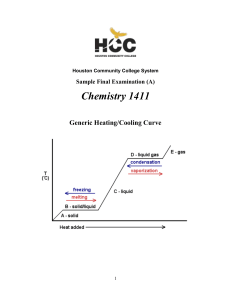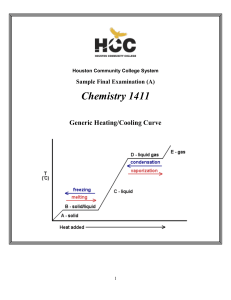
CHEM-1411 Final Practice Exam
... overall geometry is tetrahedral and the molecular geometry is trigonal pyramidal. The hybridization of the sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizatio ...
... overall geometry is tetrahedral and the molecular geometry is trigonal pyramidal. The hybridization of the sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizatio ...
10/18/11 - Note: Once it is downloaded, click SET
... (orbital- probable location of each electron - at least 2 electrons per orbital - no electrons=no orbital) Quantum Numbers- a number that describes the properties of electrons and consists of 4 numbers (Quantum- a certain number) Pauli’s Exclusion Principle- only 2 electrons can occupy a single orbi ...
... (orbital- probable location of each electron - at least 2 electrons per orbital - no electrons=no orbital) Quantum Numbers- a number that describes the properties of electrons and consists of 4 numbers (Quantum- a certain number) Pauli’s Exclusion Principle- only 2 electrons can occupy a single orbi ...
Synthesis, characterization and bonding of Ba Li Sn
... Sn (rod, Alfa, 99.999%) was used as received. All reactions were carried out in sealed niobium tubes, which were in turn jacketed and sealed under vacuum in silica ampoules. More details on the techniques and the procedures are described elsewhere [12]. Initially, Ba 3 Li 4 Sn 8 was synthesized as t ...
... Sn (rod, Alfa, 99.999%) was used as received. All reactions were carried out in sealed niobium tubes, which were in turn jacketed and sealed under vacuum in silica ampoules. More details on the techniques and the procedures are described elsewhere [12]. Initially, Ba 3 Li 4 Sn 8 was synthesized as t ...
Synthesis and structure characterization of zinc and cadmium
... The diffraction pattern of 1 was indexed giving a monoclinic unit cell (a = 29.4580 Å, b = 5.3011 Å, c = 7.4989 Å, b = 95.60241, V = 1168.2 Å3) with a figure of merit of 26.07. Systematic maxima absences suggested C2, Cm and C2/m as probable space groups. Following the fact that the starting dipeptid ...
... The diffraction pattern of 1 was indexed giving a monoclinic unit cell (a = 29.4580 Å, b = 5.3011 Å, c = 7.4989 Å, b = 95.60241, V = 1168.2 Å3) with a figure of merit of 26.07. Systematic maxima absences suggested C2, Cm and C2/m as probable space groups. Following the fact that the starting dipeptid ...
Week 1 - School of Chemical Sciences
... Electron Counting Step 1: Determine the oxidation state of the metal. To do this, balance the ligand charges with an equal opposite charge on the metal. This is the metal's formal oxidation state. ...
... Electron Counting Step 1: Determine the oxidation state of the metal. To do this, balance the ligand charges with an equal opposite charge on the metal. This is the metal's formal oxidation state. ...
Synthesis and structure characterization of zinc
... The diffraction pattern of 1 was indexed giving a monoclinic unit cell (a = 29.4580 Å, b = 5.3011 Å, c = 7.4989 Å, b = 95.60241, V = 1168.2 Å3) with a figure of merit of 26.07. Systematic maxima absences suggested C2, Cm and C2/m as probable space groups. Following the fact that the starting dipeptid ...
... The diffraction pattern of 1 was indexed giving a monoclinic unit cell (a = 29.4580 Å, b = 5.3011 Å, c = 7.4989 Å, b = 95.60241, V = 1168.2 Å3) with a figure of merit of 26.07. Systematic maxima absences suggested C2, Cm and C2/m as probable space groups. Following the fact that the starting dipeptid ...
The Transition Metal-Nitrogen Multiple Bond
... Numerous nitrido complexes of transition metals show very short metal-nitrogen bond lengths, suggesting M=N-triple bonds. At present, compounds of this type are being intensively investigated. In particular the molybdenum complexes are considered as model substances for the study of at least an inte ...
... Numerous nitrido complexes of transition metals show very short metal-nitrogen bond lengths, suggesting M=N-triple bonds. At present, compounds of this type are being intensively investigated. In particular the molybdenum complexes are considered as model substances for the study of at least an inte ...
