Answers to examination questions
... The oxygen molecule (O O) contains one double bond; the carbon dioxide molecule (O C O) contains two double bond and the tetrafluoroethene molecule contains one double bond. ...
... The oxygen molecule (O O) contains one double bond; the carbon dioxide molecule (O C O) contains two double bond and the tetrafluoroethene molecule contains one double bond. ...
4 - WebAssign
... IUPAC (International Union of Pure and Applied Chemistry) rules for naming compounds in which the cation can have more than one charge: Name the metal, give its charge in Roman numerals, in parentheses, then name the anion. Give charge only when necessary. Copyright © Houghton Mifflin Company. All r ...
... IUPAC (International Union of Pure and Applied Chemistry) rules for naming compounds in which the cation can have more than one charge: Name the metal, give its charge in Roman numerals, in parentheses, then name the anion. Give charge only when necessary. Copyright © Houghton Mifflin Company. All r ...
Communicating Research to the General Public
... chemistry’s periodic table – are examples of elements, or materials consisting of only one type of atom. There are about 115 known elements, many of which do not occur naturally. Each element contains only atoms with a characteristic number of electrons and protons. 8.2.2. Atoms can combine to make ...
... chemistry’s periodic table – are examples of elements, or materials consisting of only one type of atom. There are about 115 known elements, many of which do not occur naturally. Each element contains only atoms with a characteristic number of electrons and protons. 8.2.2. Atoms can combine to make ...
Chemistry Notes for class 12 Chapter 9 Coordination
... www.ncerthelp.com (Visit for all ncert solutions in text and videos, CBSE syllabus, note and many more) ...
... www.ncerthelp.com (Visit for all ncert solutions in text and videos, CBSE syllabus, note and many more) ...
Sample pages 2 PDF
... For centuries, human beings have found inspiration in nature, from macro-scale to micro-scale. Animals have been inspiring designs of cars, robotics, and even computational algorithms based on their behaviors. Some new super tough materials got inspired in deer antlers. Environmental analysis of pre ...
... For centuries, human beings have found inspiration in nature, from macro-scale to micro-scale. Animals have been inspiring designs of cars, robotics, and even computational algorithms based on their behaviors. Some new super tough materials got inspired in deer antlers. Environmental analysis of pre ...
Structure / Nomenclature Guide
... without charges or bonds. Structural formulae are not given for these substances in the monographs since the molecular formula is considered to be sufficient, except in the case of co-ordination compounds for which the stereochemistry of the complexes must be specified (see section A-6). If metal sa ...
... without charges or bonds. Structural formulae are not given for these substances in the monographs since the molecular formula is considered to be sufficient, except in the case of co-ordination compounds for which the stereochemistry of the complexes must be specified (see section A-6). If metal sa ...
Chapter 24 Chemistry of Coordination Compounds
... metal and the charges on the ligands, one can calculate the charge on the complex ion. Chemistry of Coordination Compounds ...
... metal and the charges on the ligands, one can calculate the charge on the complex ion. Chemistry of Coordination Compounds ...
Energetics Past Paper Questions
... heat loss (to the surroundings); insulate the reaction vessel/use a lid/draw a temperature versus time graph; 3.5°C/temperature change would be the same; amount of base reacted would be the same/excess acid would not react/ KOH is the limiting reagent amount of energy needed to break one mole of (co ...
... heat loss (to the surroundings); insulate the reaction vessel/use a lid/draw a temperature versus time graph; 3.5°C/temperature change would be the same; amount of base reacted would be the same/excess acid would not react/ KOH is the limiting reagent amount of energy needed to break one mole of (co ...
H - Deans Community High School
... The dependent variable (what you measured), Safety The method mentioning all the equipment used and measurements made, readings and variable kept constant/changed etc 7. A table (with headings) of your measurements, and a sample average and rate = 1/t calculation) and your line graph. 8. Your conclu ...
... The dependent variable (what you measured), Safety The method mentioning all the equipment used and measurements made, readings and variable kept constant/changed etc 7. A table (with headings) of your measurements, and a sample average and rate = 1/t calculation) and your line graph. 8. Your conclu ...
Ultimate removal mechanisms of elements from the ocean*
... for may indicate that Cr in pelagic clays are in part absorbed as Cr(OHh. The high Cop/C,w for F- is due to the formation of apatite in addition to the adsorption onto cx-FeO(OH), probably as = Fe-F. The Fe-F bond strength is again inversely related to log K!. Since log K! for HCI and HBr are so lar ...
... for may indicate that Cr in pelagic clays are in part absorbed as Cr(OHh. The high Cop/C,w for F- is due to the formation of apatite in addition to the adsorption onto cx-FeO(OH), probably as = Fe-F. The Fe-F bond strength is again inversely related to log K!. Since log K! for HCI and HBr are so lar ...
