Reductions of Carboxylic Acid Derivatives - IDC
... increased temperature and pressure, it is possible to reduce aldehydes and ketones to alcohols, but carboxylic acids, esters and amides are comparatively unreactive. The exceptional reactivity of acyl halides, on the other hand, facilitates their reduction under mild conditions, by using a poisoned ...
... increased temperature and pressure, it is possible to reduce aldehydes and ketones to alcohols, but carboxylic acids, esters and amides are comparatively unreactive. The exceptional reactivity of acyl halides, on the other hand, facilitates their reduction under mild conditions, by using a poisoned ...
Wed March 3 lecture
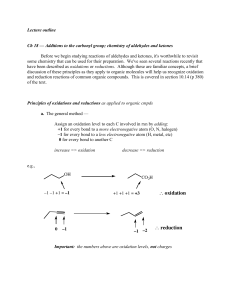
... Before we begin studying reactions of aldehydes and ketones, it's worthwhile to revisit some chemistry that can be used for their preparation. We've seen several reactions recently that have been described as oxidations or reductions. Although these are familiar concepts, a brief discussion of these ...
... Before we begin studying reactions of aldehydes and ketones, it's worthwhile to revisit some chemistry that can be used for their preparation. We've seen several reactions recently that have been described as oxidations or reductions. Although these are familiar concepts, a brief discussion of these ...
Chapter 14 Selenium reagents
... • Aryl alkyl selenides are preparable either (as above) from electrophilic selenium reagents and carbon ncleophiles or from nucleophilic selenium reagents, e.g. ArSe-Na+, and carbon electrophiles. On oxidation they give selenoxides; if these contain a -hydrogen, they may undergo spontaneous syn-add ...
... • Aryl alkyl selenides are preparable either (as above) from electrophilic selenium reagents and carbon ncleophiles or from nucleophilic selenium reagents, e.g. ArSe-Na+, and carbon electrophiles. On oxidation they give selenoxides; if these contain a -hydrogen, they may undergo spontaneous syn-add ...
+ Y
... has an electron-poor atom (e.g H+, CH3+ ) and can form a bond by accepting a pair of electrons from a nucleophile ...
... has an electron-poor atom (e.g H+, CH3+ ) and can form a bond by accepting a pair of electrons from a nucleophile ...
Lesson 4 halogenoalkanes
... CH3CH2CH2I + K+CN- CH3CH2CH2CN + I CN- is the cyanide ion. Reaction is carried out under reflux. Reaction is useful as it extends the carbon chain. ...
... CH3CH2CH2I + K+CN- CH3CH2CH2CN + I CN- is the cyanide ion. Reaction is carried out under reflux. Reaction is useful as it extends the carbon chain. ...
Chapter 21: Carboxylic Acid Derivatives
... this reaction will not proceed since Clis a weaker base than NH2O ...
... this reaction will not proceed since Clis a weaker base than NH2O ...
Ch 16 Aldehydes and Ketones I
... • Addition of primary and secondary amines – Primary amines react with ald/ket to form imines – An imine has a C-N double bond – Secondary amines react with ald/ket to form enamines – An enamine has an amino group bonded to carboncarbon double bond. ...
... • Addition of primary and secondary amines – Primary amines react with ald/ket to form imines – An imine has a C-N double bond – Secondary amines react with ald/ket to form enamines – An enamine has an amino group bonded to carboncarbon double bond. ...
Here is the Original File - University of New Hampshire
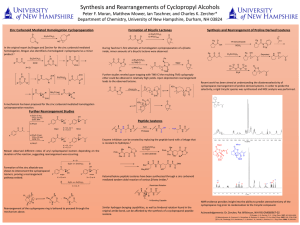
... Enzyme inhibitors can be created by replacing the peptide bond with a linkage that is resistant to hydrolysis.2 Mower observed different ratios of aryl cyclopropanol isomers depending on the duration of the reaction, suggesting rearrangement was occurring. ...
... Enzyme inhibitors can be created by replacing the peptide bond with a linkage that is resistant to hydrolysis.2 Mower observed different ratios of aryl cyclopropanol isomers depending on the duration of the reaction, suggesting rearrangement was occurring. ...
Exam 2 Review Sheet for Friday, March 2 Exam Chem 1120, Spring
... • Define and use the following terms: catenation, hybridization, homologous, saturated, unsaturated, condensed structural formula, general structural formula, radicals, isomers. • Explain why there are so many carbon compounds. • List and explain the different types of hybridization that carbon unde ...
... • Define and use the following terms: catenation, hybridization, homologous, saturated, unsaturated, condensed structural formula, general structural formula, radicals, isomers. • Explain why there are so many carbon compounds. • List and explain the different types of hybridization that carbon unde ...
