![(C3H7)3NH[CrO3X],(X=F, Cl), Reagents for Oxidation of](http://s1.studyres.com/store/data/015838257_1-b7e4138a4ed1f989d8dc5b682bb74b7a-300x300.png)
(C3H7)3NH[CrO3X],(X=F, Cl), Reagents for Oxidation of
... Oxidation of organic compounds in general, and of alcohols in particular, under mild, aprotic conditions is an important reaction in synthetic organic chemistry [1] . For this purpose different Cr(VI) based oxidants are reported in the literature. However, some of the reported chromium ...
... Oxidation of organic compounds in general, and of alcohols in particular, under mild, aprotic conditions is an important reaction in synthetic organic chemistry [1] . For this purpose different Cr(VI) based oxidants are reported in the literature. However, some of the reported chromium ...
Alcohol oxidation
... The mechanism of the Jacobsen–Katsuki epoxidation is not fully understood, but most likely a manganese(V)-species is the reactive intermediate which is formed upon the oxidation of the Mn(III)-salen complex. The most accepted mechanism is the concerted pathway mechanism. After the formation of the M ...
... The mechanism of the Jacobsen–Katsuki epoxidation is not fully understood, but most likely a manganese(V)-species is the reactive intermediate which is formed upon the oxidation of the Mn(III)-salen complex. The most accepted mechanism is the concerted pathway mechanism. After the formation of the M ...
carbonyl chemistry 1
... Thus, the carbonyl group of an aldehyde or a ketone can be protected in the form of an acetal or ketal. Deprotection following reaction on other regions of the molecule then yields the carbonyl group again – this then is the first protection/deprotection protocol we have encountered. In general, sim ...
... Thus, the carbonyl group of an aldehyde or a ketone can be protected in the form of an acetal or ketal. Deprotection following reaction on other regions of the molecule then yields the carbonyl group again – this then is the first protection/deprotection protocol we have encountered. In general, sim ...
Chapter 20 reactions of carbonyls
... Reactivity of Common Organometallic Compounds • Since both Li and Mg are very electropositive metals, organolithium (RLi) and organomagnesium (RMgX) reagents contain very polar carbon-metal bonds and are therefore very reactive reagents. • Organomagnesium reagents are called Grignard reagents. • Or ...
... Reactivity of Common Organometallic Compounds • Since both Li and Mg are very electropositive metals, organolithium (RLi) and organomagnesium (RMgX) reagents contain very polar carbon-metal bonds and are therefore very reactive reagents. • Organomagnesium reagents are called Grignard reagents. • Or ...
... epoxide opening reactions are, however, generally carried out under either acidic or basic conditions[4-6,7] and they usually require high temperatures and/or long react ion times, and side reactions, such as isomerization, epimerization and rearrangements may be induced by the alkaline conditions. ...
Phenols Like alcohols, phenols are starting materials for a wide
... o-cresol is a weaker acid than phenol; m-cresol is a stronger acid than o- or p-cresol Preparation of phenols Cannot readily replace the halide of aryl halides by -OH, since aryl halides are inert. The reaction can be performed in industry under high pressure and temperature: OH ...
... o-cresol is a weaker acid than phenol; m-cresol is a stronger acid than o- or p-cresol Preparation of phenols Cannot readily replace the halide of aryl halides by -OH, since aryl halides are inert. The reaction can be performed in industry under high pressure and temperature: OH ...
Lecture Resource ()
... • Some amines are heterocyclic compounds (or heterocycles) • Most drugs, vitamins, and many other natural products are heterocycles • A natural product is a compound synthesized by a plant or an animal ...
... • Some amines are heterocyclic compounds (or heterocycles) • Most drugs, vitamins, and many other natural products are heterocycles • A natural product is a compound synthesized by a plant or an animal ...
TOPIC 7. ELIMINATION REACTIONS (chapter 7 and parts of
... - Do the “Learning Group Problem” at the end of the chapter. - Work through the practice exam ...
... - Do the “Learning Group Problem” at the end of the chapter. - Work through the practice exam ...
ch11 by dr. Dina
... Hydroxyl groups are poor leaving groups, and as such, are often converted to alkyl halides when a good leaving group is needed Three general methods exist for conversion of alcohols to alkyl halides, depending on the classification of the alcohol and the halogen desired Reaction can occur with phos ...