Chemistry Problem Solving Drill
... XPS (also known as ESCA) can detect composition of elements in the first 1-12 nm of the surface. XPS uses X-rays to eject electrons from the sample. The energy of the ejected electron corresponds to its binding energy in the atomic electronic structure. The number of electrons ejected is related to ...
... XPS (also known as ESCA) can detect composition of elements in the first 1-12 nm of the surface. XPS uses X-rays to eject electrons from the sample. The energy of the ejected electron corresponds to its binding energy in the atomic electronic structure. The number of electrons ejected is related to ...
Hydrogen-bond supramolecular structure of group 13 Schiff base
... the different ionic radii of aluminum and gallium, and crystallize in the tetragonal space group I41 /a as solvates with disordered molecules of n-hexane. Adjacent monomeric moieties are linked by the C Himino · · ·O hydrogen bonds with the aryloxide oxygen acting as the hydrogen acceptor. This lead ...
... the different ionic radii of aluminum and gallium, and crystallize in the tetragonal space group I41 /a as solvates with disordered molecules of n-hexane. Adjacent monomeric moieties are linked by the C Himino · · ·O hydrogen bonds with the aryloxide oxygen acting as the hydrogen acceptor. This lead ...
1)A neutral atom has no overall charge, and ion is a
... 5)a)Create graph, will be gone over in class. b)These are the smallest atoms on each of their respective rows, and electrons are being removed from filled orbitals, which have strong stability, which takes a lot of energy to do. c)The valence electrons experience a smaller nuclear force of attractio ...
... 5)a)Create graph, will be gone over in class. b)These are the smallest atoms on each of their respective rows, and electrons are being removed from filled orbitals, which have strong stability, which takes a lot of energy to do. c)The valence electrons experience a smaller nuclear force of attractio ...
Fe(H2O)6
... There is no change in the occupation of the eg* orbitals, just in the high spin Fe2+ / Fe3+ system. However, the Fe2+ system with its (t2g)6 configuration has an optimal electron count for π-bonding with the bipyridyl ring: it is an 18 e- complex. Thus, even though Fe3+ has a high charge, the Fe-N b ...
... There is no change in the occupation of the eg* orbitals, just in the high spin Fe2+ / Fe3+ system. However, the Fe2+ system with its (t2g)6 configuration has an optimal electron count for π-bonding with the bipyridyl ring: it is an 18 e- complex. Thus, even though Fe3+ has a high charge, the Fe-N b ...
Local structure of LiNi0.5Mn0.5O2 cathode material probed by in
... and less toxic. The x-ray absorption near-edge structure 共XANES兲 study on this solid solution revealed that the Ni is in the Ni2+ state.1 Our recent study on the similar layered material Li共Ni1/3Co1/3Mn1/3兲O2 confirmed the involvement of the Ni2+ ↔ Ni3+ ↔ Ni4+ redox couple in the 2.7– 4 V region.19 ...
... and less toxic. The x-ray absorption near-edge structure 共XANES兲 study on this solid solution revealed that the Ni is in the Ni2+ state.1 Our recent study on the similar layered material Li共Ni1/3Co1/3Mn1/3兲O2 confirmed the involvement of the Ni2+ ↔ Ni3+ ↔ Ni4+ redox couple in the 2.7– 4 V region.19 ...
Controlling Aggregation of Copper(II)
... modes (e.g., copper(II)) are less predictable.13,14 In this case, the directing and templating effects of the ligands are weaker, and other variables, most notably the coordination or hydrogen bonding abilities of the solvent or counterion, can cause changes as significant as a different cluster nuc ...
... modes (e.g., copper(II)) are less predictable.13,14 In this case, the directing and templating effects of the ligands are weaker, and other variables, most notably the coordination or hydrogen bonding abilities of the solvent or counterion, can cause changes as significant as a different cluster nuc ...
Lecture19_Ch19_11140..
... Werner’s explanation of coordination complexes Metal ions exhibit two kinds of valence: primary and secondary valences The primary valence is the oxidation number (positive charge) of the metal (usually 2+ or 3+) The secondary valence is the number of atoms that are directly bonded (coordinated) to ...
... Werner’s explanation of coordination complexes Metal ions exhibit two kinds of valence: primary and secondary valences The primary valence is the oxidation number (positive charge) of the metal (usually 2+ or 3+) The secondary valence is the number of atoms that are directly bonded (coordinated) to ...
Cu(II) and Zn(II) Complexes with an N- and O
... structural feature in 1 is that the adjacent layers recognize each other through strongly offset π–π stacking interactions, ultimately leading to a 3D supramolecular framework (Fig. 1e). The centroid-centroid distance between the central benzene rings is 3.662 Å. ...
