PowerPoint
... 8. Dicarboxylic acids – containing two carboxyl groups Aldaric acid – a family of sugar acids Oxalic acid – found in many foods Malonic acid Malic acid – found in apples Succinic acid – a component of the citric cycle Glutaric acid 9. Tricarboxylic acid – containing a hydroxy groups Ci ...
... 8. Dicarboxylic acids – containing two carboxyl groups Aldaric acid – a family of sugar acids Oxalic acid – found in many foods Malonic acid Malic acid – found in apples Succinic acid – a component of the citric cycle Glutaric acid 9. Tricarboxylic acid – containing a hydroxy groups Ci ...
A manganese catalyst for highly reactive yet chemoselective
... significantly lower reactivity with this challenging substrate (entries 3 and 4). The enhanced reactivity for an electrophilic C–H amination reaction may be attributed to an electronic difference between these ligands, as evidence suggests that phthalocyanines are significantly better π-acceptor ligan ...
... significantly lower reactivity with this challenging substrate (entries 3 and 4). The enhanced reactivity for an electrophilic C–H amination reaction may be attributed to an electronic difference between these ligands, as evidence suggests that phthalocyanines are significantly better π-acceptor ligan ...
CH - UTH e
... • Carbanions (nucleophiles) can react with electrophilic carbon centers in favorable cases. The net result is a carbon‐‐carbon bond carbon carbon bond‐‐ ‐‐a big deal! a big deal! • Grignards and organolithium reagents react with many ...
... • Carbanions (nucleophiles) can react with electrophilic carbon centers in favorable cases. The net result is a carbon‐‐carbon bond carbon carbon bond‐‐ ‐‐a big deal! a big deal! • Grignards and organolithium reagents react with many ...
Study Guide Chapter 4 Alcohols and Alkyl Halides
... The hydroxyl group is trans to the isopropyl group and cis to the methyl group. All three substituents need not always be equatorial; instead, one or two of them may be axial. Since neomenthol is the second most stable stereoisomer, we choose the structure with one axial substituent. Furthermore, we ...
... The hydroxyl group is trans to the isopropyl group and cis to the methyl group. All three substituents need not always be equatorial; instead, one or two of them may be axial. Since neomenthol is the second most stable stereoisomer, we choose the structure with one axial substituent. Furthermore, we ...
Chapter 16 - Chemistry of Benzene
... The product is formed by loss of a proton, which is replaced by bromine FeBr3 is added as a catalyst to polarize the bromine reagent ...
... The product is formed by loss of a proton, which is replaced by bromine FeBr3 is added as a catalyst to polarize the bromine reagent ...
Alcohols, Phenols, and Thiols
... classified as primary, secondary, or tertiary depending on whether one, two, or three organic groups are attached to the hydroxyl-bearing carbon. The nomenclature of alcohols and phenols is summarized in Secs. 7.1–7.3. Alcohols and phenols form hydrogen bonds. These bonds account for the relatively ...
... classified as primary, secondary, or tertiary depending on whether one, two, or three organic groups are attached to the hydroxyl-bearing carbon. The nomenclature of alcohols and phenols is summarized in Secs. 7.1–7.3. Alcohols and phenols form hydrogen bonds. These bonds account for the relatively ...
Chapter 13, sections 13.5 - Properties of Aldehydes and Ketones
... which contains Cu2+, reacts with aldehydes that have an adjacent OH group. • An aldehyde is oxidized to a carboxylic acid, while Cu2+ is reduced to give red Cu2O(s). ...
... which contains Cu2+, reacts with aldehydes that have an adjacent OH group. • An aldehyde is oxidized to a carboxylic acid, while Cu2+ is reduced to give red Cu2O(s). ...
chapter 4 -aromatic compounds
... • A monosubstituted benzene has the formula C6H5G where G is the group that replaces a hydrogen atom. • All hydrogens in benzene are equivalent. ...
... • A monosubstituted benzene has the formula C6H5G where G is the group that replaces a hydrogen atom. • All hydrogens in benzene are equivalent. ...
Dehydration of n-propanol and methanol to produce
... Abstract: An ether is an organic compound that consists of an oxygen atom bonded to two alkyl or aryl groups. In this work, we investigate the bimolecular dehydration of two alcohols, n-propanol and methanol with catalysts that are used in transesterification. Experiments were carried out to evaluat ...