Chapter 7 - Alkenes and Alkynes I less substituted alkene due to
... - Carrying out dehydrohalogenations with a bulky base, such as tert-butxide, favors the formation of the less substituted alkene due to sterics - When an elimination yields the less substituted alkene, it follows the Hofmann Rule - The anticoplanar transition state is preferred, but the syn coplanar ...
... - Carrying out dehydrohalogenations with a bulky base, such as tert-butxide, favors the formation of the less substituted alkene due to sterics - When an elimination yields the less substituted alkene, it follows the Hofmann Rule - The anticoplanar transition state is preferred, but the syn coplanar ...
EXP-7
... The Cannizzaro reaction is that of aldehydes that do not contain alpha hydrogens to give carboxylic acids and alcohols (alpha hydrogens cause an Aldol reaction to take place). This occurs in the presence of a strong base. Benzaldehyde, which does not contain alpha hydrogens, was used for this reacti ...
... The Cannizzaro reaction is that of aldehydes that do not contain alpha hydrogens to give carboxylic acids and alcohols (alpha hydrogens cause an Aldol reaction to take place). This occurs in the presence of a strong base. Benzaldehyde, which does not contain alpha hydrogens, was used for this reacti ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034
... 5. Explain the change in the geometry of excited state molecule in a photochemical process and the variation in its physical property. 6. What is Norrish type I and II cleavage reactions? Give suitable examples. 7. What are the important guidelines to be followed while choosing alternate synthetic r ...
... 5. Explain the change in the geometry of excited state molecule in a photochemical process and the variation in its physical property. 6. What is Norrish type I and II cleavage reactions? Give suitable examples. 7. What are the important guidelines to be followed while choosing alternate synthetic r ...
Tests for functional groups
... Carboxylic acids contain a hydroxyl (-OH) group. They therefore react with PCl5 in the same manner as described for alcohols above ii) Reaction with water Carboxylic acids are weak acids, i.e. they are only partially ionised in aqueous solution. Most are reasonably soluble, giving a solution with a ...
... Carboxylic acids contain a hydroxyl (-OH) group. They therefore react with PCl5 in the same manner as described for alcohols above ii) Reaction with water Carboxylic acids are weak acids, i.e. they are only partially ionised in aqueous solution. Most are reasonably soluble, giving a solution with a ...
Ch 19 Aldehydes and Ketones
... - Aldehydes are more reactive than ketones for both steric and electronic reasons. - First, the H creates less steric hindrance so that the carbonyl C is more accessible. - Second, an organic group provides e- donating induction which stabilizes the + carbonyl C and makes it less reactive. - Formal ...
... - Aldehydes are more reactive than ketones for both steric and electronic reasons. - First, the H creates less steric hindrance so that the carbonyl C is more accessible. - Second, an organic group provides e- donating induction which stabilizes the + carbonyl C and makes it less reactive. - Formal ...
Ethers, Sulfides, Epoxides
... Most of the carbocation reacts to give the 1,2 product because of the smaller Ea leading to the 1,2 product. This is true at all temperatures. At higher temperatures the reverse reactions occur leading from the 1,2 or 1,4 product to the carbocation. Note that the 1,2 product is more easily converte ...
... Most of the carbocation reacts to give the 1,2 product because of the smaller Ea leading to the 1,2 product. This is true at all temperatures. At higher temperatures the reverse reactions occur leading from the 1,2 or 1,4 product to the carbocation. Note that the 1,2 product is more easily converte ...
Samantha Landolfa Amy Ryan Section 10 Experiment 9 – Alkenes
... products follow Zaitsev’s Rule, which states that the major product will be the most substituted alkene because it is the most stable. In this experiment, the most stable alkene is 2-methly-2-butene, which is tri-substituted about the double bond. The minor product is 2-methyl-1-butene, a di-substit ...
... products follow Zaitsev’s Rule, which states that the major product will be the most substituted alkene because it is the most stable. In this experiment, the most stable alkene is 2-methly-2-butene, which is tri-substituted about the double bond. The minor product is 2-methyl-1-butene, a di-substit ...
Chapter 7: Alkenes and Alkynes – Properties and Synthesis
... Allylic substitution, Allyl radical, Allylic chlorination Allylic bromination, N-Bromosuccinimide MO of allyl radical and allyl cation Rules for writing resonance structures ...
... Allylic substitution, Allyl radical, Allylic chlorination Allylic bromination, N-Bromosuccinimide MO of allyl radical and allyl cation Rules for writing resonance structures ...
