Chapter One
... known, its chemical composition can be expressed as the mass percent of each element in the compound (percent composition), and vice versa. When solving this kind of problems, we can use masses expressed in a.m.u. or in g/mol ...
... known, its chemical composition can be expressed as the mass percent of each element in the compound (percent composition), and vice versa. When solving this kind of problems, we can use masses expressed in a.m.u. or in g/mol ...
Writing Chemical Equations KClO3 O2 (g) + KCl (s) Balancing
... In this example we started with 2 atoms of hydrogen and we ended with 2 atoms of hydrogen. But we started with only one atom of oxygen and ended with 2 atoms of oxygen. We cannot change the numbers at the bottom of the symbols because that is how the formulas must be written. To balance the equation ...
... In this example we started with 2 atoms of hydrogen and we ended with 2 atoms of hydrogen. But we started with only one atom of oxygen and ended with 2 atoms of oxygen. We cannot change the numbers at the bottom of the symbols because that is how the formulas must be written. To balance the equation ...
Chapter 23 (Section 3) Pregnancy, Birth, and Childhood (Pages 735
... c. some MATTER exists in elemental form [(e.g.) gold [Au] = not chemically REACTIVE)] *d. ELEMENTS individually or combined form everything in the universe including HUMANS *1. Human body’s most abundant ELEMENTS: carbon [C], oxygen [O], hydrogen [H], and nitrogen [N]; for teeth & BONES = calcium [C ...
... c. some MATTER exists in elemental form [(e.g.) gold [Au] = not chemically REACTIVE)] *d. ELEMENTS individually or combined form everything in the universe including HUMANS *1. Human body’s most abundant ELEMENTS: carbon [C], oxygen [O], hydrogen [H], and nitrogen [N]; for teeth & BONES = calcium [C ...
Fall Exam 1
... Assuming that you could travel at this speed on the Hyperloop, how long would it take to go from Lexington to Chicago, IL (358.8 miles)? A. 36.0 minutes C. 102 minutes B. 6.0 minutes D. 60.0 minutes 14. A box has a volume of 2.83 ft3. What is its volume in liters? A. 0.0863 L C. 80.1 L B. 0.100 L D. ...
... Assuming that you could travel at this speed on the Hyperloop, how long would it take to go from Lexington to Chicago, IL (358.8 miles)? A. 36.0 minutes C. 102 minutes B. 6.0 minutes D. 60.0 minutes 14. A box has a volume of 2.83 ft3. What is its volume in liters? A. 0.0863 L C. 80.1 L B. 0.100 L D. ...
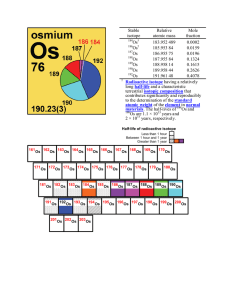
Stable isotope Relative atomic mass Mole fraction Os 183.952 489
... normal material – a reasonably possible source for an element or its compounds in commerce, for industry or science; the material is not itself studied for some extraordinary anomaly and its mole fractions (isotopic abundances) have not been modified significantly in a geologically brief period [4] ...
... normal material – a reasonably possible source for an element or its compounds in commerce, for industry or science; the material is not itself studied for some extraordinary anomaly and its mole fractions (isotopic abundances) have not been modified significantly in a geologically brief period [4] ...
Chemistry Major Understandings
... 3.1q Matter is classified as a pure substance or as a mixture of substances. 3.1r Apure substance (element or compound) has a constant composition and constant properties throughout a given sample, and from sample to sample. 3.1s Mixtures are composed of two or more different substances that can be ...
... 3.1q Matter is classified as a pure substance or as a mixture of substances. 3.1r Apure substance (element or compound) has a constant composition and constant properties throughout a given sample, and from sample to sample. 3.1s Mixtures are composed of two or more different substances that can be ...
File - Mr. Holz`s Website
... Monomer – A molecule that is able to bond with other identical molecules to form polymers Polymer – A molecule that is made up of two or more monomers Dehydration Synthesis – Process by which monomers are covalently bonded to form polymers; water is a byproduct Hydrolysis – Process by which polymers ...
