
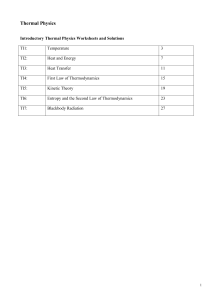
- Pcpolytechnic
... external source, the temperature of the gas will increases and piston will rise. As piston rises , the boundary of the system moves. In other words the heat and work energy crosses the boundary of the system during this process. But there is no addition or loss of original mass of the working substa ...
... external source, the temperature of the gas will increases and piston will rise. As piston rises , the boundary of the system moves. In other words the heat and work energy crosses the boundary of the system during this process. But there is no addition or loss of original mass of the working substa ...
Chemical Engineering Thermodynamics
... to spontaneously change (i.e. there are no net driving forces for change). As such, equilibrium can only occur for closed (and isolated) systems. If an open system does not change with time as it undergoes a process, it is said to be in steady-state. With this, we will again define some important co ...
... to spontaneously change (i.e. there are no net driving forces for change). As such, equilibrium can only occur for closed (and isolated) systems. If an open system does not change with time as it undergoes a process, it is said to be in steady-state. With this, we will again define some important co ...
Energy and the First Law of Thermodynamics
... • The internal energy is a state function, which is dependent only on the present state of the system, and not on the pathway by which it got to that state. – Some examples of state functions include energy (and many other thermodynamic terms), pressure, volume, altitude, distance, etc. • An energy ...
... • The internal energy is a state function, which is dependent only on the present state of the system, and not on the pathway by which it got to that state. – Some examples of state functions include energy (and many other thermodynamic terms), pressure, volume, altitude, distance, etc. • An energy ...
Screen Version
... One way of stating the Second Law of Thermodynamics is: “only by transferring heat from a warmer to a colder body can heat can be converted into work in a cyclic process.” It can be shown that no engine can be more efficient than a reversible engine working between the same limits of temperature, a ...
... One way of stating the Second Law of Thermodynamics is: “only by transferring heat from a warmer to a colder body can heat can be converted into work in a cyclic process.” It can be shown that no engine can be more efficient than a reversible engine working between the same limits of temperature, a ...
27 Oct. 2010 - PHA Science
... Being exothermic (“exothermicity”) favors spontaneity, but does not guarantee it! An endothermic reaction can be spontaneous. Not all exothermic reactions are spontaneous. So how do we predict if a reaction is spontaneous under given conditions if ∆H doesn't help us?! ...
... Being exothermic (“exothermicity”) favors spontaneity, but does not guarantee it! An endothermic reaction can be spontaneous. Not all exothermic reactions are spontaneous. So how do we predict if a reaction is spontaneous under given conditions if ∆H doesn't help us?! ...
Unit II - Chemical Thermodynamics
... The method by which the state of a system is changed is called a “Process”. It can be effected by changing any one of the state variables viz P,T,C etc. Isothermal process: It is a process carried out at const temperature. Exchange of heat takes place between system and surroundings dT = 0 Adiabatic ...
... The method by which the state of a system is changed is called a “Process”. It can be effected by changing any one of the state variables viz P,T,C etc. Isothermal process: It is a process carried out at const temperature. Exchange of heat takes place between system and surroundings dT = 0 Adiabatic ...
heat
... engine transform the potential energy into useful work, which can be used to propel the car. ...
... engine transform the potential energy into useful work, which can be used to propel the car. ...
Chapter 22-1 - UCF College of Sciences
... Now, suppose that the block hits a fixed wall and stops. This inelastic collision is clearly an irreversible process. The ordered mechanical energy of the gas is converted into random internal energy and the temperature of the gas rises. The gas still has the same total energy, but now all of the ...
... Now, suppose that the block hits a fixed wall and stops. This inelastic collision is clearly an irreversible process. The ordered mechanical energy of the gas is converted into random internal energy and the temperature of the gas rises. The gas still has the same total energy, but now all of the ...
The Second Law of Thermodynamics and Entropy
... cylinder is held at a pressure largerth,an the ambient pressure by means of a piston loaded down with a weight, as shown in Figure 46.4(a), To simplify the example, jet us also assume that the system is originally at ambient temperature and that it is perfectly thermally insulated from the environme ...
... cylinder is held at a pressure largerth,an the ambient pressure by means of a piston loaded down with a weight, as shown in Figure 46.4(a), To simplify the example, jet us also assume that the system is originally at ambient temperature and that it is perfectly thermally insulated from the environme ...
2 nd Law of Thermodynamics
... General Features of the Entropy S • It is a state function, so that ΔS between given macrostates is independent of the path. • It is a quantitative measure of the disorder in a system. • It gives a criterion for the direction of a process, since an isolated system will reach a state of maximum entr ...
... General Features of the Entropy S • It is a state function, so that ΔS between given macrostates is independent of the path. • It is a quantitative measure of the disorder in a system. • It gives a criterion for the direction of a process, since an isolated system will reach a state of maximum entr ...
II. THE FIRST LAW OF THERMODYNAMICS AND RELATED
... where q is again the net thermal energy (per unit mass) passing into the system from the surroundings. [Thermal energy can be defined as the potential and kinetic energies that change as a consequence of a temperature change.] Note that this last expression deals with the classification of energy pa ...
... where q is again the net thermal energy (per unit mass) passing into the system from the surroundings. [Thermal energy can be defined as the potential and kinetic energies that change as a consequence of a temperature change.] Note that this last expression deals with the classification of energy pa ...
Ans_PS08b_full_121 F16
... “gets there” it is no longer considered to be “heat”—it merely contributes to the internal energy getting larger.) ...
... “gets there” it is no longer considered to be “heat”—it merely contributes to the internal energy getting larger.) ...
06_00 AP PPT Thermochemistry
... EU 3.C – Chemical and physical transformations may be observed in several ways and typically involve a change in energy. EK 3.C.2 – Net changes in energy for a chemical reaction can be endothermic or exothermic. EU 5.A – Two systems with different temperatures that are in thermal contact will excha ...
... EU 3.C – Chemical and physical transformations may be observed in several ways and typically involve a change in energy. EK 3.C.2 – Net changes in energy for a chemical reaction can be endothermic or exothermic. EU 5.A – Two systems with different temperatures that are in thermal contact will excha ...
Heat Engines and the First Law of Thermodynamics
... it. Observations like these caused physicists and engineers in the middle of the nineteenth century to conclude that heat is just a form of energy, the form that flows when there is a temperature difference between two objects. We want to examine the mathematical relationship between the heat energy ...
... it. Observations like these caused physicists and engineers in the middle of the nineteenth century to conclude that heat is just a form of energy, the form that flows when there is a temperature difference between two objects. We want to examine the mathematical relationship between the heat energy ...
Heat transfer

Heat transfer is the exchange of thermal energy between physical systems, depending on the temperature and pressure, by dissipating heat. The fundamental modes of heat transfer are conduction or diffusion, convection and radiation.Heat transfer always occurs from a region of high temperature to another region of lower temperature. Heat transfer changes the internal energy of both systems involved according to the First Law of Thermodynamics. The Second Law of Thermodynamics defines the concept of thermodynamic entropy, by measurable heat transfer.Thermal equilibrium is reached when all involved bodies and the surroundings reach the same temperature. Thermal expansion is the tendency of matter to change in volume in response to a change in temperature.










![BTD QUESTION BANK[1].](http://s1.studyres.com/store/data/009330461_1-f5de3108f7a7a17ebe3a8cbd391865db-300x300.png)











