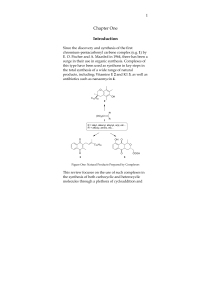
Reductive Deoxygenation of Ketones and Secondary Alcohols by
... ranging between 20% and 90%.4 However, the reduction of ketones to methylene derivatives has been achieved by a number of other reagents: prominent among which are zinc with acid as in the Clemmensen reduction,&sodium borohydride with trifluoroacetic acid, as found by Gribble and co-workers,’jb and ...
... ranging between 20% and 90%.4 However, the reduction of ketones to methylene derivatives has been achieved by a number of other reagents: prominent among which are zinc with acid as in the Clemmensen reduction,&sodium borohydride with trifluoroacetic acid, as found by Gribble and co-workers,’jb and ...
Solvent and Temperature Effects on the Reduction and Amination
... To further examine the product distributions resulting from iPr-LAB, 1 M in THF, prepared from 1 equiv of nBuLi (Scheme 3), a series of reactions of iPr-LAB were performed, varying halides, temperatures, and solvents. The LAB reagent was prepared in THF and varying amounts of dioxane were added to c ...
... To further examine the product distributions resulting from iPr-LAB, 1 M in THF, prepared from 1 equiv of nBuLi (Scheme 3), a series of reactions of iPr-LAB were performed, varying halides, temperatures, and solvents. The LAB reagent was prepared in THF and varying amounts of dioxane were added to c ...
Nucleophilic Additions to Carbonyl Group
... Copyright 1996-2005 by Richard F. Daley & Sally J. Daley ...
... Copyright 1996-2005 by Richard F. Daley & Sally J. Daley ...
RELATIONSHIP BETWEEN Sn1 and E1 REACTIONS
... to form HBr, which is an inorganic product of the reaction. However, we will not focus on inorganic products. As a matter of fact, inorganic products are frequently left out when writing organic reactions and mechanisms to avoid clutter and keep the focus on the organic products. b) Rearrange to a m ...
... to form HBr, which is an inorganic product of the reaction. However, we will not focus on inorganic products. As a matter of fact, inorganic products are frequently left out when writing organic reactions and mechanisms to avoid clutter and keep the focus on the organic products. b) Rearrange to a m ...
Aromatic Compounds
... substitution is more stable than a typical alkyl carbocation because of resonance but much less stable than the starting benzene ring Comparison of alkene addition and aromatic substitution ...
... substitution is more stable than a typical alkyl carbocation because of resonance but much less stable than the starting benzene ring Comparison of alkene addition and aromatic substitution ...
Chapter 1
... • What are the major and minor products when 3methyl-2-butanol is dehydrated? • Zaitsev’s rule states that in an elimination reaction the alkene with the greatest number of alkyl groups on the double bonded carbon is the major product of the reaction ...
... • What are the major and minor products when 3methyl-2-butanol is dehydrated? • Zaitsev’s rule states that in an elimination reaction the alkene with the greatest number of alkyl groups on the double bonded carbon is the major product of the reaction ...
Unit-8-Alcohols-Aldehydes-Ketones
... organic molecules that play important roles in biochemistry; looking both at their physical and chemical properties. The Group VIA elements, oxygen and sulfur, typically form two covalent bonds to attain a filled valence shell. The families that include oxygen and sulfur with two single bonds includ ...
... organic molecules that play important roles in biochemistry; looking both at their physical and chemical properties. The Group VIA elements, oxygen and sulfur, typically form two covalent bonds to attain a filled valence shell. The families that include oxygen and sulfur with two single bonds includ ...
17: Oxidation and Reduction
... We can imagine that chromate ion (CrO4-2) forms from dichromate (Cr2O7-2) by loss of chromium trioxide (CrO3), or that it forms from addition of H2O to CrO3 followed by deprotonation. The three "chromate" species, or the three "dichromate" species, are simply differently protonated froms of CrO4-2 o ...
... We can imagine that chromate ion (CrO4-2) forms from dichromate (Cr2O7-2) by loss of chromium trioxide (CrO3), or that it forms from addition of H2O to CrO3 followed by deprotonation. The three "chromate" species, or the three "dichromate" species, are simply differently protonated froms of CrO4-2 o ...
