CLASS X carbon and its compound
... Ethanol gets dehydrated to ethene when heated with conc. sulphuric acid at 443 K (170°C). Methylated spirit is ethanol in which a small amount of methanol is mixed. This makes it unfit for drinking purposes, but is extensively used in paints and varnish industry. Pure ethanoic acid is a corrosive co ...
... Ethanol gets dehydrated to ethene when heated with conc. sulphuric acid at 443 K (170°C). Methylated spirit is ethanol in which a small amount of methanol is mixed. This makes it unfit for drinking purposes, but is extensively used in paints and varnish industry. Pure ethanoic acid is a corrosive co ...
AP Chemistry Review Packet 1 CO2(g) + H2(g) « H2O(g) + CO(g
... (c) Without performing calculations, describe how the resulting data should be used to obtain the standard molar enthalpy of neutralization. (d) When a class of students performed this experiment, the average of the results was -55.0 kilojoules per mole. The accepted value for the standard molar ent ...
... (c) Without performing calculations, describe how the resulting data should be used to obtain the standard molar enthalpy of neutralization. (d) When a class of students performed this experiment, the average of the results was -55.0 kilojoules per mole. The accepted value for the standard molar ent ...
Chemistry Review2
... will take the H+. Mixing acid and bases will create water and a salt. Sometimes acids and bases are reactants, conjugate bases and conjugate acids are products. HCO3-(aq) + H20(l) CO3 -2 (aq) + H3O+ (aq), Identify the acid, base, conjugate acid and conjugate base. Acids and Bases are on a pH scale ...
... will take the H+. Mixing acid and bases will create water and a salt. Sometimes acids and bases are reactants, conjugate bases and conjugate acids are products. HCO3-(aq) + H20(l) CO3 -2 (aq) + H3O+ (aq), Identify the acid, base, conjugate acid and conjugate base. Acids and Bases are on a pH scale ...
R - MSU Chemistry
... The starting material is C8H8O2 so A has an extra C7H4O. This looks like the addition of PhCOCl with the loss of HCl. The most obvious reaction is acylation of the phenolic oxygen rather th ...
... The starting material is C8H8O2 so A has an extra C7H4O. This looks like the addition of PhCOCl with the loss of HCl. The most obvious reaction is acylation of the phenolic oxygen rather th ...
Chemistry - StudyTime NZ
... Both atoms have the same atomic number. Because different elements are defined by their atomic number, we can say that both isotopes are the element Carbon. This means they have the same number of p ...
... Both atoms have the same atomic number. Because different elements are defined by their atomic number, we can say that both isotopes are the element Carbon. This means they have the same number of p ...
FINAL EXAM Spring 2012
... The last page of this examination is a periodic table [Gas constant = 8.314 J/mol K; 0.08206 L*atm/mole*K,1 faraday = 96500 J/V mol e-; at 25oC] 1) The reaction has the rate law, Rate = k[A][B]2. Which will cause the rate to increase the most? A) doubling [A] B) doubling [B] C) tripling [B] D) quadr ...
... The last page of this examination is a periodic table [Gas constant = 8.314 J/mol K; 0.08206 L*atm/mole*K,1 faraday = 96500 J/V mol e-; at 25oC] 1) The reaction has the rate law, Rate = k[A][B]2. Which will cause the rate to increase the most? A) doubling [A] B) doubling [B] C) tripling [B] D) quadr ...
Practice Exam #2
... 19) Which one of the following conditions would always result in an increase in the internal energy of a system? A) The system loses heat and has work done on it by the surroundings. B) The system loses heat and does work on the surroundings. C) The system gains heat and does work on the surrounding ...
... 19) Which one of the following conditions would always result in an increase in the internal energy of a system? A) The system loses heat and has work done on it by the surroundings. B) The system loses heat and does work on the surroundings. C) The system gains heat and does work on the surrounding ...
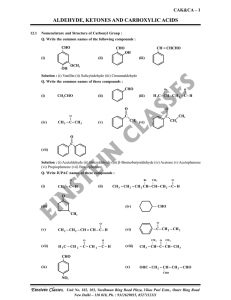
aldehyde, ketones and carboxylic acids
... approximately perpendicular to the plane of sp2 hybridised orbitals of carbonyl carbon. The hybridisation of carbon changes from sp2 to sp3 in this process, and a tetrahedral alkoxide intermediate is produced. This intermediate captures a proton from the reaction medium to give the electrically neut ...
