An Unified Approach to Thermodynamics of Power Yield in Thermal
... mass transferred through ‘conductances’ or boundary layers. The energy flux (power) is created in each generator located between the resource stream (‘upper’ fluid 1) and, say, an waste stream (‘lower’ fluid, 2). Basically, both transfer mechanisms, flows and values of conductances of boundary layer ...
... mass transferred through ‘conductances’ or boundary layers. The energy flux (power) is created in each generator located between the resource stream (‘upper’ fluid 1) and, say, an waste stream (‘lower’ fluid, 2). Basically, both transfer mechanisms, flows and values of conductances of boundary layer ...
Energy Conservation in Ethanol-Water Distillation
... compressed by the application of work to a higher pressure. The resulting high pressure vapour flows to the condenser where it condenses, giving up its latent heat at a high temperature, before expanding back to a low pressure liquid. The heat pump cycle may be connected to a distillation column in ...
... compressed by the application of work to a higher pressure. The resulting high pressure vapour flows to the condenser where it condenses, giving up its latent heat at a high temperature, before expanding back to a low pressure liquid. The heat pump cycle may be connected to a distillation column in ...
Second Law of Thermodynamics
... All the processes listed above require energy input to occur just as a refrigerator requires electrical energy to run. Systems can become more ordered and lower their entropy as time passes. However, this can happen only as the entropy of the environment increases, just as we found out in the case o ...
... All the processes listed above require energy input to occur just as a refrigerator requires electrical energy to run. Systems can become more ordered and lower their entropy as time passes. However, this can happen only as the entropy of the environment increases, just as we found out in the case o ...
Pdf - Text of NPTEL IIT Video Lectures
... The compressor which compresses the air to a higher pressure and temperature then the high pressure and temperature air goes to a combustion chamber where fuel is being injected and burned with the air and a temperature is raised to a much higher value the state point3. The fluid or working system ...
... The compressor which compresses the air to a higher pressure and temperature then the high pressure and temperature air goes to a combustion chamber where fuel is being injected and burned with the air and a temperature is raised to a much higher value the state point3. The fluid or working system ...
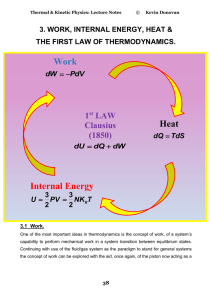
Internal Energy Work Heat
... represented by a perfect differential while an imperfect differential represents how that change was carried out (a process or flow). NB W is NOT a function of state! If a large change is made in the volume it is necessary to find the net effect through integration and care has to be taken in evalua ...
... represented by a perfect differential while an imperfect differential represents how that change was carried out (a process or flow). NB W is NOT a function of state! If a large change is made in the volume it is necessary to find the net effect through integration and care has to be taken in evalua ...
Thermodynamics - WordPress.com
... calorimeter and ∆U was found to be -742.7 kJ mol-1 of cyanamide at 298K. Calculate the enthalpy change for the reaction at 298 K. NH2CN(s) + 3/2 O2 (g) → N2(g) + CO2(g) +H2O( l ) 2. Calculate the number of kJ necessary to raise the temperature of 60 g of aluminium from 35o to 55o C. Molar heat capac ...
... calorimeter and ∆U was found to be -742.7 kJ mol-1 of cyanamide at 298K. Calculate the enthalpy change for the reaction at 298 K. NH2CN(s) + 3/2 O2 (g) → N2(g) + CO2(g) +H2O( l ) 2. Calculate the number of kJ necessary to raise the temperature of 60 g of aluminium from 35o to 55o C. Molar heat capac ...
HEAT AND TEMPERATURE
... speed). From this argument it is clear that there has to be a lower limit on temperature. As a gas is cooled, the molecules move more and more slowly until they eventually have no kinetic energy left - i.e. they are stationary. At this point the gas can no longer exert a pressure; as gases exert pre ...
