.1 – Predict the ground state electronic configurations and/or orbital diagram for a given atom or ion. Standard: 3.2.C.A2 – Relate the position of an el ...
... for the first twenty elements of the periodic table. Anchor: CHEM.A.2.2 – Describe the behavior of electrons in atoms. Eligible Content: CHEM.A.2.2.1 – Predict the ground state electronic configurations and/or orbital diagram for a given atom or ion. Standard: 3.2.C.A2 – Relate the position of an el ...
Learning Outcomes
... (b) describe, with the aid of diagrams, the structure of an atom as containing protons and neutrons (nucleons) in the nucleus and electrons arranged in shells (energy levels) (Knowledge of s, p, d and f classification is not required; a copy of the Periodic Table will be available in Papers 1 and 2) ...
... (b) describe, with the aid of diagrams, the structure of an atom as containing protons and neutrons (nucleons) in the nucleus and electrons arranged in shells (energy levels) (Knowledge of s, p, d and f classification is not required; a copy of the Periodic Table will be available in Papers 1 and 2) ...
Chapter 2 - Atoms and the Periodic Table (test bank)
... A. Odd number of neutrons and odd number of protons B. Even number of neutrons and odd number of protons C. Odd number of neutrons and even number of protons D. Even number of neutrons and even number of protons E. None of the options above results in significantly more stable nuclei. ...
... A. Odd number of neutrons and odd number of protons B. Even number of neutrons and odd number of protons C. Odd number of neutrons and even number of protons D. Even number of neutrons and even number of protons E. None of the options above results in significantly more stable nuclei. ...
Chapter 2 Atoms, Molecules, and Ions
... Ø If two elements, A and B, form more than one compound, the masses of B that combine with a given mass of A are in the ratio of small whole numbers. Ø Dalton predicted this law and observed it while developing his atomic theory. Ø When two or more compounds exist from the same elements, they can ...
... Ø If two elements, A and B, form more than one compound, the masses of B that combine with a given mass of A are in the ratio of small whole numbers. Ø Dalton predicted this law and observed it while developing his atomic theory. Ø When two or more compounds exist from the same elements, they can ...
Surviving Chemistry - Bremen High School District 228
... For example, there are a few different atoms of the element Lithium. All atoms of Lithium contain the same number of protons in their nucleus. The difference between these atoms is the number of neutrons. Since all Lithium atoms have the same number of protons (3), they all have the same atomic numb ...
... For example, there are a few different atoms of the element Lithium. All atoms of Lithium contain the same number of protons in their nucleus. The difference between these atoms is the number of neutrons. Since all Lithium atoms have the same number of protons (3), they all have the same atomic numb ...
Final Exam Review Notes
... Postulate from John Dalton’s Model of the Atom 1. All matter is composed of indivisible atoms. 2. An element is composed of only one type of atom – All atoms of one type of element always behave the same way. – Atoms of different elements do not behave the same way. 3. Two or more elements combine t ...
... Postulate from John Dalton’s Model of the Atom 1. All matter is composed of indivisible atoms. 2. An element is composed of only one type of atom – All atoms of one type of element always behave the same way. – Atoms of different elements do not behave the same way. 3. Two or more elements combine t ...
Chapter 3
... Foundations of Atomic Theory, continued • Law of definite proportions: a chemical compound contains the same elements in exactly the same proportions by mass regardless of the size of the sample or source of the compound • Law of multiple proportions: if two or more different compounds are composed ...
... Foundations of Atomic Theory, continued • Law of definite proportions: a chemical compound contains the same elements in exactly the same proportions by mass regardless of the size of the sample or source of the compound • Law of multiple proportions: if two or more different compounds are composed ...
Chapter 3
... Foundations of Atomic Theory, continued • Law of definite proportions: a chemical compound contains the same elements in exactly the same proportions by mass regardless of the size of the sample or source of the compound • Law of multiple proportions: if two or more different compounds are composed ...
... Foundations of Atomic Theory, continued • Law of definite proportions: a chemical compound contains the same elements in exactly the same proportions by mass regardless of the size of the sample or source of the compound • Law of multiple proportions: if two or more different compounds are composed ...
Chemistry: A Molecular Approach
... in its nucleus • the number of protons in the nucleus of an atom is called the atomic number the elements are arranged on the Periodic Table in order of their atomic numbers ...
... in its nucleus • the number of protons in the nucleus of an atom is called the atomic number the elements are arranged on the Periodic Table in order of their atomic numbers ...
DCY1B - Manonmaniam Sundaranar University
... high tensile strength. They are good conductors of heat and electricity (iii) Melting and Boiling points: They have high melting and boiling points except Zn, Cd and Hg. The low melting point of Zn, Cd and Hg may be attributed to the completely filled d-level. (iv) Atomic (covalent) radii: Atomic ra ...
... high tensile strength. They are good conductors of heat and electricity (iii) Melting and Boiling points: They have high melting and boiling points except Zn, Cd and Hg. The low melting point of Zn, Cd and Hg may be attributed to the completely filled d-level. (iv) Atomic (covalent) radii: Atomic ra ...
US Army medical course General Chemistry
... wall may be constructed from a basic unit, the brick. In trying to find this basic unit, they separated matter by all the methods (chemical and physical) available to them until they could not separate it any further. They felt this separation must result in the building block of matter, which they ...
... wall may be constructed from a basic unit, the brick. In trying to find this basic unit, they separated matter by all the methods (chemical and physical) available to them until they could not separate it any further. They felt this separation must result in the building block of matter, which they ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.