Latest Publication (still not complete)
... observed reaction pathways and hence is crucial to the total understanding of the chemistry of chromium-pentacarbonyl carbene complexes. Fischer carbene complexes exhibit two characteristic features that are important in understanding their respective chemistry. The first of these features is the fa ...
... observed reaction pathways and hence is crucial to the total understanding of the chemistry of chromium-pentacarbonyl carbene complexes. Fischer carbene complexes exhibit two characteristic features that are important in understanding their respective chemistry. The first of these features is the fa ...
Chapter 11: Reactions of Alcohols
... The halides should be sterically unhindered (primary is best). The more steric factors, the least amount of ether will be obtained. ...
... The halides should be sterically unhindered (primary is best). The more steric factors, the least amount of ether will be obtained. ...
CBSEGuess.com
... Explain why (a) the dipole moment of chlorobenzne is lower than that of cyclohexyl chloride? i. (b) grignard reagent should be prepared under anhydrous condition. ...
... Explain why (a) the dipole moment of chlorobenzne is lower than that of cyclohexyl chloride? i. (b) grignard reagent should be prepared under anhydrous condition. ...
20 More About Oxidation–Reduction Reactions
... The carbonyl group of an amide is reduced to a methylene group (CH 2) by lithium aluminum hydride (Section 18.5). Primary, secondary, and tertiary amines are formed, depending on the number of substituents bonded to the nitrogen of the amide. To obtain the amine in its neutral basic form, acid is no ...
... The carbonyl group of an amide is reduced to a methylene group (CH 2) by lithium aluminum hydride (Section 18.5). Primary, secondary, and tertiary amines are formed, depending on the number of substituents bonded to the nitrogen of the amide. To obtain the amine in its neutral basic form, acid is no ...
Structurally diagnostic ion/molecule reactions
... In 1989, Kenttamaa and Cooks33 reported the first classselective reaction applied with efficiency to considerably complex molecules. They found that protonated ˇhydroxycarbonyl compounds react selectively with ethyl vinyl ether to form characteristic cyclic products (Scheme 8), and demonstrated its ...
... In 1989, Kenttamaa and Cooks33 reported the first classselective reaction applied with efficiency to considerably complex molecules. They found that protonated ˇhydroxycarbonyl compounds react selectively with ethyl vinyl ether to form characteristic cyclic products (Scheme 8), and demonstrated its ...
Chapter 4 Aqueous Reactions and Solution Stoichiometry
... Zn (s) + 2 CuNO3 (aq) 2 Cu (s) + Zn(NO3)2 (aq) Cu (s) + 2 AgNO3 (aq) 2 Ag (s) + Cu(NO3)2 (aq) HCl (aq) + NaOH (aq) NaCl (aq) + H2O (l) ...
... Zn (s) + 2 CuNO3 (aq) 2 Cu (s) + Zn(NO3)2 (aq) Cu (s) + 2 AgNO3 (aq) 2 Ag (s) + Cu(NO3)2 (aq) HCl (aq) + NaOH (aq) NaCl (aq) + H2O (l) ...
The Acid Hydrolysis Mechanism of Acetals Catalyzed
... host molecules have been utilized to affect the hydrolysis of substrates in their interior cavities. The alcoholysis of alkyl halides has been accomplished with size selectivity with assemblies able to concentrate solvent molecules inside of their interior cavities.20 Similarly, β-cyclodextrin has b ...
... host molecules have been utilized to affect the hydrolysis of substrates in their interior cavities. The alcoholysis of alkyl halides has been accomplished with size selectivity with assemblies able to concentrate solvent molecules inside of their interior cavities.20 Similarly, β-cyclodextrin has b ...
applied sciences Chiral β-Amino Alcohols as Ligands for the N
... Since a proportion Ru-dimer:aminoalcohol:base = 1:4:10 is common for the ATH of ketones, we chose that proportion as the starting point of our study, using 5 mol% of the Ru-dimer. Under those conditions, the Ru complex bearing L1 was able to reduce the imine 1a in isopropyl alcohol in the presence o ...
... Since a proportion Ru-dimer:aminoalcohol:base = 1:4:10 is common for the ATH of ketones, we chose that proportion as the starting point of our study, using 5 mol% of the Ru-dimer. Under those conditions, the Ru complex bearing L1 was able to reduce the imine 1a in isopropyl alcohol in the presence o ...
Acidity of Alcohols
... bond, whichever is closer to the end of the chain determines the direction of numbering. • In the case where each would have the same position number, the double bond takes the lower number. • In the name, “ene” comes before “yne” because of alphabetization. ...
