Ex - Bosna Sema
... In a chemical reaction, the limiting reagent is the substance which is totally consumed when the chemical reaction is complete. The amount of product formed is limited by this reagent since the reaction cannot proceed further without it. The other reagents may be present in excess of the quantities ...
... In a chemical reaction, the limiting reagent is the substance which is totally consumed when the chemical reaction is complete. The amount of product formed is limited by this reagent since the reaction cannot proceed further without it. The other reagents may be present in excess of the quantities ...
Hydrocarbons - msottchemistry
... • Does not act as 3 single bonds and 3 double bonds • It’s 6 identical bonds of intermediate length • Due to hybridization (delocalized, shared electrons) ...
... • Does not act as 3 single bonds and 3 double bonds • It’s 6 identical bonds of intermediate length • Due to hybridization (delocalized, shared electrons) ...
Exam Review_Key_All Topics.082
... 1. What is the purpose of a fractionating column and how does it work? A Fractionating column is used to separate a mixture into its component parts by differences in boiling points. The fractionating column is filled with beads that allow for condensation. The bottom of the flask is heated and the ...
... 1. What is the purpose of a fractionating column and how does it work? A Fractionating column is used to separate a mixture into its component parts by differences in boiling points. The fractionating column is filled with beads that allow for condensation. The bottom of the flask is heated and the ...
1. Chemical Energetics March
... and so there will be extra bonds to break and an extra CO2 and H2O produced per increment – perhaps you can work out the expected values starting from methanol) ...
... and so there will be extra bonds to break and an extra CO2 and H2O produced per increment – perhaps you can work out the expected values starting from methanol) ...
Aldehydes and Ketones
... Naming Aldehydes IUPAC Replace the -e in the alkane name with –al Common Add aldehyde to the prefixes form (1C), acet (2C), propion(3), and butry(4C) O ...
... Naming Aldehydes IUPAC Replace the -e in the alkane name with –al Common Add aldehyde to the prefixes form (1C), acet (2C), propion(3), and butry(4C) O ...
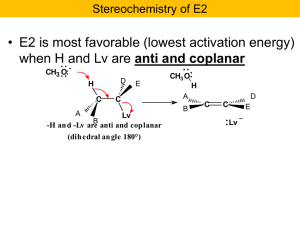
Nucleophilic Substitution and b
... Recall the problem: If reaction occurs only at the C bearing the Cl the other should remain chiral! Hmmmm? But now notice that the intermediate sulfonium ion is achiral. It has a mirror plane of symmetry. Only optically inactive products will result. ...
... Recall the problem: If reaction occurs only at the C bearing the Cl the other should remain chiral! Hmmmm? But now notice that the intermediate sulfonium ion is achiral. It has a mirror plane of symmetry. Only optically inactive products will result. ...
Indian National Chemistry Olympiad Theory 2014
... One of them is the Hoffman’s exhaustive methylation method for determining the nature of the nitrogen heterocylic ring. The heterocyclic ring is opened up with loss of nitrogen as an amine and formation of the less substituted olefin. The reactions involved are shown below. ...
... One of them is the Hoffman’s exhaustive methylation method for determining the nature of the nitrogen heterocylic ring. The heterocyclic ring is opened up with loss of nitrogen as an amine and formation of the less substituted olefin. The reactions involved are shown below. ...
1 Chemical Reactions and Equations
... Example II: Let us take a little more difficult equation, when iron is combined with steam (H2O). (i) The skeleton equation for the above reaction is Fe + H2O → Fe3O4 + H2 (ii) Then, Fe3O4 is selected which contains the maximum of atoms. It contains 4 oxygen atoms whereas there is only one oxygen ...
... Example II: Let us take a little more difficult equation, when iron is combined with steam (H2O). (i) The skeleton equation for the above reaction is Fe + H2O → Fe3O4 + H2 (ii) Then, Fe3O4 is selected which contains the maximum of atoms. It contains 4 oxygen atoms whereas there is only one oxygen ...
FahadH. Ahmad (Contact: +92 323 509 4443)
... carbon atoms because the intermolecular forces are much stronger. Carboxylic acids are weak acids, so they can donate a hydrogen ion(H+) in acidbase reactions: This means that they will react with carbonates to produce a salt, water and carbon dioxide: They will also react with reactive metals to pr ...
... carbon atoms because the intermolecular forces are much stronger. Carboxylic acids are weak acids, so they can donate a hydrogen ion(H+) in acidbase reactions: This means that they will react with carbonates to produce a salt, water and carbon dioxide: They will also react with reactive metals to pr ...
2010 Exam
... Explain using bonding theories why sodium metal bends when struck with a hammer while sodium chloride shatters. Support your answer with a labeled diagram. ...