... Monomer – A molecule that is able to bond with other identical molecules to form polymers Polymer – A molecule that is made up of two or more monomers Dehydration Synthesis – Process by which monomers are covalently bonded to form polymers; water is a byproduct Hydrolysis – Process by which polymers ...
South Pasadena • AP Chemistry
... Which measurement below is NOT written with three significant digits? a) 2.00 cm c) 0.003 L b) 550. grams d) 12.7 mm ...
... Which measurement below is NOT written with three significant digits? a) 2.00 cm c) 0.003 L b) 550. grams d) 12.7 mm ...
Chemistry Midterm Review Study Guide 2012
... 4. a. Which has the larger radius, Al or In? In b. Which has the larger radius, Se or Ca? Ca c. Which has a larger radius, Ca or Ca+2 Ca (would get smaller if lost 2 e-) d. Which has greater ionization energies as a class, metals or nonmetals? nonmetals e. Which has the greater ionization energy, As ...
... 4. a. Which has the larger radius, Al or In? In b. Which has the larger radius, Se or Ca? Ca c. Which has a larger radius, Ca or Ca+2 Ca (would get smaller if lost 2 e-) d. Which has greater ionization energies as a class, metals or nonmetals? nonmetals e. Which has the greater ionization energy, As ...
CP Chemistry Final Exam Review Sheet
... 52. What is a cation (and how is it created)? positively charged ion formed by losing of electrons example: Ca+2 is a cation which loses two negatively charged electrons to become positively charged 53. What is an anion (and how is it created)? negatively charged ion formed from the gaining of elect ...
... 52. What is a cation (and how is it created)? positively charged ion formed by losing of electrons example: Ca+2 is a cation which loses two negatively charged electrons to become positively charged 53. What is an anion (and how is it created)? negatively charged ion formed from the gaining of elect ...
South Pasadena · AP Chemistry
... 3. Round 2.6765 to two significant figures. 4. Write the following numbers in scientific notation. a. 560000 b. 33400 c. 0.0004120 8. A student measures the mass of a beaker filled with corn oil. The mass reading averages 215.6 g. The mass of the beaker is 110.4 g. a. What is the mass of the corn oi ...
... 3. Round 2.6765 to two significant figures. 4. Write the following numbers in scientific notation. a. 560000 b. 33400 c. 0.0004120 8. A student measures the mass of a beaker filled with corn oil. The mass reading averages 215.6 g. The mass of the beaker is 110.4 g. a. What is the mass of the corn oi ...
Chemistry Comes Alive: Part A
... • Energy may be converted from one form to another • Conversion is inefficient because some energy is ―lost‖ as heat Composition of Matter • Elements • Cannot be broken down by ordinary chemical means • Each has unique properties: • Physical properties • Are detectable with our senses, or are measur ...
... • Energy may be converted from one form to another • Conversion is inefficient because some energy is ―lost‖ as heat Composition of Matter • Elements • Cannot be broken down by ordinary chemical means • Each has unique properties: • Physical properties • Are detectable with our senses, or are measur ...
Safety - Wando High School
... 1. What makes a covalent bond? What makes an ionic bond? 2. What happens with the electrons in an ionic and covalent bond? 3. Why do atoms bond? 4. In a chemical formula what do the symbols and numbers represent? 5. What is a molecule? Is CO2 a molecule? Is NaCl a molecule? 6. What is an elements ox ...
... 1. What makes a covalent bond? What makes an ionic bond? 2. What happens with the electrons in an ionic and covalent bond? 3. Why do atoms bond? 4. In a chemical formula what do the symbols and numbers represent? 5. What is a molecule? Is CO2 a molecule? Is NaCl a molecule? 6. What is an elements ox ...
Unit 2 matter - Kowenscience.com
... established that all samples of a given compound have the same proportions, by mass, of the elements present in the compound. • A chemical compound always contains the same elements in the ...
... established that all samples of a given compound have the same proportions, by mass, of the elements present in the compound. • A chemical compound always contains the same elements in the ...
What You Need to Know to Pass the Chemistry
... Substances in a mixture may be separated by their size, polarity, density, boiling and freezing points, and solubility (among others). Filtration and distillation are examples of processes used to separate mixtures. 2. An element is a substance composed of atoms with the same atomic number. They ...
