Boron Reagents in Process Chemistry: Excellent
... trialkylboranes. With more hindered olefins, the hydroboration can be stopped at the mono- or disubstituted organoborane. For example, hydroboration of 2-methyl-2butene proceeds only to the dialkyl stage, generating the corresponding disiamylborane. A very useful regioselective hydroborating agent, ...
... trialkylboranes. With more hindered olefins, the hydroboration can be stopped at the mono- or disubstituted organoborane. For example, hydroboration of 2-methyl-2butene proceeds only to the dialkyl stage, generating the corresponding disiamylborane. A very useful regioselective hydroborating agent, ...
Document
... Many are chiral and occur in single enantiomer form. Some of these compounds are pharmacologically important, e. g., quinine and atropine. Others have dangerous natures, e. g., morphine and strychnine. Some alkaloids have been used to separate the enantiomers of chiral carboxylic acids. This resolut ...
... Many are chiral and occur in single enantiomer form. Some of these compounds are pharmacologically important, e. g., quinine and atropine. Others have dangerous natures, e. g., morphine and strychnine. Some alkaloids have been used to separate the enantiomers of chiral carboxylic acids. This resolut ...
On The catalytic Hydrogenation of Co2 and Carboxylic acid esters
... hydrogenation of CO2 and dehydrogenation of FA, these systems have three major drawbacks. Firstly, these catalysts contain polydentate phosphine ligands, that can greatly contribute to the catalyst price. Taken together with a non-trivial handling of phosphine complexes, this diminishes the benefits ...
... hydrogenation of CO2 and dehydrogenation of FA, these systems have three major drawbacks. Firstly, these catalysts contain polydentate phosphine ligands, that can greatly contribute to the catalyst price. Taken together with a non-trivial handling of phosphine complexes, this diminishes the benefits ...
iNTRODUCTiON TO ORGANiC COMPOUNDS
... hydroxy group is responsible for the characteristic properties of alcohols so we refer to it as the functional group for alcohols. There are three different methods for naming alcohols, but we will use only the IUPAC system. The rules that you used for naming alkanes and alkenes (in Chapter 25) are ...
... hydroxy group is responsible for the characteristic properties of alcohols so we refer to it as the functional group for alcohols. There are three different methods for naming alcohols, but we will use only the IUPAC system. The rules that you used for naming alkanes and alkenes (in Chapter 25) are ...
A review of new developments in the Friedel–Crafts - Beilstein
... formed during this transformation, namely water. Waste water treatment is an ongoing and expensive issue in large scale chemical processes; therefore, methodologies without the formation of water or other by-products would be even more desirable. As such, hydroarylations and hydroalkylations with a ...
... formed during this transformation, namely water. Waste water treatment is an ongoing and expensive issue in large scale chemical processes; therefore, methodologies without the formation of water or other by-products would be even more desirable. As such, hydroarylations and hydroalkylations with a ...
Resolution of Diols via Catalytic Asymmetric Acetalization
... of different aldehydes revealed that efficient kinetic resolution of diol rac-2a can be achieved in the presence of only 1−2 mol% of 1a to give a variety of 1,3-dioxolanes 4 with excellent enantioselectivities and diastereoselectivities (Table 1). Replacing isovaleraldehyde with less bulky n-pentanal o ...
... of different aldehydes revealed that efficient kinetic resolution of diol rac-2a can be achieved in the presence of only 1−2 mol% of 1a to give a variety of 1,3-dioxolanes 4 with excellent enantioselectivities and diastereoselectivities (Table 1). Replacing isovaleraldehyde with less bulky n-pentanal o ...
Organic Chemistry Package 2011
... becomes hopeless when larger alkanes are considered. For example there are 5 isomers of hexane (C6H14), 9 isomers of heptane (C7H16) and 75 isomers of decane (C10H22). Another problem arises as far as nomenclature is concerned. Hydrogens can be replaced or substituted by'other elements or groups. Fo ...
... becomes hopeless when larger alkanes are considered. For example there are 5 isomers of hexane (C6H14), 9 isomers of heptane (C7H16) and 75 isomers of decane (C10H22). Another problem arises as far as nomenclature is concerned. Hydrogens can be replaced or substituted by'other elements or groups. Fo ...
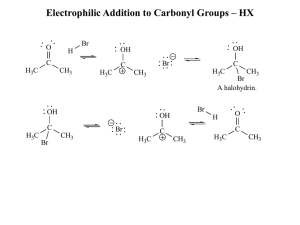
Electrophilic Addition to Carbonyl Groups – HX
... • If an aldol reaction is worked up under acidic conditions, an E2 reaction will follow, giving a double bond conjugated to the carbonyl group: ...