... Explain using bonding theories why sodium metal bends when struck with a hammer while sodium chloride shatters. Support your answer with a labeled diagram. ...
9.2 Oxidation Numbers
... Phosphates, like ammonium phosphate, are important components of fertilizers used to stimulate the growth of agricultural crops and to make our gardens green. Their commercial synthesis requires elemental phosphorus, which can be acquired by heating phosphate rock (containing calcium phosphate) with ...
... Phosphates, like ammonium phosphate, are important components of fertilizers used to stimulate the growth of agricultural crops and to make our gardens green. Their commercial synthesis requires elemental phosphorus, which can be acquired by heating phosphate rock (containing calcium phosphate) with ...
2013 Final Exam File - Fiji National University
... For each molecule, arrange the 4 substituents around the stereocentre in order of priority, according to the Cahn-Ingold-Prelog Rules, and then assign the stereocentre as R or S. ...
... For each molecule, arrange the 4 substituents around the stereocentre in order of priority, according to the Cahn-Ingold-Prelog Rules, and then assign the stereocentre as R or S. ...
Chapter 25 The Chemistry of Life: Organic Chemistry 25.1 Some
... Branched-chain hydrocarbons are possible for alkanes with four or more C atoms. Structures with different branches can be written for the same formula: ...
... Branched-chain hydrocarbons are possible for alkanes with four or more C atoms. Structures with different branches can be written for the same formula: ...
KINETICS questions
... Predict the effect of each of the following changes on the initial rate of the reaction and explain your prediction. (a) Addition of hydrogen gas at constant temperature and volume (b) Increase in volume of the reaction vessel at constant temperature (c) Addition of catalyst. In your explanation, in ...
... Predict the effect of each of the following changes on the initial rate of the reaction and explain your prediction. (a) Addition of hydrogen gas at constant temperature and volume (b) Increase in volume of the reaction vessel at constant temperature (c) Addition of catalyst. In your explanation, in ...
Chapter 10
... A cyclic transition state occurs instead where one of the B-H bonds is transferred to the carbon, this process stabilizes the structure as the carbon never bears a full positive charge ...
... A cyclic transition state occurs instead where one of the B-H bonds is transferred to the carbon, this process stabilizes the structure as the carbon never bears a full positive charge ...
Unit 8 Packet
... reactant or product and percent yield using particle models from a balanced chemical equation and amount of one substance in the reaction. Given quantities of multiple reactants, students will be able to determine and use the limiting reactant. ...
... reactant or product and percent yield using particle models from a balanced chemical equation and amount of one substance in the reaction. Given quantities of multiple reactants, students will be able to determine and use the limiting reactant. ...
Synthesis of enantiopure alcohols
... Previously we have reported that the enantioselectivity (E) decreased during esterifications of a range of secondary alcohols (1-4) catalyzed by immobilized lipase B from Candida antarctica (Novozym 435) and that addition of enantiopure (R)alcohols, (R)-1, (R)-2, (R)-5, (R)-6 and (R)-7, induced incr ...
... Previously we have reported that the enantioselectivity (E) decreased during esterifications of a range of secondary alcohols (1-4) catalyzed by immobilized lipase B from Candida antarctica (Novozym 435) and that addition of enantiopure (R)alcohols, (R)-1, (R)-2, (R)-5, (R)-6 and (R)-7, induced incr ...
Document
... processes include freezing and boiling, in which a material changes form with no chemical reaction ...
... processes include freezing and boiling, in which a material changes form with no chemical reaction ...
Chloroperbenzoic_aci..
... m-CPBA.26b In this reaction, substantial amounts of nitrone as byproduct are expected. (The best method for the preparation of hydroxylamines is to oxidize the secondary amine with 2-(phenylsulfonyl)-3-aryloxaziridine (see e.g. (±) trans-2(Phenylsulfonyl)-3-phenyloxaziridine) to the nitrone, and the ...
... m-CPBA.26b In this reaction, substantial amounts of nitrone as byproduct are expected. (The best method for the preparation of hydroxylamines is to oxidize the secondary amine with 2-(phenylsulfonyl)-3-aryloxaziridine (see e.g. (±) trans-2(Phenylsulfonyl)-3-phenyloxaziridine) to the nitrone, and the ...
Requirements And Guidelines For Abstract (Title May be
... eliminate persistent organic pollutants, which cannot be completely removed with an conventional wastewater treatment methods (like adsorption, flocculation, air stripping, etc). Among these advanced technologies, TiO2 heterogeneous photocatalytic oxidation with ultraviolet (UV) irradiation source i ...
... eliminate persistent organic pollutants, which cannot be completely removed with an conventional wastewater treatment methods (like adsorption, flocculation, air stripping, etc). Among these advanced technologies, TiO2 heterogeneous photocatalytic oxidation with ultraviolet (UV) irradiation source i ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.