... speed). From this argument it is clear that there has to be a lower limit on temperature. As a gas is cooled, the molecules move more and more slowly until they eventually have no kinetic energy left - i.e. they are stationary. At this point the gas can no longer exert a pressure; as gases exert pre ...
THERMODYNAMICS LECTURE NOTES
... compared to molecular dimensions, and therefore a system (to be defined next) contains many molecules, and this is called continuum. The concept of continuum loses validity when the mean free path of molecules approaches the order of typical system dimensions. The State Postulate The state of the sy ...
... compared to molecular dimensions, and therefore a system (to be defined next) contains many molecules, and this is called continuum. The concept of continuum loses validity when the mean free path of molecules approaches the order of typical system dimensions. The State Postulate The state of the sy ...
EGR 107 FALL 2001
... methods this can be solved for an ideal gas (tables or constant specific heats). Discuss two methods this can be solved for liquids (tables or constant specific heats). b. For a constant pressure process in a closed system q h ; list any assumptions required. Discuss two methods this can be solve ...
... methods this can be solved for an ideal gas (tables or constant specific heats). Discuss two methods this can be solved for liquids (tables or constant specific heats). b. For a constant pressure process in a closed system q h ; list any assumptions required. Discuss two methods this can be solve ...
Meaning of Entropy in Classical Thermodynamics
... e is similar to an energy parameter that Gibbs called the modulus θ. The equation of state above is obtained using the kinetic theory, which is part of statistical mechanics. This attests to the central role of statistical physics in the foundation of classical thermodynamics. For an adiabatic proce ...
... e is similar to an energy parameter that Gibbs called the modulus θ. The equation of state above is obtained using the kinetic theory, which is part of statistical mechanics. This attests to the central role of statistical physics in the foundation of classical thermodynamics. For an adiabatic proce ...
Fluids and Thermo Review
... A fluid is any substance that flows, typically a liquid or a gas. Hydrostatics is the study of fluids at rest, such as the pressure of a fluid at a particular depth, or the buoyant force acting on an object in a fluid. Archimedes principle states that the buoyant force acting on an object in a fluid ...
... A fluid is any substance that flows, typically a liquid or a gas. Hydrostatics is the study of fluids at rest, such as the pressure of a fluid at a particular depth, or the buoyant force acting on an object in a fluid. Archimedes principle states that the buoyant force acting on an object in a fluid ...
First Law of Thermodynamics
... liquid are loosely bound and may mix with one another freely. (While a liquid has a definite volume, it still takes the shape of its container. The molecules of a gas interact with each other slightly, but usually move at higher speeds than that of solid of liquid. In all three states of matter the ...
... liquid are loosely bound and may mix with one another freely. (While a liquid has a definite volume, it still takes the shape of its container. The molecules of a gas interact with each other slightly, but usually move at higher speeds than that of solid of liquid. In all three states of matter the ...
Fundamentals of Thermodynamics Applied to Thermal
... point as seen in Fig. 1. Therefore, above the critical pressure and temperature there will be no liquid–vapor phase change. The process illustrated in Fig. 1 takes place at a constant pressure, known as isobaric, which is imposed on the system by the piston weight plus local atmospheric pressure. Ot ...
... point as seen in Fig. 1. Therefore, above the critical pressure and temperature there will be no liquid–vapor phase change. The process illustrated in Fig. 1 takes place at a constant pressure, known as isobaric, which is imposed on the system by the piston weight plus local atmospheric pressure. Ot ...
Heat transfer

Heat transfer is the exchange of thermal energy between physical systems, depending on the temperature and pressure, by dissipating heat. The fundamental modes of heat transfer are conduction or diffusion, convection and radiation.Heat transfer always occurs from a region of high temperature to another region of lower temperature. Heat transfer changes the internal energy of both systems involved according to the First Law of Thermodynamics. The Second Law of Thermodynamics defines the concept of thermodynamic entropy, by measurable heat transfer.Thermal equilibrium is reached when all involved bodies and the surroundings reach the same temperature. Thermal expansion is the tendency of matter to change in volume in response to a change in temperature.