(i) Coordination entity
... attached to the central metal atom/ion. They are commonly Octahedral, Square-planar or Tetrahedral Oxidation number: The charge that the central atom would carry if all the ligands are removed along with their pairs of electrons shared with the central atom. It is represented in parenthesis. viii) H ...
... attached to the central metal atom/ion. They are commonly Octahedral, Square-planar or Tetrahedral Oxidation number: The charge that the central atom would carry if all the ligands are removed along with their pairs of electrons shared with the central atom. It is represented in parenthesis. viii) H ...
The intersecting-state model: a link between
... exponential factors of Eq. (1) by m 2; or as smaller bond extension, given by the division of d ³ by m 2: The classical rate constants calculated by our formalism are in good agreement with the experimental data collected in the 300±1000 K temperature range. This is illustrated in Fig. 2 using t ...
... exponential factors of Eq. (1) by m 2; or as smaller bond extension, given by the division of d ³ by m 2: The classical rate constants calculated by our formalism are in good agreement with the experimental data collected in the 300±1000 K temperature range. This is illustrated in Fig. 2 using t ...
1411FINALSAMPLEs and Key
... overall geometry is tetrahedral and the molecular geometry is trigonal pyramidal. The hybridization of the sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizatio ...
... overall geometry is tetrahedral and the molecular geometry is trigonal pyramidal. The hybridization of the sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizatio ...
5.1-Organizing the elements - Environmental-Chemistry
... The role of electrons in formation of positive and negative ions • The periodic trends in the periodic table are the result of electron arrangement. • Valence electron: Electrons in outer most energy level or outer most shell in an atom are called valance electrons. • Valence electrons account for ...
... The role of electrons in formation of positive and negative ions • The periodic trends in the periodic table are the result of electron arrangement. • Valence electron: Electrons in outer most energy level or outer most shell in an atom are called valance electrons. • Valence electrons account for ...
Chapter 23 - Transition Metals and Coordination Chemistry
... - secondary valence : coordination # number of atoms bonded to metal ion - 6 for these cobalt complexes - explained the two forms of [Co(NH3)4Cl2]+ complexes cis and trans ...
... - secondary valence : coordination # number of atoms bonded to metal ion - 6 for these cobalt complexes - explained the two forms of [Co(NH3)4Cl2]+ complexes cis and trans ...
Chem 11 Notes Booklet (pdf version)
... atom. It is an averaged mass number called the atomic mass and will be used later. 4. Ions a) Creating Ions We know that atoms are electrically neutral because they have equal numbers of protons (p+) and electrons (e-). When atoms join together, though, they can lose or gain electrons. This caus ...
... atom. It is an averaged mass number called the atomic mass and will be used later. 4. Ions a) Creating Ions We know that atoms are electrically neutral because they have equal numbers of protons (p+) and electrons (e-). When atoms join together, though, they can lose or gain electrons. This caus ...
Tuning PCP-Ir Complexes: The impact of an N
... multiplets that correspond to the diastereotopic protons of the two NCH2 groups appear at δ 4.41–4.24 and 4.69–4.54 ppm for 2, and δ 5.00–4.84 and 4.64–4.50 ppm for 3. The 13C NMR spectra show triplets at δ 160.6 ppm for 2 and 161.2 ppm for 3, both with a JC-P of 3.4 Hz, which can be attributed to t ...
... multiplets that correspond to the diastereotopic protons of the two NCH2 groups appear at δ 4.41–4.24 and 4.69–4.54 ppm for 2, and δ 5.00–4.84 and 4.64–4.50 ppm for 3. The 13C NMR spectra show triplets at δ 160.6 ppm for 2 and 161.2 ppm for 3, both with a JC-P of 3.4 Hz, which can be attributed to t ...
Density functional studies on lanthanide (III) texaphyrins (Ln
... is at most 0.5 eV), whereas for basis A the difference to B and C mounts up to 1.4 eV. Therefore the average values for basis B and C are calculated and used for the discussions below. 3.1. Molecular structures The most important geometrical parameters of the fully optimized structures are listed in ...
... is at most 0.5 eV), whereas for basis A the difference to B and C mounts up to 1.4 eV. Therefore the average values for basis B and C are calculated and used for the discussions below. 3.1. Molecular structures The most important geometrical parameters of the fully optimized structures are listed in ...
Openstax - Chemistry - Answer Key
... 1. The starting materials consist of one green sphere and two purple spheres. The products consist of two green spheres and two purple spheres. This violates Dalton’s postulate that that atoms are not created during a chemical change, but are merely redistributed. 3. This statement violates Dalton’s ...
