Thermodynamics
... a. Enthalpy can be a constant pressure system and defined as the summation of a system + the pressure and volume. b. If there is an increase in energy of a system: (H = E + PV), you will find that H is nothing but the heat energy of the system. The first law of thermodynamics tells you enthalpy ...
... a. Enthalpy can be a constant pressure system and defined as the summation of a system + the pressure and volume. b. If there is an increase in energy of a system: (H = E + PV), you will find that H is nothing but the heat energy of the system. The first law of thermodynamics tells you enthalpy ...
2. Thermodynamic Processes and Quantities Defined
... at the expense of the internal energy of the gas causing the temperature of the gas to drop. The figure below shows P-V diagrams for these two processes. ...
... at the expense of the internal energy of the gas causing the temperature of the gas to drop. The figure below shows P-V diagrams for these two processes. ...
Properties and Classification of Matter
... ◦ A new substance is produced with its own properties ◦ Involved a flow of energy (endothermic & exothermic reactions) ◦ Many cause a phase change (i.e. gas formation) ...
... ◦ A new substance is produced with its own properties ◦ Involved a flow of energy (endothermic & exothermic reactions) ◦ Many cause a phase change (i.e. gas formation) ...
Temperature and Thermal Energy
... thermometer in your mouth and wait for a few minutes before checking the thermometer for your temperature reading. The atomic level process involved in measuring temperature involves collisions and energy transfers between the thermometer and your body. ...
... thermometer in your mouth and wait for a few minutes before checking the thermometer for your temperature reading. The atomic level process involved in measuring temperature involves collisions and energy transfers between the thermometer and your body. ...
Thermodynamics Chapter 4
... • Kinetic energy is possessed by a molecule as it moves through space. It transfers this energy to other systems by means of collisions in which its linear momentum changes. Collisions with such things as thermometers and thermocouples are the basis for temperature measurement. • It is a characteris ...
... • Kinetic energy is possessed by a molecule as it moves through space. It transfers this energy to other systems by means of collisions in which its linear momentum changes. Collisions with such things as thermometers and thermocouples are the basis for temperature measurement. • It is a characteris ...
Temperature, Thermal Energy, and Heat
... a good example of how convection works. As the water at the bottom of the pot heats up, the molecules begin to move faster and their kinetic energy increases, causing them to spread apart. The water expands and becomes less dense than the surrounding water. As a result, it rises to the surface, wher ...
... a good example of how convection works. As the water at the bottom of the pot heats up, the molecules begin to move faster and their kinetic energy increases, causing them to spread apart. The water expands and becomes less dense than the surrounding water. As a result, it rises to the surface, wher ...
Le Châtelier`s Principle
... Increase in the concentration of sulfur dioxide: The equilibrium shifts right to use up the extra SO2, producing more sulfur trioxide and using up some oxygen. Decreasing the partial pressure of sulfur trioxide: This is equivalent to decreasing the concentration of sulfur trioxide. The equilibrium w ...
... Increase in the concentration of sulfur dioxide: The equilibrium shifts right to use up the extra SO2, producing more sulfur trioxide and using up some oxygen. Decreasing the partial pressure of sulfur trioxide: This is equivalent to decreasing the concentration of sulfur trioxide. The equilibrium w ...
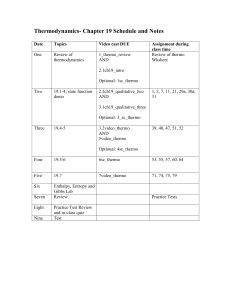
10 PRE-LABORATORY ASSIGNMENT EXPERIMENT 7 1. Is t
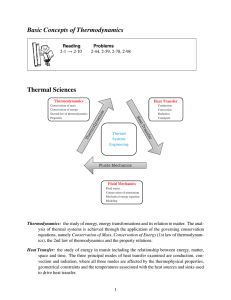
... Thermochemistry is the study of the relationship between chemical reactions and energy changes. Thermochemistry has many practical applications. For example, using thermochemistry: (1) mining engineers can calculate how much fuel will be needed to prepare metals from their ores, (2) structural ...
... Thermochemistry is the study of the relationship between chemical reactions and energy changes. Thermochemistry has many practical applications. For example, using thermochemistry: (1) mining engineers can calculate how much fuel will be needed to prepare metals from their ores, (2) structural ...
SAT Subject Physics Formula Reference Kinematics
... A heat engine essentially converts heat into work. The engine does work by absorbing heat from a hot reservoir and discarding some heat to a cold reservoir. The formula gives the quality (“efficiency”) of the engine. ...
... A heat engine essentially converts heat into work. The engine does work by absorbing heat from a hot reservoir and discarding some heat to a cold reservoir. The formula gives the quality (“efficiency”) of the engine. ...
transport processes and cross-coupling effects in non
... reactions; the state-to-state approach considering the internal (vibrational or ro-vibrational) energy exchanges as slow processes and thus suitable for extremely non-equilibrium situations (the price for this is the great number of fluid-dynamic equations which have to be solved for the populations ...
... reactions; the state-to-state approach considering the internal (vibrational or ro-vibrational) energy exchanges as slow processes and thus suitable for extremely non-equilibrium situations (the price for this is the great number of fluid-dynamic equations which have to be solved for the populations ...
Chapter 4 - UniMAP Portal
... The equations in the figure are valid for any substance undergoing any process. ...
... The equations in the figure are valid for any substance undergoing any process. ...
Notes #2 Chem 341
... Heat effect is measured by measuring a temp. change in the liquid surrounding the reaction vessel. The heat capacity of the calorimeter (surroundings) is determined in a separate experiment by passing a given amount of electrical energy through a heating coil immersed in the same reaction vessel. ...
... Heat effect is measured by measuring a temp. change in the liquid surrounding the reaction vessel. The heat capacity of the calorimeter (surroundings) is determined in a separate experiment by passing a given amount of electrical energy through a heating coil immersed in the same reaction vessel. ...
Chap-3
... LW; cooling by emission of LW) – latent heating (associated with water phase changes). ...
... LW; cooling by emission of LW) – latent heating (associated with water phase changes). ...
1 CHAPTER 8 HEAT CAPACITY, AND THE EXPANSION OF GASES
... gases was one of the several failures of classical theory that helped to give rise to the birth of quantum theory. Quantum theory in fact accounts spectacularly well and in detail for the specific heat capacities of molecules and how the heat capacities vary with temperature. This topic is often dea ...
... gases was one of the several failures of classical theory that helped to give rise to the birth of quantum theory. Quantum theory in fact accounts spectacularly well and in detail for the specific heat capacities of molecules and how the heat capacities vary with temperature. This topic is often dea ...