... • If an aldol reaction is worked up under acidic conditions, an E2 reaction will follow, giving a double bond conjugated to the carbonyl group: ...
Oxidative reactions ppt - Senior Chemistry
... •This reaction is generally done by passing the vapors of the alcohol through a tube furnace in a stream of inert carrier gas. •This is not a practical laboratory method -- it is better suited to industrial processes. •The reaction stops at the aldehyde stage -- no more removal of hydrogen can take ...
... •This reaction is generally done by passing the vapors of the alcohol through a tube furnace in a stream of inert carrier gas. •This is not a practical laboratory method -- it is better suited to industrial processes. •The reaction stops at the aldehyde stage -- no more removal of hydrogen can take ...
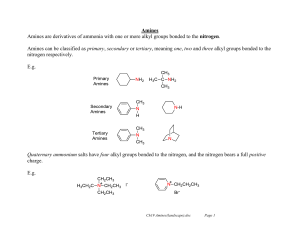
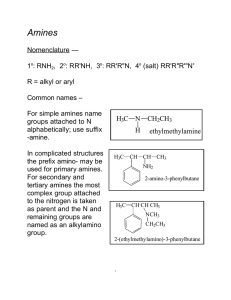
Amines Amines are derivatives of ammonia with one or more alkyl
... To achieve a conformation suitable for E2 to occur, C-3 must rotate and place a Hydrogen anti to the bulky leaving group. To remove the proton from C-1 ( Hofmann product), any of the three staggered conformations allow the E2 mechanism to operate. The Hofmann product dominates since elimination of ...
... To achieve a conformation suitable for E2 to occur, C-3 must rotate and place a Hydrogen anti to the bulky leaving group. To remove the proton from C-1 ( Hofmann product), any of the three staggered conformations allow the E2 mechanism to operate. The Hofmann product dominates since elimination of ...
Retrosynthetic Analysis of Nabumetone
... Grignard reactions that produce secondary alcohols, will require oxidation to the corresponding ketone to arrive at the desired product. Starting with the bond between the ring and the side chain, we are given two pairs of synthons as shown in Scheme 2. The synthetic equivalent to the aromatic anion ...
... Grignard reactions that produce secondary alcohols, will require oxidation to the corresponding ketone to arrive at the desired product. Starting with the bond between the ring and the side chain, we are given two pairs of synthons as shown in Scheme 2. The synthetic equivalent to the aromatic anion ...
Proofs to - Research Explorer
... [Fe{=C(H)Me}(dppe)Cp*][PF6], [25]. The X-ray crystal structures of the oxygen heteroatomstabilised carbene complexes 3[I] and 5[PF6] have therefore been determined and important bond lengths and angles are summarised in Table 1 together with comparative data for related cyclopentadienyl iron and rut ...
... [Fe{=C(H)Me}(dppe)Cp*][PF6], [25]. The X-ray crystal structures of the oxygen heteroatomstabilised carbene complexes 3[I] and 5[PF6] have therefore been determined and important bond lengths and angles are summarised in Table 1 together with comparative data for related cyclopentadienyl iron and rut ...
1. Alcohol Oxidations
... Oxidation of 1° Alcohols to Aldehydes [Cu(II)/O2] Marko, I. E.; Giles, P. R.; Tsukazaki, M.; Brown, S. M.; Urch, C. J. Science 1996, 274, 2044. Oxidation proceeds with O2 in the presence of a catalytic amount (5 mol%) of a copper complex, which can be formed in situ from CuCl2, phenanthroline, and a ...
... Oxidation of 1° Alcohols to Aldehydes [Cu(II)/O2] Marko, I. E.; Giles, P. R.; Tsukazaki, M.; Brown, S. M.; Urch, C. J. Science 1996, 274, 2044. Oxidation proceeds with O2 in the presence of a catalytic amount (5 mol%) of a copper complex, which can be formed in situ from CuCl2, phenanthroline, and a ...
A. 3-chloro-1-ethoxybutane B. p
... 12. (3 points) The boiling points of aldehydes and ketones differ from those of alcohols and alkenes due to: a. Hydrogen bonding between like molecules (aldehyde with aldehyde) b. Hydrogen bonding with water c. Dipolar bonding between like molecules (aldehyde with aldehyde) d. Both b and c e. Both a ...
