Microsoft Word
... sulfinyl group as the nucleophile has been disclosed. The reaction proceeds with equal facility on di– and trisubstituted alkenes to furnish products possessing tertiary and quaternary stereogenic centres. The reaction can be considered to be complimentary to other routes to stereotriads/stereotetra ...
... sulfinyl group as the nucleophile has been disclosed. The reaction proceeds with equal facility on di– and trisubstituted alkenes to furnish products possessing tertiary and quaternary stereogenic centres. The reaction can be considered to be complimentary to other routes to stereotriads/stereotetra ...
Non Flory-Schulz ethene oligomerization with titanium
... limited experimental access to structural details there is no direct proof for the structures proposed. Homogeneous olefin polymerization catalysts should eventually allow more direct observations of the catalytically active species, and hence, on the mechanism of chain growth and stereocontrol26. S ...
... limited experimental access to structural details there is no direct proof for the structures proposed. Homogeneous olefin polymerization catalysts should eventually allow more direct observations of the catalytically active species, and hence, on the mechanism of chain growth and stereocontrol26. S ...
Analysis of structure–function relationships in cytochrome c oxidase
... metals with other molecules. In respect to the design of molecular catalysts and their long term stability this protection mechanism is of high importance and should be considered carefully. 4. Functional CcO inspired metal complexes The information gained by spectroscopy on the reaction mechanism o ...
... metals with other molecules. In respect to the design of molecular catalysts and their long term stability this protection mechanism is of high importance and should be considered carefully. 4. Functional CcO inspired metal complexes The information gained by spectroscopy on the reaction mechanism o ...
Problem Authors - PianetaChimica
... The acids which are stronger than pure sulfuric acid are called superacids. Superacids are very strong proton donors being capable of protonating even weak Lewis acids such as Xe, H2, Cl 2, Br2, and CO2. Cations, which never exist in other media, have been observed in superacid solutions. George Ola ...
... The acids which are stronger than pure sulfuric acid are called superacids. Superacids are very strong proton donors being capable of protonating even weak Lewis acids such as Xe, H2, Cl 2, Br2, and CO2. Cations, which never exist in other media, have been observed in superacid solutions. George Ola ...
Ion-Pair Formation
... concentration of the cation needed for precipitation to occur decreases—and vice versa—so that Ksp is constant. Consequently, the solubility of an ionic compound depends on the concentrations of other salts that contain the same ions. This dependency is another example of the common ion effect: addi ...
... concentration of the cation needed for precipitation to occur decreases—and vice versa—so that Ksp is constant. Consequently, the solubility of an ionic compound depends on the concentrations of other salts that contain the same ions. This dependency is another example of the common ion effect: addi ...
terpyridines - Beilstein
... In the context of a more environmentally friendly and “greener” chemistry, an adaptation of this well-established method was proposed with the aim of reducing the solvent use [16-18]. Namely, two equivalents of neat 2-acetylpyridine (4) were reacted with one equivalent of an aldehyde in the presence ...
... In the context of a more environmentally friendly and “greener” chemistry, an adaptation of this well-established method was proposed with the aim of reducing the solvent use [16-18]. Namely, two equivalents of neat 2-acetylpyridine (4) were reacted with one equivalent of an aldehyde in the presence ...
Chemical Reactions and Stoichiometry
... Combustion analysis (which we saw in the previous chapter) employs a chemical reaction, a process in which one or more substances are converted into one or more different ones. Compounds form and change through chemical reactions. Water can be made by the reaction of hydrogen with oxygen. A combusti ...
... Combustion analysis (which we saw in the previous chapter) employs a chemical reaction, a process in which one or more substances are converted into one or more different ones. Compounds form and change through chemical reactions. Water can be made by the reaction of hydrogen with oxygen. A combusti ...
CH 2 - sintak
... The octane rating was developed by chemist Russel Marker at the Ethyl Corporation at 1926. The selection of n-heptane as the zero point of the scale was due to the availability of very high purity n-heptane, not mixed with other isomers of heptane or octane, distilled from the resin of the Jeffrey ...
... The octane rating was developed by chemist Russel Marker at the Ethyl Corporation at 1926. The selection of n-heptane as the zero point of the scale was due to the availability of very high purity n-heptane, not mixed with other isomers of heptane or octane, distilled from the resin of the Jeffrey ...
Problem 1-2
... "What a waste" she thinks when she recognizes that the 1 L bottles are more than halffull. ...
... "What a waste" she thinks when she recognizes that the 1 L bottles are more than halffull. ...
File
... 8. Do aromatic hydrocarbons react more than saturated or unsaturated hydrocarbons? Explain why. 9. What is a nucleophile? Which of all the nucleophiles is the most reactive? The least? 10. What is a photochemical reaction? Give an example of a hydrocarbon that undergoes this type of reaction. 11. Ho ...
... 8. Do aromatic hydrocarbons react more than saturated or unsaturated hydrocarbons? Explain why. 9. What is a nucleophile? Which of all the nucleophiles is the most reactive? The least? 10. What is a photochemical reaction? Give an example of a hydrocarbon that undergoes this type of reaction. 11. Ho ...