... Abstract: An ether is an organic compound that consists of an oxygen atom bonded to two alkyl or aryl groups. In this work, we investigate the bimolecular dehydration of two alcohols, n-propanol and methanol with catalysts that are used in transesterification. Experiments were carried out to evaluat ...
Slide 1
... • A monosubstituted benzene has the formula C6H5G where G is the group that replaces a hydrogen atom. • All hydrogens in benzene are equivalent. ...
... • A monosubstituted benzene has the formula C6H5G where G is the group that replaces a hydrogen atom. • All hydrogens in benzene are equivalent. ...
chapter27
... – Alkynes with only one C C bond have the formula CnH2n-2. • Each carbon atom in a C C bond is sp hybridized. – Each sp hybrid contains two bonds and two p bonds. – The carbon atom will have one single bond and one triple bond. ...
... – Alkynes with only one C C bond have the formula CnH2n-2. • Each carbon atom in a C C bond is sp hybridized. – Each sp hybrid contains two bonds and two p bonds. – The carbon atom will have one single bond and one triple bond. ...
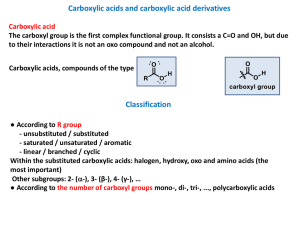
carboxylic acid
... connected to main hydrocarbon chain - In this case the group COOH is not numbered! When a carboxyl group is attached to a ring, the parent ring is named (retaining the final -e) and the suffix -carboxylic acid is added. Compounds with two carboxyl groups are distinguished by the suffix -dioic acid o ...
... connected to main hydrocarbon chain - In this case the group COOH is not numbered! When a carboxyl group is attached to a ring, the parent ring is named (retaining the final -e) and the suffix -carboxylic acid is added. Compounds with two carboxyl groups are distinguished by the suffix -dioic acid o ...
Tro Chemistry a Molecular Approach, 3E
... CHECK The units of the answer are correct. The magnitude of the answer (25.8 g) is less than the initial mass of CO2 (37.8 g). This is reasonable because each carbon in CO2 has two oxygen atoms associated with it, while in C6 H12 O6 each carbon has only one oxygen atom associated with it and two hyd ...
... CHECK The units of the answer are correct. The magnitude of the answer (25.8 g) is less than the initial mass of CO2 (37.8 g). This is reasonable because each carbon in CO2 has two oxygen atoms associated with it, while in C6 H12 O6 each carbon has only one oxygen atom associated with it and two hyd ...
Chm 2
... d. 11.0 mol For the reaction represented by the equation Cl2 + 2KBr 2KCl + Br2, how many moles of potassium chloride are produced from 119 g of potassium bromide? a. 0.119 mol c. 0.581 mol b. 0.236 mol d. 1.00 mol For the reaction represented by the equation 3Fe + 4H2O Fe3O4 + 4H2, how many mole ...
... d. 11.0 mol For the reaction represented by the equation Cl2 + 2KBr 2KCl + Br2, how many moles of potassium chloride are produced from 119 g of potassium bromide? a. 0.119 mol c. 0.581 mol b. 0.236 mol d. 1.00 mol For the reaction represented by the equation 3Fe + 4H2O Fe3O4 + 4H2, how many mole ...
2 - C7Chemistry
... 2 moles of ammonia are produced, along with 1 mole of carbon dioxide and 1 mole of water vapor. ...
... 2 moles of ammonia are produced, along with 1 mole of carbon dioxide and 1 mole of water vapor. ...
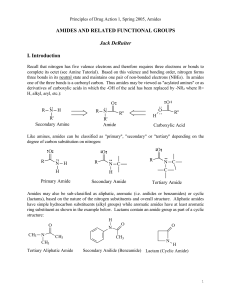
AMIDES AND RELATED FUNCTIONAL GROUPS
... secondary amides also contain two- and one N-H dipoles, respectively. Because of the π-bonding arrangement of the carbonyl and the greater electronegativity of oxygen, the carbonyl (C=O) is a stronger dipole than the N-C dipole. The presence of a C=O dipole and, to a lesser extent a N-C dipole, allo ...
... secondary amides also contain two- and one N-H dipoles, respectively. Because of the π-bonding arrangement of the carbonyl and the greater electronegativity of oxygen, the carbonyl (C=O) is a stronger dipole than the N-C dipole. The presence of a C=O dipole and, to a lesser extent a N-C dipole, allo ...