... 1. The starting materials consist of one green sphere and two purple spheres. The products consist of two green spheres and two purple spheres. This violates Dalton’s postulate that that atoms are not created during a chemical change, but are merely redistributed. 3. This statement violates Dalton’s ...
1 Course Code– CH1141 Semester – I Credit
... 8. Which is more stable O2 or O22+ ? 9. What is the enthalpy for elementary substances in their standard state? 10. What is the unit of dipole moment? 10x1 = 10 marks ...
... 8. Which is more stable O2 or O22+ ? 9. What is the enthalpy for elementary substances in their standard state? 10. What is the unit of dipole moment? 10x1 = 10 marks ...
structures of simple solids
... The Ionic Model by Goldschmidt states that are ions are essentially charged, incompressible, nonpolarisable spheres. More sophisticated models assume ions are composed of two parts: A central, hard, unperturbable core, where most electron density is concentrated. A soft, polarisable outer sphere, wh ...
... The Ionic Model by Goldschmidt states that are ions are essentially charged, incompressible, nonpolarisable spheres. More sophisticated models assume ions are composed of two parts: A central, hard, unperturbable core, where most electron density is concentrated. A soft, polarisable outer sphere, wh ...
Complexes of the Group 5 Elements
... to the V24+ species as long as it is protected from oxygen. The V–V distance of 1.924(2) Å is the same within 3m to that in K(THF)3[V2(DPhF)4] (1.929(1) Å). This supports the notion that the presence or absence of the alkali metals in the pockets between paddles does not alter the metal–metal intera ...
... to the V24+ species as long as it is protected from oxygen. The V–V distance of 1.924(2) Å is the same within 3m to that in K(THF)3[V2(DPhF)4] (1.929(1) Å). This supports the notion that the presence or absence of the alkali metals in the pockets between paddles does not alter the metal–metal intera ...
Atomic Orbitals
... – The heavier elements, particularly the 6th period and beyond (Cs, Ba, La, …). We notice it most for Tl - Bi. ...
... – The heavier elements, particularly the 6th period and beyond (Cs, Ba, La, …). We notice it most for Tl - Bi. ...
Interactlons of NO and CO with Pd and Pt Atoms
... Gavezzotti et alez6performed Hartree-Fock (HF) calculations = 0.95). Relativistic effective core potentials (RECPs) and the on PtCO using a relativistic pseudopotential for Pt. Rohfling and Pd and Pt basis sets of Hay and Wadt31awere used to represent Hay27performed unrestricted Hartree-Fock (UHF) w ...
... Gavezzotti et alez6performed Hartree-Fock (HF) calculations = 0.95). Relativistic effective core potentials (RECPs) and the on PtCO using a relativistic pseudopotential for Pt. Rohfling and Pd and Pt basis sets of Hay and Wadt31awere used to represent Hay27performed unrestricted Hartree-Fock (UHF) w ...
AP Chemistry - Oak Park Unified School District
... being at a particular point in space is given by its (3). An orbital is described by a combination of four quantum numbers. The principal quantum number, n, is indicated by the integers 1, 2, 3. . . 7. This quantum number relates to the (4) and energy of the orbital. The sublevel quantum number, l, ...
... being at a particular point in space is given by its (3). An orbital is described by a combination of four quantum numbers. The principal quantum number, n, is indicated by the integers 1, 2, 3. . . 7. This quantum number relates to the (4) and energy of the orbital. The sublevel quantum number, l, ...
Grade 11 Review Package
... This statement can be used to predict trends in the properties of the elements. Figure R.5 summarizes the periodic trends of four properties of atoms: atomic size, ionization energy, electron affinity, and electronegativity. These four properties affect the structure of molecules and ions, and they ...
... This statement can be used to predict trends in the properties of the elements. Figure R.5 summarizes the periodic trends of four properties of atoms: atomic size, ionization energy, electron affinity, and electronegativity. These four properties affect the structure of molecules and ions, and they ...
covalent - Typepad
... Identify the letter of the choice that best completes the statement or answers the question. 1. In the compound sodium fluoride, NaF, the sodium atom loses one electron and the fluorine atom gains one electron to form ions that are isoelectric with a. helium. c. neon. b. oxygen. d. calcium. 2. Atoms ...
... Identify the letter of the choice that best completes the statement or answers the question. 1. In the compound sodium fluoride, NaF, the sodium atom loses one electron and the fluorine atom gains one electron to form ions that are isoelectric with a. helium. c. neon. b. oxygen. d. calcium. 2. Atoms ...