Ch 06
... electric field between them). If the molecules have enough kinetic energy, they can approach closely enough for their bond structures to rearrange to form an activated complex. The activated complex is an unstable molecule with a particular geometry. It is unstable because it possesses the maximum p ...
... electric field between them). If the molecules have enough kinetic energy, they can approach closely enough for their bond structures to rearrange to form an activated complex. The activated complex is an unstable molecule with a particular geometry. It is unstable because it possesses the maximum p ...
Homogeneously catalysed hydrogenation of unsaturated fatty acids
... Fatty alcohols can be prepared b y heating the corresponding metal soaps under hydrogen. Pb and Cd soaps of unsaturated f a t t y acids give as reaction product unsaturated f a t t y alcohols, the Cu- and Sn-soaps yield saturated alcohols and mixtures of Cu- and Cdsoaps again give unsaturated f a t ...
... Fatty alcohols can be prepared b y heating the corresponding metal soaps under hydrogen. Pb and Cd soaps of unsaturated f a t t y acids give as reaction product unsaturated f a t t y alcohols, the Cu- and Sn-soaps yield saturated alcohols and mixtures of Cu- and Cdsoaps again give unsaturated f a t ...
Organic Chemistry
... Organic chemistry is the branch of chemistry that deals with the study of carbon based compounds. Bonds between carbon atoms are covalent; each carbon is capable of forming four bonds: – Four single bonds = tertrahedral geometry – Two single, one double = trigonal planar geometry – Two double = line ...
... Organic chemistry is the branch of chemistry that deals with the study of carbon based compounds. Bonds between carbon atoms are covalent; each carbon is capable of forming four bonds: – Four single bonds = tertrahedral geometry – Two single, one double = trigonal planar geometry – Two double = line ...
Organic Chemistry
... • Resulted in 10,000’s of birth defects • (R) form is effective, but the (S) form can bind to DNA to produce mutagenetic effects. • Many organic reactions produce a racemic mixture of equal parts. ...
... • Resulted in 10,000’s of birth defects • (R) form is effective, but the (S) form can bind to DNA to produce mutagenetic effects. • Many organic reactions produce a racemic mixture of equal parts. ...
32. The reaction described by this equilibrium is
... hydrogen gas is less expensive and is readily available for use the molar heat of combustion for hydrogen gas is greater than those of the hydrocarbon fuels ...
... hydrogen gas is less expensive and is readily available for use the molar heat of combustion for hydrogen gas is greater than those of the hydrocarbon fuels ...
Subject Materials for Chemistry
... 27. How is steel manufactured by Bessemer process? Ans: For fig ref.page number 22 fig.number2.6. Steel is manufactured from pig iron in a Bessemer converter, which is a pear shaped furnace lined inside with silicon. Molten pig iron is taken in Bessemer converter is heated with a hot blast of air. O ...
... 27. How is steel manufactured by Bessemer process? Ans: For fig ref.page number 22 fig.number2.6. Steel is manufactured from pig iron in a Bessemer converter, which is a pear shaped furnace lined inside with silicon. Molten pig iron is taken in Bessemer converter is heated with a hot blast of air. O ...
CHEM 263 Oct 14 revised
... Reactions of Substituted Benzene – Continued How can Basic Brown be synthesized from toluene? (The process of figuring out what starting materials could be used to synthesize a target molecule is known as ...
... Reactions of Substituted Benzene – Continued How can Basic Brown be synthesized from toluene? (The process of figuring out what starting materials could be used to synthesize a target molecule is known as ...
alcohol
... Phenyl cations are energetically unstable and are difficult to form. Phenols can also never undergo displacement by the SN2 mechanism ...
... Phenyl cations are energetically unstable and are difficult to form. Phenols can also never undergo displacement by the SN2 mechanism ...
AP Chemistry - West Bloomfield School District
... In a certain experiment, 6.00 g of aluminum is burned in 24.0 g of bromine. What is the maximum amount of aluminum bromide that can be produced? 67. Acid-base neutralization reactions are very common in industrial processes. This is the reaction of sulfuric acid with sodium hydroxide: H2SO4 (aq) + 2 ...
... In a certain experiment, 6.00 g of aluminum is burned in 24.0 g of bromine. What is the maximum amount of aluminum bromide that can be produced? 67. Acid-base neutralization reactions are very common in industrial processes. This is the reaction of sulfuric acid with sodium hydroxide: H2SO4 (aq) + 2 ...
Alkenes
... system. The trans double bond is in a ten membered ring. Compound (b) is a Bredt’s rule violation and is not stable. The largest ring contains six carbon atoms, and the trans double bond cannot be stable in this bridgehead position. Compound (c) (norbornene) is stable. The (cis) double bond is not a ...
... system. The trans double bond is in a ten membered ring. Compound (b) is a Bredt’s rule violation and is not stable. The largest ring contains six carbon atoms, and the trans double bond cannot be stable in this bridgehead position. Compound (c) (norbornene) is stable. The (cis) double bond is not a ...
Atomic Structure
... Bond order is a concept in the molecular orbital theory. It depends on the number of electrons in the bonding and antibonding orbitals. Which of the following statements is true about it? The bond order (a) Cannot be a negative quantity (b) Always has an integral value (c) Can assume any value, posi ...