C h e m g u i d e ... CARBOXYLIC ACIDS: PREPARATION
... Cr2O72- + 14H+ + 6e(i) Work out the electron-half-equation for the conversion of propan-1-ol to propanoic acid. (ii) Combine that with the electron-half-equation given above to work out the ionic equation for the overall reaction. e) Although it would be less commonly done, you could also use the sa ...
... Cr2O72- + 14H+ + 6e(i) Work out the electron-half-equation for the conversion of propan-1-ol to propanoic acid. (ii) Combine that with the electron-half-equation given above to work out the ionic equation for the overall reaction. e) Although it would be less commonly done, you could also use the sa ...
Isoborneol from Camphor: A Reduction
... while they are an unresolved singlet in borneol. 2. The reduction mechanism is often shown with a hydride ion (H:-) attacking the carbonyl carbon. Why might one assign a partial negative charge to the hydrogens in sodium borohydride? Cautions: This reaction involves diethyl ether which is extremely ...
... while they are an unresolved singlet in borneol. 2. The reduction mechanism is often shown with a hydride ion (H:-) attacking the carbonyl carbon. Why might one assign a partial negative charge to the hydrogens in sodium borohydride? Cautions: This reaction involves diethyl ether which is extremely ...
Fragrant Esters Esters are prepared in a reaction between a
... can prepare esters that produce different scents. Sometimes only small changes are needed to get a completely different fragrance. For example experiment 1 (see below) yields an ester that smells like apple. A slightly longer chain produces an orange scent. Having a branched chain leads to banana oi ...
... can prepare esters that produce different scents. Sometimes only small changes are needed to get a completely different fragrance. For example experiment 1 (see below) yields an ester that smells like apple. A slightly longer chain produces an orange scent. Having a branched chain leads to banana oi ...
Mild Reduction of Carboxylic Acids to Alcohols
... © 1999ElsevierScienceLtd. All rightsreserved. Keywords: Reduction, Carboxylic acids, Alcohols, Amino acids. ...
... © 1999ElsevierScienceLtd. All rightsreserved. Keywords: Reduction, Carboxylic acids, Alcohols, Amino acids. ...
Dehydration notes
... Dehydration of alcohols is … Acid catalyzed – creates a good leaving group (i.e. water) Carbocation intermediate formation. First two steps of the mechanism at the same as for SN1. Carbocation will rearrange for increased stability, if possible. 5. Protons can be removed from any adjacent position ...
... Dehydration of alcohols is … Acid catalyzed – creates a good leaving group (i.e. water) Carbocation intermediate formation. First two steps of the mechanism at the same as for SN1. Carbocation will rearrange for increased stability, if possible. 5. Protons can be removed from any adjacent position ...
National 5 Whole Course Revision Questions Unit 1 Chemical
... 1. Describe how temperature, concentration and particle size affect the rate of a chemical reaction- mention collision theory in your answer. 2. a) How do catalysts affect the rate of a chemical reaction? b) Name the types of catalysts and describe how they differ from each other? 3. What is an enzy ...
... 1. Describe how temperature, concentration and particle size affect the rate of a chemical reaction- mention collision theory in your answer. 2. a) How do catalysts affect the rate of a chemical reaction? b) Name the types of catalysts and describe how they differ from each other? 3. What is an enzy ...
Unit 3: Reactions of Alkenes. Thermodynamics and Kinetics
... This symbol indicates that the reaction takes place under standard conditions --all species at 1 M, 25 OC, and 1 atm. ...
... This symbol indicates that the reaction takes place under standard conditions --all species at 1 M, 25 OC, and 1 atm. ...
Wolff–Kishner reduction

The Wolff–Kishner reduction is a reaction used in organic chemistry to convert carbonyl functionalities into methylene groups. In the context of complex molecule synthesis, it is most frequently employed to remove a carbonyl group after it has served its synthetic purpose of activating an intermediate in a preceding step. As such, there is no obvious retron for this reaction. Originally reported by Nikolai Kischner in 1911 and Ludwig Wolff in 1912, it has been applied to the total synthesis of scopadulcic acid B, aspidospermidine and dysidiolide.In general, the reaction mechanism first involves the in situ generation of a hydrazone by condensation of hydrazine with the ketone or aldehyde substrate. Sometimes it is however advantageous to use a pre-formed hydrazone as substrate (see modifications). The hydrazone is deprotonated by alkoxide base followed by a concerted, rate-determining step in which a diimide anion is formed. Collapse of this alkyldiimde with loss of N2 leads to formation of an alkylanion which can be protonated by solvent to give the desired product.Because the Wolff–Kishner reduction requires highly basic conditions, it is unsuitable for base-sensitive substrates. However, this method can be superior over the related Clemmensen reduction for acid-sensitive compounds such as pyrroles and for high-molecular weight compounds.