... Substances in a mixture may be separated by their size, polarity, density, boiling and freezing points, and solubility (among others). Filtration and distillation are examples of processes used to separate mixtures. 2. An element is a substance composed of atoms with the same atomic number. They ...
Exam Review - hrsbstaff.ednet.ns.ca
... d) names 20. The period number in the periodic table designates the __ for the row. a) total nuclear charge c) maximum number of outer electrons b) maximum number of nucleons d) highest energy level 21. The radii of the atoms become smaller from sodium to chlorine across period 3. This is primarily ...
... d) names 20. The period number in the periodic table designates the __ for the row. a) total nuclear charge c) maximum number of outer electrons b) maximum number of nucleons d) highest energy level 21. The radii of the atoms become smaller from sodium to chlorine across period 3. This is primarily ...
Exam Review – Part 1
... • e.g. NaCl is called sodium chloride • Al2O3 is called aluminum oxide • MgCl3 is called _________________ • Do you get it??? • You should memorize the first 20 elements (charges)so that you don’t have to waste time looking them up. ...
... • e.g. NaCl is called sodium chloride • Al2O3 is called aluminum oxide • MgCl3 is called _________________ • Do you get it??? • You should memorize the first 20 elements (charges)so that you don’t have to waste time looking them up. ...
Chapter 1 Chemistry: the study of the composition of matter and the
... Conversion questions. For example, how many grams in 27.4 kilograms? Significant figures a. Count them, round to a specific significant figure, add/subtract/multiply/divide Write in scientific notation Is it accurate? Precise? What is the percent error? Be able to solve for the missing value in a de ...
... Conversion questions. For example, how many grams in 27.4 kilograms? Significant figures a. Count them, round to a specific significant figure, add/subtract/multiply/divide Write in scientific notation Is it accurate? Precise? What is the percent error? Be able to solve for the missing value in a de ...
File - docstover.org
... Atomic number = _______, Mass number = _____ # of protons = ________ # of electrons = ______, # of neutrons = ______. Atomic Masses: What is the difference between the mass number for Carbon–14 and carbon’s atomic mass of 12.011 amu? ...
... Atomic number = _______, Mass number = _____ # of protons = ________ # of electrons = ______, # of neutrons = ______. Atomic Masses: What is the difference between the mass number for Carbon–14 and carbon’s atomic mass of 12.011 amu? ...
Figure 2: Alternative Periodic Table
... b) If a new element, X, with atomic weight 25.84 is discovered, what would its properties be? Where would it fit in the periodic table you constructed? Placed in table above using blue electrons. We predict it to be a colorless gas with low electrical conductivity and high electrical reactivity. c) ...
... b) If a new element, X, with atomic weight 25.84 is discovered, what would its properties be? Where would it fit in the periodic table you constructed? Placed in table above using blue electrons. We predict it to be a colorless gas with low electrical conductivity and high electrical reactivity. c) ...
Lecture 2 - Columbia University
... space relative to one another in three dimensions. The H 4 hydrogen atoms point to the vertices of a tetrahedron. ...
... space relative to one another in three dimensions. The H 4 hydrogen atoms point to the vertices of a tetrahedron. ...
Flexbook - What is Matter?
... in air, while chlorine is a very deadly, poisonous gas. When these two are combined, we get table salt (sodium chloride). When sodium chloride is in solid form, many units join together, as illustrated above on the right. Elements: Names and Symbols ...
... in air, while chlorine is a very deadly, poisonous gas. When these two are combined, we get table salt (sodium chloride). When sodium chloride is in solid form, many units join together, as illustrated above on the right. Elements: Names and Symbols ...
Fundamentals Fall Final Review
... 21. Know how to calculate the number of protons, neutrons and electrons in an atom when given its isotope mass and atomic number. As an example: Iron (Fe) has an atomic number of 26. An isotope of iron has a mass of 57 amu. Calculate the number of protons, neutrons and electrons in an atom of this i ...
... 21. Know how to calculate the number of protons, neutrons and electrons in an atom when given its isotope mass and atomic number. As an example: Iron (Fe) has an atomic number of 26. An isotope of iron has a mass of 57 amu. Calculate the number of protons, neutrons and electrons in an atom of this i ...