... Hydroxyl groups are poor leaving groups, and as such, are often converted to alkyl halides when a good leaving group is needed Three general methods exist for conversion of alcohols to alkyl halides, depending on the classification of the alcohol and the halogen desired Reaction can occur with phos ...
Problems - Department of Chemistry HKU
... where p0 is the initial pressure and p is the final pressure of cyclopropane. What is the order and rate constant for the reaction under these conditions? 21.10 The addition of hydrogen halides to alkenes has played a fundamental role in the investigation of organic reaction mechanisms. In one study ...
... where p0 is the initial pressure and p is the final pressure of cyclopropane. What is the order and rate constant for the reaction under these conditions? 21.10 The addition of hydrogen halides to alkenes has played a fundamental role in the investigation of organic reaction mechanisms. In one study ...
Experiment #3: Asymmetric Synthesis – Use of a Chiral Manganese
... involves the oxidation of olefins (or alkenes) to epoxides, which serve as synthetic intermediates towards a wide variety of oxygen-bearing functionalities.1 For example, epoxides react with strong nucleophiles (e.g. RS- , RSi-) under basic conditions and even weak nucleophiles (e.g., H2O) will reac ...
... involves the oxidation of olefins (or alkenes) to epoxides, which serve as synthetic intermediates towards a wide variety of oxygen-bearing functionalities.1 For example, epoxides react with strong nucleophiles (e.g. RS- , RSi-) under basic conditions and even weak nucleophiles (e.g., H2O) will reac ...
Chapter 12 Alcohols from Carbonyl Compounds: Oxidation
... The starting material may be a ketone or an ester There are two routes that start with ketones (one is shown) ...
... The starting material may be a ketone or an ester There are two routes that start with ketones (one is shown) ...
Chapter 1-
... The starting material may be a ketone or an ester There are two routes that start with ketones (one is shown) ...
... The starting material may be a ketone or an ester There are two routes that start with ketones (one is shown) ...
Preparation of Aldehydes and Ketones
... NaBH4 and LiAlH4 reduce carbonyl groups but not carbon-carbon double bonds: ...
... NaBH4 and LiAlH4 reduce carbonyl groups but not carbon-carbon double bonds: ...
Ch 10 Haloalkanes n haloarenes
... KCN is ionic they can attach through C or N but C-C bond is strong than C-N bond. So alkyl cyanide is the major product but AgCN is covalent so more electronegative N can attach to C and forms isocyanides. Ans 2. Because Cl donates electron to the benzene ring by + R effect and electron density incr ...
... KCN is ionic they can attach through C or N but C-C bond is strong than C-N bond. So alkyl cyanide is the major product but AgCN is covalent so more electronegative N can attach to C and forms isocyanides. Ans 2. Because Cl donates electron to the benzene ring by + R effect and electron density incr ...
Silicon hydrides in organic synthesis
... The acceptor properties of a,@unsaturated carbonyl compounds make them excellent ligands for low-valent transition metals and, obviously, good substrates for selective reduction with group-14 hydrides. Indeed, we found that a combination of tributyltin hydride, Pd(0) catalyst and a weak acid such as ...
... The acceptor properties of a,@unsaturated carbonyl compounds make them excellent ligands for low-valent transition metals and, obviously, good substrates for selective reduction with group-14 hydrides. Indeed, we found that a combination of tributyltin hydride, Pd(0) catalyst and a weak acid such as ...
Exam 2
... 1. (10 pts) Circle what is true about Substitution and elimination reactions. a. Strong Bases (B") have conjugate acids (BH) that have small pKa values (<5) ...
... 1. (10 pts) Circle what is true about Substitution and elimination reactions. a. Strong Bases (B") have conjugate acids (BH) that have small pKa values (<5) ...
Exam 1 Review Sheet Chapter 15 Chemistry 110b
... composition and reaction (review the mechanism). Ozonolysis of alkenes (review from first semester). DIBAL reduction of esters; know the structure of the reagent and mechanism of reaction. Extend your mechanistic insights to the DIBAL reduction of nitriles. Li(Ot-Bu)3AlH reduction of acid chlorides ...