Unfamiliar Oxidation States and Tkeir Stabilization
... magnetic susceptibility measurements; and (4) X-ray studies. In some instances a combination of two or more of these has been necessary for the complete characterization of the oxidation state in question. Analytical data, in conjunction with a study of the chemical properties of the substance, freq ...
... magnetic susceptibility measurements; and (4) X-ray studies. In some instances a combination of two or more of these has been necessary for the complete characterization of the oxidation state in question. Analytical data, in conjunction with a study of the chemical properties of the substance, freq ...
- Kendriya Vidyalaya Jamuna Colliery
... 3. Niobium crystallizes in bcc structure. If its density is 8.55 cm-3, calculate atomic radius of [At. Mass of Niobium = 92.9u, NA = 6.022 x 1023 atoms mol-1 ]. 4. If radius of octahedral void is r and radius of atom in close packing is R, derive the relationship between r and R. 5. Non stoichiomet ...
... 3. Niobium crystallizes in bcc structure. If its density is 8.55 cm-3, calculate atomic radius of [At. Mass of Niobium = 92.9u, NA = 6.022 x 1023 atoms mol-1 ]. 4. If radius of octahedral void is r and radius of atom in close packing is R, derive the relationship between r and R. 5. Non stoichiomet ...
Recent advances in homogeneous nickel catalysis
... field have been reported. For example, Kocienski and Dixon developed the first effective Ni(0)catalysed cross-coupling of vinyl carbamates with organomagnesium reagents in 198921, and several years later Snieckus and co-workers described the Ni(0)catalysed cross-coupling of aryl carbamates and organ ...
... field have been reported. For example, Kocienski and Dixon developed the first effective Ni(0)catalysed cross-coupling of vinyl carbamates with organomagnesium reagents in 198921, and several years later Snieckus and co-workers described the Ni(0)catalysed cross-coupling of aryl carbamates and organ ...
Recognize the functional group and give a characteristic of this
... A group of covalently bonded elements that when bonded to a carbon chain give it a unique quality. Compounds with similar functional groups will have similar qualities and properties. ...
... A group of covalently bonded elements that when bonded to a carbon chain give it a unique quality. Compounds with similar functional groups will have similar qualities and properties. ...
chapter 5 soaps and detergents
... The materials are sold as a-olefinsulfonates (AOS) because of the use of olefinic precursors. AOS has not yet made great strides in the heavy-duty laundry field but is being used successfully for light duty detergents, hand dishwashing shampoos, bubble baths and synthetic soap bars. ...
... The materials are sold as a-olefinsulfonates (AOS) because of the use of olefinic precursors. AOS has not yet made great strides in the heavy-duty laundry field but is being used successfully for light duty detergents, hand dishwashing shampoos, bubble baths and synthetic soap bars. ...
431 KB / 47 pages
... (a) We have seen (Investigate This 10.2) that electrolysis of a dilute aqueous solution of an ionic compound (magnesium sulfate) produces a gas at both electrodes and a basic solution at the cathode and acidic solution at the anode, just as the problem statement says is observed here for a dilute aq ...
... (a) We have seen (Investigate This 10.2) that electrolysis of a dilute aqueous solution of an ionic compound (magnesium sulfate) produces a gas at both electrodes and a basic solution at the cathode and acidic solution at the anode, just as the problem statement says is observed here for a dilute aq ...
ap 2005 chemistry_b scoring guidelines - AP Central
... Write the formulas to show the reactants and the products for any FIVE of the laboratory situations described below. Answers to more than five choices will not be graded. In all cases, a reaction occurs. Assume that solutions are aqueous unless otherwise indicated. Represent substances in solution a ...
... Write the formulas to show the reactants and the products for any FIVE of the laboratory situations described below. Answers to more than five choices will not be graded. In all cases, a reaction occurs. Assume that solutions are aqueous unless otherwise indicated. Represent substances in solution a ...
Lecture - Ch 16
... carbonyl group toward reduction – An aryl alkyl ketone can be converted into an alkylbenzene by catalytic hydrogenation over a palladium catalyst ...
... carbonyl group toward reduction – An aryl alkyl ketone can be converted into an alkylbenzene by catalytic hydrogenation over a palladium catalyst ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.