... 12. (3 points) The boiling points of aldehydes and ketones differ from those of alcohols and alkenes due to: a. Hydrogen bonding between like molecules (aldehyde with aldehyde) b. Hydrogen bonding with water c. Dipolar bonding between like molecules (aldehyde with aldehyde) d. Both b and c e. Both a ...
Catalytic Asymmetric Induction. Highly Enantioselective Addition of
... Benzaldehyde does not react with diethylzinc at 0 "C in toluene. When a 1:1 :1 or 1 :2:2 mixture of benzaldehyde, diethylzinc, and (-)-DAIB in toluene was allowed to stand at 0 "C, the aldehyde was consumed slowly but only benzyl alcohol was obtained. No ethylation product could be detected. If, how ...
... Benzaldehyde does not react with diethylzinc at 0 "C in toluene. When a 1:1 :1 or 1 :2:2 mixture of benzaldehyde, diethylzinc, and (-)-DAIB in toluene was allowed to stand at 0 "C, the aldehyde was consumed slowly but only benzyl alcohol was obtained. No ethylation product could be detected. If, how ...
226 amines lec
... Azo compounds are often used as dyes. They are colored owing to extensive conjugation. For example, butter yellow was at one time used to color margarine yellow. It has been found to be carcinogenic and is no longer used for this purpose. ...
... Azo compounds are often used as dyes. They are colored owing to extensive conjugation. For example, butter yellow was at one time used to color margarine yellow. It has been found to be carcinogenic and is no longer used for this purpose. ...
AMIDES AND AMINES: ORGANIC NITROGEN COMPOUNDS
... Amines: Low molar-mass amines are flammable gases with an ammonia-like odor. Aliphatic amines up to six carbon atoms are water soluble. Many amines have a “fishy” odor and many have very foul odors. Aromatic amines occur as liquids and solids. Soluble aliphatic amines give basic solutions. Aromatic ...
... Amines: Low molar-mass amines are flammable gases with an ammonia-like odor. Aliphatic amines up to six carbon atoms are water soluble. Many amines have a “fishy” odor and many have very foul odors. Aromatic amines occur as liquids and solids. Soluble aliphatic amines give basic solutions. Aromatic ...
Ethers - Home - KSU Faculty Member websites
... Cleavage of ethers by hot concentrated acids Ethers are quite stable compounds. Ethers react only under strongly acidic condition.When ethers are heated in concentrated acid ...
... Cleavage of ethers by hot concentrated acids Ethers are quite stable compounds. Ethers react only under strongly acidic condition.When ethers are heated in concentrated acid ...
OChem1 Course Pack
... The better the possible carbocation, the faster the SN1 reaction; 3° > 2° > 1° > CH3 ...
... The better the possible carbocation, the faster the SN1 reaction; 3° > 2° > 1° > CH3 ...
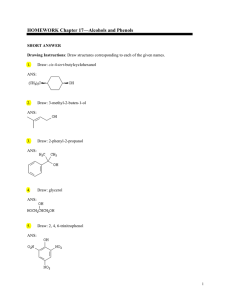
HOMEWORK Chapter 17—Alcohols and Phenols
... In E2 elimination, dehydration proceeds most readily when the two groups to be eliminated have a trans-diaxial relationship. In this compound, the only hydrogen with the proper geometric relationship to the −OH group is at C6 so the major product of this reaction is 3methylcyclohexene. ...
... In E2 elimination, dehydration proceeds most readily when the two groups to be eliminated have a trans-diaxial relationship. In this compound, the only hydrogen with the proper geometric relationship to the −OH group is at C6 so the major product of this reaction is 3methylcyclohexene. ...
Alkene

In organic chemistry, an alkene is an unsaturated hydrocarbon that contains at least one carbon–carbon double bond. Alkene, olefin, and olefine are used often interchangeably (see nomenclature section below). Acyclic alkenes, with only one double bond and no other functional groups, known as mono-enes, form a homologous series of hydrocarbons with the general formula CnH2n. Alkenes have two hydrogen atoms less than the corresponding alkane (with the same number of carbon atoms). The simplest alkene, ethylene (C2H4), which has the International Union of Pure and Applied Chemistry (IUPAC) name ethene is the organic compound produced on the largest scale industrially. Aromatic compounds are often drawn as cyclic alkenes, but their structure and properties are different and they are not considered to be alkenes.



![[Ru(Triphos)H2(CO)] Characterisation - Durham e](http://s1.studyres.com/store/data/017676948_1-4352644236c53cc416f065328f560d26-300x300.png)