... approximately perpendicular to the plane of sp2 hybridised orbitals of carbonyl carbon. The hybridisation of carbon changes from sp2 to sp3 in this process, and a tetrahedral alkoxide intermediate is produced. This intermediate captures a proton from the reaction medium to give the electrically neut ...
organic sample test
... carboxylic acids can be reacted with alcohols selected to make esters which do not have foul odours and are inviting to bacteria which will consume them react the carboxylic acids with a base to make an insoluble salt which can be removed from the bottom of the lagoon, and the clear water on top can ...
... carboxylic acids can be reacted with alcohols selected to make esters which do not have foul odours and are inviting to bacteria which will consume them react the carboxylic acids with a base to make an insoluble salt which can be removed from the bottom of the lagoon, and the clear water on top can ...
Chemistry 40S – Exam Review
... 15. The [OH-] in a solution is 1.0 x 10-4 mol/L. What is the pH of the solution? 16. What is the [H3O+] in a 0.20 mol/L NaOH solution? 17. A solution has a [H+] of 0.0010 M. What is the [OH-]? 18. What is the [H3O+] in a 0.020 M Ba(OH)2(aq) solution? 19. A base added to a neutral solution will: a) ...
... 15. The [OH-] in a solution is 1.0 x 10-4 mol/L. What is the pH of the solution? 16. What is the [H3O+] in a 0.20 mol/L NaOH solution? 17. A solution has a [H+] of 0.0010 M. What is the [OH-]? 18. What is the [H3O+] in a 0.020 M Ba(OH)2(aq) solution? 19. A base added to a neutral solution will: a) ...
inorganic-chemistry-gp-i-alkali-metals
... The colour of the superoxide’s is due to the paramagnetic behaviour, the O2- is having two covalent bonds and a single electron, which when move from one to other atom releases photon of visible range giving the compounds colour, and also the paramagnetic behaviour The stability of peroxides and s ...
... The colour of the superoxide’s is due to the paramagnetic behaviour, the O2- is having two covalent bonds and a single electron, which when move from one to other atom releases photon of visible range giving the compounds colour, and also the paramagnetic behaviour The stability of peroxides and s ...
CHEMISTRY
... Recall – compounds are pure substances that contain more than one type of element that are bonded together in a fixed ratio. Water (H2O) is an example of a compound. In water, the two hydrogen atoms are stuck to the oxygen and can’t be separated without undergoing a chemical change. There are two ty ...
... Recall – compounds are pure substances that contain more than one type of element that are bonded together in a fixed ratio. Water (H2O) is an example of a compound. In water, the two hydrogen atoms are stuck to the oxygen and can’t be separated without undergoing a chemical change. There are two ty ...
Alcohols and Phenols
... 1H NMR: electron-withdrawing effect of the nearby oxygen, absorbs at 3.5 to 4 (See Figure 17-13) ...
... 1H NMR: electron-withdrawing effect of the nearby oxygen, absorbs at 3.5 to 4 (See Figure 17-13) ...
Fundamentals of General Chemistry and Physical Chemistry for
... We can see that the reaction between Pb(NO3)2 and KI is really a reaction between ions, and can be approximately referred to as an ionic reaction. ...
... We can see that the reaction between Pb(NO3)2 and KI is really a reaction between ions, and can be approximately referred to as an ionic reaction. ...
An Overview of Organic Reactions
... transition-state structure for the first step The π bond between carbons begins to break § The C–H bond begins to form § The H–Br bond begins to break ...
... transition-state structure for the first step The π bond between carbons begins to break § The C–H bond begins to form § The H–Br bond begins to break ...
- University at Albany
... ethoxide ion is a stronger base than t-butoxide ion). Also, alkyl halide reactivity decreases from methyl to 10 to 20 to 30. In fact, 30 alkyl halides do not react by SN2. Leaving group: The substrate should have a good leaving group. A good leaving group should be electron withdrawing, relatively s ...
... ethoxide ion is a stronger base than t-butoxide ion). Also, alkyl halide reactivity decreases from methyl to 10 to 20 to 30. In fact, 30 alkyl halides do not react by SN2. Leaving group: The substrate should have a good leaving group. A good leaving group should be electron withdrawing, relatively s ...