... structural feature in 1 is that the adjacent layers recognize each other through strongly offset π–π stacking interactions, ultimately leading to a 3D supramolecular framework (Fig. 1e). The centroid-centroid distance between the central benzene rings is 3.662 Å. ...
PDF w - American Chemical Society
... that the electrostatic contribution is of larger importance than charge transfer since the lighter inorganic halides exhibit a higher negative electrostatic potential. It is, however, not known whether these trends are completely consistent for all transition metals M−X acceptors as only a limited n ...
... that the electrostatic contribution is of larger importance than charge transfer since the lighter inorganic halides exhibit a higher negative electrostatic potential. It is, however, not known whether these trends are completely consistent for all transition metals M−X acceptors as only a limited n ...
Activation of C–F bonds using Cp*2ZrH2: a
... Cp*2ZrH2 also reacts rapidly with certain fluorinated olefins. For example, 1 (3 equiv.) and 1,1-difluoroethylene react at ⫺80 ⬚C to give Cp*2Zr(C2H5)H and Cp*2ZrHF. No intermediates are observed, and attempts to use limiting quantities of 1 result in smaller conversions to the same products. This reac ...
... Cp*2ZrH2 also reacts rapidly with certain fluorinated olefins. For example, 1 (3 equiv.) and 1,1-difluoroethylene react at ⫺80 ⬚C to give Cp*2Zr(C2H5)H and Cp*2ZrHF. No intermediates are observed, and attempts to use limiting quantities of 1 result in smaller conversions to the same products. This reac ...
- Free Documents
... The first stable dihydrogen metal complex was isolated by Kubas. The lengthened HH bond . is greater than the HH bond length in free H .. This is thought to arise from metal backbonding into the HH orbital. ...
... The first stable dihydrogen metal complex was isolated by Kubas. The lengthened HH bond . is greater than the HH bond length in free H .. This is thought to arise from metal backbonding into the HH orbital. ...
Metal Contaminants - environmentalgeochemistry
... ■ The principles of HSAB can be used to predict the speciation of transition metals in subsurface systems as well as their relative toxicity. ■ The speciation of transition metals is more affect by the presence of natural organic matter than the speciation of the base cations. ■ Likewise, changes in ...
... ■ The principles of HSAB can be used to predict the speciation of transition metals in subsurface systems as well as their relative toxicity. ■ The speciation of transition metals is more affect by the presence of natural organic matter than the speciation of the base cations. ■ Likewise, changes in ...
2.6 M - Thierry Karsenti
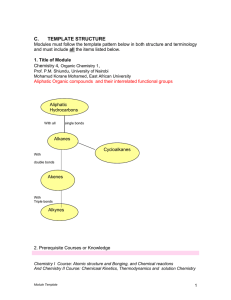
... atoms in the cyclic structure is other than carbon. Heterocyclic componds may be aliphatic or aromatic 15. Isomers: These are different compounds that have the same molecular formula. Isomers are further subdivided into: (a) structural isomers, (b) geometrical isomers and (c) stereoisomers(optical i ...
... atoms in the cyclic structure is other than carbon. Heterocyclic componds may be aliphatic or aromatic 15. Isomers: These are different compounds that have the same molecular formula. Isomers are further subdivided into: (a) structural isomers, (b) geometrical isomers and (c) stereoisomers(optical i ...
Answers - University of Waterloo
... Use the following information and diagram to answer questions 7-10. A galvanic cell is constructed by placing a strip of zinc into a 1.0 mol L−1 solution of zinc nitrate and a strip of aluminum into a 1.0 mol L−1 solution of aluminum nitrate. The two metal strips are connected to a voltmeter by wire ...
... Use the following information and diagram to answer questions 7-10. A galvanic cell is constructed by placing a strip of zinc into a 1.0 mol L−1 solution of zinc nitrate and a strip of aluminum into a 1.0 mol L−1 solution of aluminum nitrate. The two metal strips are connected to a voltmeter by wire ...
Four-Electron Oxidative Formation of Aryl Diazenes Using a
... was localized, giving a localized quinone–phenol structure for the ligand, which was discernable in the ligand C C and C O bond lengths.[17] We suspect that the bond lengths observed for the [ONOq] ligand of 6 reflect averaged values resulting from both orientations of the [ONOq] ligand in the solid ...
... was localized, giving a localized quinone–phenol structure for the ligand, which was discernable in the ligand C C and C O bond lengths.[17] We suspect that the bond lengths observed for the [ONOq] ligand of 6 reflect averaged values resulting from both orientations of the [ONOq] ligand in the solid ...