... Bond order is a concept in the molecular orbital theory. It depends on the number of electrons in the bonding and antibonding orbitals. Which of the following statements is true about it? The bond order (a) Cannot be a negative quantity (b) Always has an integral value (c) Can assume any value, posi ...
Organic Models
... bond with each other as well as with atoms of other elements. Each carbon atom, however, may make only four covalent bonds. When carbon atoms bond with each other, they form a carbon chain. Compounds containing carbon chains may become a homologous series. A homologous series is a set of compounds t ...
... bond with each other as well as with atoms of other elements. Each carbon atom, however, may make only four covalent bonds. When carbon atoms bond with each other, they form a carbon chain. Compounds containing carbon chains may become a homologous series. A homologous series is a set of compounds t ...
AP Chemistry Chapter 16
... - Go is a state function since can be determined using the same method as H -remember the relationship G = H - T S - Gf is the symbol used for standard free energy of formation -for any element in its standard state under normal conditions (1 atm, 25.0 C) the free state value is zero -reme ...
... - Go is a state function since can be determined using the same method as H -remember the relationship G = H - T S - Gf is the symbol used for standard free energy of formation -for any element in its standard state under normal conditions (1 atm, 25.0 C) the free state value is zero -reme ...
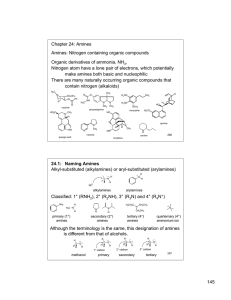
145 Chapter 24: Amines Amines: Nitrogen containing organic
... Alkyl ammonium ions, R3NH+ X-, have pKa values in the range of 10-11 (ammonium ion, H4N+ X-, has a pKa ~ 9.25) The ammonium ions of aryl amines and heterocyclic aromatic amines are considerably less basic than alkyl amines (pKa ~ 5 or less). The nitrogen lone pair is less basic if it is in an sp2 hy ...
... Alkyl ammonium ions, R3NH+ X-, have pKa values in the range of 10-11 (ammonium ion, H4N+ X-, has a pKa ~ 9.25) The ammonium ions of aryl amines and heterocyclic aromatic amines are considerably less basic than alkyl amines (pKa ~ 5 or less). The nitrogen lone pair is less basic if it is in an sp2 hy ...
Organic Chemical Reactions
... thermodynamics and kinetics must be favorable, that is the reaction must have a negative ΔG and should occur in a relatively fast manner. As far as kinetics is concerned the reactants must go through an energy barrier, called the free energy of activation ΔG‡. Once this energy is gained, partial bon ...
... thermodynamics and kinetics must be favorable, that is the reaction must have a negative ΔG and should occur in a relatively fast manner. As far as kinetics is concerned the reactants must go through an energy barrier, called the free energy of activation ΔG‡. Once this energy is gained, partial bon ...
Redox Flash Cards - No Brain Too Small
... the process by which ionic compounds are split into their atoms using electric currents electrolysis ...
... the process by which ionic compounds are split into their atoms using electric currents electrolysis ...
File
... the hydroxide it attached to. If the hydroxide is on the end of a chain and only connected to a carbon atom that is attached to only one other carbon atom it is known as a primary alcohol. If the hydroxide group is attached to a carbon atom which is also attached to two other carbons it is known as ...
... the hydroxide it attached to. If the hydroxide is on the end of a chain and only connected to a carbon atom that is attached to only one other carbon atom it is known as a primary alcohol. If the hydroxide group is attached to a carbon atom which is also attached to two other carbons it is known as ...
A2 LEVEL CHEMISTRY 4.1.1 ARENES TEST Answer all questions
... In this question, one mark is available for the quality of use and organisation of scientific terms. Describe how benzene could be converted into nitrobenzene. State the reagents and conditions, give a balanced equation for each stage and show the structure of the product. ...
... In this question, one mark is available for the quality of use and organisation of scientific terms. Describe how benzene could be converted into nitrobenzene. State the reagents and conditions, give a balanced equation for each stage and show the structure of the product. ...
Practice Problem - HCC Southeast Commons
... resonance effects that reinforce each other – The ortho and para intermediates are destabilized – The positive charge of the carbocation intermediate in ortho and para attack is directly on the carbon that bears the deactivating group and resonance ...
... resonance effects that reinforce each other – The ortho and para intermediates are destabilized – The positive charge of the carbocation intermediate in ortho and para attack is directly on the carbon that bears the deactivating group and resonance ...
Mechanistic Studies of the Reactions of Silicon
... formation of a silene-alcohol complex, followed by intracomplex proton transfer. The latter is rate-determining in all cases but acetic acid, for which it is proposed that complexation is the rate-determining step for reaction. Proton transfer from the complex to a second molecule of alcohol compete ...
... formation of a silene-alcohol complex, followed by intracomplex proton transfer. The latter is rate-determining in all cases but acetic acid, for which it is proposed that complexation is the rate-determining step for reaction. Proton transfer from the complex to a second molecule of alcohol compete ...
AP BIOLOGY Chapter 4 - Bremen High School District 228
... the ß (beta) form so every other glucose is upside down the respect to its neighbors; joined with 1,4 linkages; straight-never branched; hydrogen bonds join parallel molecules into microfibrils; ...
... the ß (beta) form so every other glucose is upside down the respect to its neighbors; joined with 1,4 linkages; straight-never branched; hydrogen bonds join parallel molecules into microfibrils; ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.