... composition and reaction (review the mechanism). Ozonolysis of alkenes (review from first semester). DIBAL reduction of esters; know the structure of the reagent and mechanism of reaction. Extend your mechanistic insights to the DIBAL reduction of nitriles. Li(Ot-Bu)3AlH reduction of acid chlorides ...
Classes and Nomenclature of Halogen Compounds
... C) Formation of organometallic compounds. Most organic chlorides, bromides, and iodides react with certain metals to give organometallic compounds, molecules with carbon-metal bonds. Grignard reagents are obtained by the reaction of alkyl or aryl halides with metallic magnesium in dry ether as t ...
... C) Formation of organometallic compounds. Most organic chlorides, bromides, and iodides react with certain metals to give organometallic compounds, molecules with carbon-metal bonds. Grignard reagents are obtained by the reaction of alkyl or aryl halides with metallic magnesium in dry ether as t ...
synthpp - Knockhardy
... Many industrial processes involve a multi stage process where functional groups are converted into other functional groups. When planning a synthetic route, chemists must consider... • the reagents required to convert one functional group into another • the presence of other functional groups - in c ...
... Many industrial processes involve a multi stage process where functional groups are converted into other functional groups. When planning a synthetic route, chemists must consider... • the reagents required to convert one functional group into another • the presence of other functional groups - in c ...
Page 1 - WordPress.com
... (i) Name the type of mechanism involved in this reaction and write an equation for each of the steps named below. ...
... (i) Name the type of mechanism involved in this reaction and write an equation for each of the steps named below. ...
CHM2210 Organic Chemistry 1
... 3. evaluating potential energy diagrams to determine the relative energy of reactants and products and to establish whether a reaction is endothermic, exothermic, endergonic or exergonic. 4. evaluating potential energy diagrams to determine the relative stability of conformers. 5. justifying the ...
... 3. evaluating potential energy diagrams to determine the relative energy of reactants and products and to establish whether a reaction is endothermic, exothermic, endergonic or exergonic. 4. evaluating potential energy diagrams to determine the relative stability of conformers. 5. justifying the ...
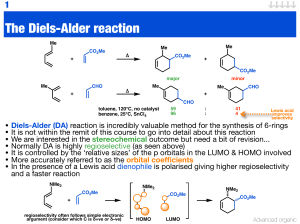
The Diels-Alder reaction
... • The fact the Diels-Alder reaction is mediated or catalysed by Lewis acids means enantioselective variants are readily carried out • The aluminium catalyst above has been utilised in enolate chemistry (aldol) reaction and is very effective in this Diels-Alder reaction ...
... • The fact the Diels-Alder reaction is mediated or catalysed by Lewis acids means enantioselective variants are readily carried out • The aluminium catalyst above has been utilised in enolate chemistry (aldol) reaction and is very effective in this Diels-Alder reaction ...
Stille reaction

The Stille reaction, or the Migita-Kosugi-Stille coupling, is a chemical reaction widely used in organic synthesis which involves the coupling of an organotin compound (also known as organostannanes) with a variety of organic electrophiles via palladium-catalyzed coupling reaction.The R1 group attached to the trialkyltin is normally sp2-hybridized, including alkenes, and aryl groups; however, conditions have been devised to incorporate both sp3-hybridized groups, such as allylic and benzylic substituents, and sp-hybridized alkynes. These organostannanes are also stable to both air and moisture, and many of these reagents are either commercially available or can be synthesized from literature precedent. However, these tin reagents tend to be highly toxic. X is typically a halide, such as Cl, Br, I, yet pseudohalides such as triflates and sulfonates and phosphates can also be used.The groundwork for the Stille reaction was laid by Colin Eaborn, Toshihiko Migita, and Masanori Kosugi in 1976 and 1977, who explored numerous palladium catalyzed couplings involving organotin reagents. John Stille and David Milstein developed a much milder and more broadly applicable procedure in 1978. Stille’s work on this area might have earned him a share of the 2010 Nobel Prize, which was awarded to Richard Heck, Ei-ichi Negishi, and Akira Suzuki for their work on the Heck, Negishi, and Suzuki coupling reactions. However, Stille died in the plane crash of United Airlines Flight 232 in 1989.Several reviews have been published on the Stille reaction.