8 - THE DETERMINATION OF THE CONCENTRATION
... equivalence point is less than 0.10 mL. Neutralize all titrant solutions with citric acid until the pH paper turns light green or orange before discarding the solution in the drain. Pour the small amount of waste NaOH used to rinse the burette into the labeled waste container. Do not pour un-neutral ...
... equivalence point is less than 0.10 mL. Neutralize all titrant solutions with citric acid until the pH paper turns light green or orange before discarding the solution in the drain. Pour the small amount of waste NaOH used to rinse the burette into the labeled waste container. Do not pour un-neutral ...
2016 Pre Course CHEMISTRY - Calday Grange Grammar School
... Diamond and graphite are both forms of carbon. Diamond is able to scratch almost all other substances, whereas graphite may be used as a lubricant. Diamond and graphite both have high melting points. Explain each of these properties of diamond and graphite in terms of structure and bonding. Give one ...
... Diamond and graphite are both forms of carbon. Diamond is able to scratch almost all other substances, whereas graphite may be used as a lubricant. Diamond and graphite both have high melting points. Explain each of these properties of diamond and graphite in terms of structure and bonding. Give one ...
AGE article for Sept 2013
... 63.6 kJ/L at SLC (25˚C and 1 atm). Are you able to use data from the Data Book to confirm these values? Note that they are only given to 3 significant figures; do you know why? The confusion may arise because of the way the data is presented in the Data Book. Heats of Combustion are given in the fo ...
... 63.6 kJ/L at SLC (25˚C and 1 atm). Are you able to use data from the Data Book to confirm these values? Note that they are only given to 3 significant figures; do you know why? The confusion may arise because of the way the data is presented in the Data Book. Heats of Combustion are given in the fo ...
chapter 4 review: types of chemical reactions and solution
... 3. Identify the precipitate formed in each of the following reactions: (a) potassium chloride (aq) + lead (II) nitrate (aq) (b) silver nitrate (aq) + magnesium bromide (aq) (c) calcium hydroxide (aq) + ferric chloride (aq) Write molecular, complete ionic, and net ionic equations. 4. Aqueous nickel ( ...
... 3. Identify the precipitate formed in each of the following reactions: (a) potassium chloride (aq) + lead (II) nitrate (aq) (b) silver nitrate (aq) + magnesium bromide (aq) (c) calcium hydroxide (aq) + ferric chloride (aq) Write molecular, complete ionic, and net ionic equations. 4. Aqueous nickel ( ...
Chapter 6: Chemical Reactions – Study Guide
... 5. For each of the following chemical reactions, write a balanced chemical equation. Be sure to show the state of each reactant and product. Classify each reaction in as many ways as possible. a. Solid lead (II) sulfide reacts with aqueous hydrochloric acid to form solid lead (II) chloride and dihyd ...
... 5. For each of the following chemical reactions, write a balanced chemical equation. Be sure to show the state of each reactant and product. Classify each reaction in as many ways as possible. a. Solid lead (II) sulfide reacts with aqueous hydrochloric acid to form solid lead (II) chloride and dihyd ...
Organic Chemistry
... This enables us to interconvert different R-C(=O)-Z compounds. This is not possible with aldehydes or ketones since the H or R groups attached to C=O are not leaving groups. Acid Chloride Interconversions. Examples of these interconversions are transformations of acid chlorides into carboxylic acids ...
... This enables us to interconvert different R-C(=O)-Z compounds. This is not possible with aldehydes or ketones since the H or R groups attached to C=O are not leaving groups. Acid Chloride Interconversions. Examples of these interconversions are transformations of acid chlorides into carboxylic acids ...
examples of chemical and physical reactions.
... 3. You are given three test tubes containing samples of hydrogen, carbon dioxide and oxygen. What tests do you do to find out which is which? a. Hydrogen – ____________________________________ b. Carbon dioxide - _______________________________ c. Oxygen - _____________________________________ ...
... 3. You are given three test tubes containing samples of hydrogen, carbon dioxide and oxygen. What tests do you do to find out which is which? a. Hydrogen – ____________________________________ b. Carbon dioxide - _______________________________ c. Oxygen - _____________________________________ ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.