... bond, whichever is closer to the end of the chain determines the direction of numbering. • In the case where each would have the same position number, the double bond takes the lower number. • In the name, “ene” comes before “yne” because of alphabetization. ...
Topic 8 notes - A
... A free radical is a species which contains an unpaired electron. Free radicals are very reactive species and will bond with almost anything they come into contact with. In this reaction the free radical which starts the reaction is a chlorine atom, formed when a chlorine molecule breaks up in the pr ...
... A free radical is a species which contains an unpaired electron. Free radicals are very reactive species and will bond with almost anything they come into contact with. In this reaction the free radical which starts the reaction is a chlorine atom, formed when a chlorine molecule breaks up in the pr ...
examination paper - University of Calgary
... Parts 1 - 6 consist of a series of multiple choice questions numbered 1 - 45, which are to be answered on your computer answer sheet. Indicate your answer by blackening out the appropriate space, A, B, C, D or E on the answer sheet. Use a pencil only and not ink. In some cases it is required that yo ...
... Parts 1 - 6 consist of a series of multiple choice questions numbered 1 - 45, which are to be answered on your computer answer sheet. Indicate your answer by blackening out the appropriate space, A, B, C, D or E on the answer sheet. Use a pencil only and not ink. In some cases it is required that yo ...
Learning Guide for Chapter 16
... Synthesis using epoxides What kinds of products can be made using epoxides? compounds with an alcohol next to something that can be attached using a Nu N HO ...
... Synthesis using epoxides What kinds of products can be made using epoxides? compounds with an alcohol next to something that can be attached using a Nu N HO ...
Efficient Homogeneous Catalysis in the Reduction of CO to CO
... CO is produced industrially from fossil fuels.3 Even with strong reducing agents, however, overcoming the OdCO bond enthalpy of 532 kJ/mol4 often presents kinetic difficulties.5,6 Certain metal complexes abstract oxygen readily from CO2,7 but the resulting metal-oxygen bonds are necessarily strong, ...
... CO is produced industrially from fossil fuels.3 Even with strong reducing agents, however, overcoming the OdCO bond enthalpy of 532 kJ/mol4 often presents kinetic difficulties.5,6 Certain metal complexes abstract oxygen readily from CO2,7 but the resulting metal-oxygen bonds are necessarily strong, ...
Document
... General procedure for 2a-2j. To a magnetically stirred solution of aldehyde (1 mmol), and potassium bromide (0.5 mmol) in MeOH (10 mL) was added dropwise 0.5 N HCl (2 mL) followed by iodosobenzene (1.5 mmol; 330 mg) and the solution stirred. Progress of the reaction was monitored by TLC. After compl ...
... General procedure for 2a-2j. To a magnetically stirred solution of aldehyde (1 mmol), and potassium bromide (0.5 mmol) in MeOH (10 mL) was added dropwise 0.5 N HCl (2 mL) followed by iodosobenzene (1.5 mmol; 330 mg) and the solution stirred. Progress of the reaction was monitored by TLC. After compl ...
Acid derivatives
... individual steps in these mechanisms vary, but the essential characteristic of the overall transformation is that of addition followed by elimination. Acid catalysts act to increase the electrophilicity of the acyl reactant; whereas, base catalysts act on the nucleophilic reactant to increase its re ...
... individual steps in these mechanisms vary, but the essential characteristic of the overall transformation is that of addition followed by elimination. Acid catalysts act to increase the electrophilicity of the acyl reactant; whereas, base catalysts act on the nucleophilic reactant to increase its re ...
Ene reaction

The ene reaction (also known as the Alder-ene reaction) is a chemical reaction between an alkene with an allylic hydrogen (the ene) and a compound containing a multiple bond (the enophile), in order to form a new σ-bond with migration of the ene double bond and 1,5 hydrogen shift. The product is a substituted alkene with the double bond shifted to the allylic position.This transformation is a group transfer pericyclic reaction, and therefore, usually requires highly activated substrates and/or high temperatures. Nonetheless, the reaction is compatible with a wide variety of functional groups that can be appended to the ene and enophile moieties. Also,many useful Lewis acid-catalyzed ene reactions have been developed which can afford high yields and selectivities at significantly lower temperatures, making the ene reaction a useful C–C forming tool for the synthesis of complex molecules and natural products.