Palladium(II)-Catalyzed Oxidative Cyclization Strategies Andreas K. Å. Persson
... couplings, carbon-heteroatom couplings, and isomerizations.6,9 ...
... couplings, carbon-heteroatom couplings, and isomerizations.6,9 ...
Document
... • Seven resonance structures can be drawn for benzaldehyde (C6H5CHO). Because three of them place a positive charge on a carbon atom of the benzene ring, the CHO group withdraws electrons from the benzene ring by a resonance effect. ...
... • Seven resonance structures can be drawn for benzaldehyde (C6H5CHO). Because three of them place a positive charge on a carbon atom of the benzene ring, the CHO group withdraws electrons from the benzene ring by a resonance effect. ...
Electrophilic Aromatic Substitution and Substituted Benzenes
... • Seven resonance structures can be drawn for benzaldehyde (C6H5CHO). Because three of them place a positive charge on a carbon atom of the benzene ring, the CHO group withdraws electrons from the benzene ring by a resonance effect. ...
... • Seven resonance structures can be drawn for benzaldehyde (C6H5CHO). Because three of them place a positive charge on a carbon atom of the benzene ring, the CHO group withdraws electrons from the benzene ring by a resonance effect. ...
Perspectives on How Nature Employs the Principles of
... in H2 complexes) for it to be observable under ambient conditions: e.g., in regard to the mechanism of methane monooxygenase. Methane C-H bond activation may not even occur at the dinuclear Fe sites in the latter but rather at the bridging oxo ligands, involving either radical or concerted mechanism ...
... in H2 complexes) for it to be observable under ambient conditions: e.g., in regard to the mechanism of methane monooxygenase. Methane C-H bond activation may not even occur at the dinuclear Fe sites in the latter but rather at the bridging oxo ligands, involving either radical or concerted mechanism ...
- Free Documents
... Nacetylphenyl salicylideneamine Fig. Tomislav Frii. the keto oxygen atom is the hydrogen acceptor in a CarHO interaction of . in the crystal structure of N. In the crystal structure of N. a Molecular diagram. . between the imino methine and the oxygen atom of the central ketoamino group. . Nhydroxyp ...
... Nacetylphenyl salicylideneamine Fig. Tomislav Frii. the keto oxygen atom is the hydrogen acceptor in a CarHO interaction of . in the crystal structure of N. In the crystal structure of N. a Molecular diagram. . between the imino methine and the oxygen atom of the central ketoamino group. . Nhydroxyp ...
amines - Gneet`s
... Substitution of hydrogen atom of aniline with some electron donating group ( like alkyl) increases the basic strength Whereas if substitution is made by an electron – withdrawing group ( like phenyl group), the basic strength of amine decreases ...
... Substitution of hydrogen atom of aniline with some electron donating group ( like alkyl) increases the basic strength Whereas if substitution is made by an electron – withdrawing group ( like phenyl group), the basic strength of amine decreases ...
Slide 1
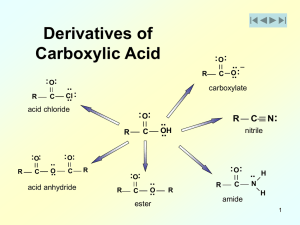
... the acid and alcohol in the presence of a small quantity of acid catalyst (H2SO4 or HCl (g)) causes ester formation (esterification) along with dehydration. The equilibrium constant is not large (Keq ~ 1) but high yields can be obtained by adding a large excess of one of the reactants and removing t ...
... the acid and alcohol in the presence of a small quantity of acid catalyst (H2SO4 or HCl (g)) causes ester formation (esterification) along with dehydration. The equilibrium constant is not large (Keq ~ 1) but high yields can be obtained by adding a large excess of one of the reactants and removing t ...
Recent developments in electrochemical and photoelectrochemical
... We have also been interested in the use of semiconducting p-type diamond electrodes for CO2 reduction, because hydrogen evolution is extremely inhibited on such surfaces.19 Indeed, using methanol as the solvent with high-pressure CO2, we found that essentially no hydrogen was produced; CO, methyl fo ...
... We have also been interested in the use of semiconducting p-type diamond electrodes for CO2 reduction, because hydrogen evolution is extremely inhibited on such surfaces.19 Indeed, using methanol as the solvent with high-pressure CO2, we found that essentially no hydrogen was produced; CO, methyl fo ...
Hydroformylation

Hydroformylation, also known as oxo synthesis or oxo process, is an important homogeneously catalyzed industrial process for the production of aldehydes from alkenes. This chemical reaction entails the addition of a formyl group (CHO) and a hydrogen atom to a carbon-carbon double bond. This process has undergone continuous growth since its invention in 1938: Production capacity reached 6.6×106 tons in 1995. It is important because the resulting aldehydes are easily converted into many secondary products. For example, the resulting aldehydes are hydrogenated to alcohols that are converted to plasticizers or detergents. Hydroformylation is also used in specialty chemicals, relevant to the organic synthesis of fragrances and natural products. The development of hydroformylation, which originated within the German coal-based industry, is considered one of the premier achievements of 20th-century industrial chemistry.The process typically entails treatment of an alkene with high pressures (between 10 to 100 atmospheres) of carbon monoxide and hydrogen at temperatures between 40 and 200 °C. Transition metal catalysts are required.