Syllabus and Regulations for 2-year, 4
... shall forward assessment in respect of every candidate to the Principal / Controller of Examination / Coordinator P. G. Courses (as the case may be) for tabulation of the results. 3.(a) The entire course of 1000 marks has been divided in to 12 papers of 75/80/85/90/100/ marks of which 6 papers (Pape ...
... shall forward assessment in respect of every candidate to the Principal / Controller of Examination / Coordinator P. G. Courses (as the case may be) for tabulation of the results. 3.(a) The entire course of 1000 marks has been divided in to 12 papers of 75/80/85/90/100/ marks of which 6 papers (Pape ...
013-IC-2008-47-11228..
... Syntheses and Structure of Square-Pyramidal [FeIIMe2 + I((tame-N3)S2 )] (1), [FeIII(Et-N2S2Me2)]22- (2), [FeIII(EtN2S2Me2)(py)]1- (3), and [FeIII((tame-N2S)S2Me2)]2- (15). Imine-ligated [FeIII ((tame-N3)S2Me2)] (PF6) · PhCN (1) was synthesized under anaerobic conditions via an Fe2+templated, Schiff- ...
... Syntheses and Structure of Square-Pyramidal [FeIIMe2 + I((tame-N3)S2 )] (1), [FeIII(Et-N2S2Me2)]22- (2), [FeIII(EtN2S2Me2)(py)]1- (3), and [FeIII((tame-N2S)S2Me2)]2- (15). Imine-ligated [FeIII ((tame-N3)S2Me2)] (PF6) · PhCN (1) was synthesized under anaerobic conditions via an Fe2+templated, Schiff- ...
Vibrational spectroscopy of bare and solvated ionic complexes of
... radiation, one must be able to induce multiple photon absorption. Although the trap increases the interaction time, more powerful IR laser sources are generally required. Beauchamp and coworkers first demonstrated IR multiple photon dissociation (IRMPD) spectroscopy of mass-selected ions using a tun ...
... radiation, one must be able to induce multiple photon absorption. Although the trap increases the interaction time, more powerful IR laser sources are generally required. Beauchamp and coworkers first demonstrated IR multiple photon dissociation (IRMPD) spectroscopy of mass-selected ions using a tun ...
Computational aspects of hydroformylation
... more and more accurately by employing various quantum chemical techniques, especially density functional theory (DFT) methods. In general, the more sophisticated the computational model, the more expensive in terms of computational resources. Therefore, the computational chemist needs to nd the gol ...
... more and more accurately by employing various quantum chemical techniques, especially density functional theory (DFT) methods. In general, the more sophisticated the computational model, the more expensive in terms of computational resources. Therefore, the computational chemist needs to nd the gol ...
this PDF file - Journal of the Indian Institute of Science
... metal 0-donor bond and a metal to ligand n-acceptor bond. Back donation of the electron density ro the ?(*orbitals of the C-C multiple bond systems can thus take place. The M=C=C group is almost linear. The angle at C, is between 1.25 and 1.41 %1 which corresponds to a b n d order between two and th ...
... metal 0-donor bond and a metal to ligand n-acceptor bond. Back donation of the electron density ro the ?(*orbitals of the C-C multiple bond systems can thus take place. The M=C=C group is almost linear. The angle at C, is between 1.25 and 1.41 %1 which corresponds to a b n d order between two and th ...
Fulltext PDF - Indian Academy of Sciences
... chemical hardness of Aun Cd (n=1–12) clusters based on the framework of the density functional theory using relativistic all-electron methods. Low-lying energy structures include two-dimensional and three-dimensional geometries. Especially, all the lowest-energy structures of Aun Cd (n=1−12) cluster ...
... chemical hardness of Aun Cd (n=1–12) clusters based on the framework of the density functional theory using relativistic all-electron methods. Low-lying energy structures include two-dimensional and three-dimensional geometries. Especially, all the lowest-energy structures of Aun Cd (n=1−12) cluster ...
Revised_for_DT_final-1 - Pure
... intermediates. The molecular structure of [12]BF4 (as its pyridine solvate) showed that this species was a novel pyridylidene complex had been formed by activation of a C-H bond of the pyridine group in [11]BF4, incorporation of a second pyridine molecule and formal loss of HF, the fate of which cou ...
... intermediates. The molecular structure of [12]BF4 (as its pyridine solvate) showed that this species was a novel pyridylidene complex had been formed by activation of a C-H bond of the pyridine group in [11]BF4, incorporation of a second pyridine molecule and formal loss of HF, the fate of which cou ...
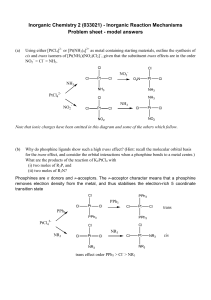
Inorganic Chemistry 2 (033021) - Inorganic Reaction
... This is an inner sphere electron transfer reaction, and will proceed via the species: [(NH3)5Co---NCS--Fe(H2O)5]4+ which will transfer an electron between the two metals, giving labile Co(II) and inert Fe(III). Water will substitute all the Co(II) ligands, giving [Co(H2O)6]2+ as ...
... This is an inner sphere electron transfer reaction, and will proceed via the species: [(NH3)5Co---NCS--Fe(H2O)5]4+ which will transfer an electron between the two metals, giving labile Co(II) and inert Fe(III). Water will substitute all the Co(II) ligands, giving [Co(H2O)6]2+ as ...
The Kubas Interaction in Transition Metal Based Hydrogen Storage
... The binding sites were modelled as fragments representing the active sites in the extended structures. Evidence has been found for the hydrogen binding through the Kubas interaction and the results were benchmarked against the available experimental data. The transition metals of the binding sites a ...
... The binding sites were modelled as fragments representing the active sites in the extended structures. Evidence has been found for the hydrogen binding through the Kubas interaction and the results were benchmarked against the available experimental data. The transition metals of the binding sites a ...
Introduction - University of Pretoria
... different methods: (i) transformation of a non-carbene ligand and (ii) addition of a carbene ligand precursor to a metal complex from pre-existing carbene complexes, of which five reactions are possible. They are (i) the transfer of a carbene ligand from one metal to another, (ii) modification of th ...
... different methods: (i) transformation of a non-carbene ligand and (ii) addition of a carbene ligand precursor to a metal complex from pre-existing carbene complexes, of which five reactions are possible. They are (i) the transfer of a carbene ligand from one metal to another, (ii) modification of th ...
- Maynooth University ePrints and eTheses Archive
... d6-DMSO solutions with Me4Si reference, unless indicated otherwise, with resolutions of 0.18 Hz and 0.01 ppm, respectively. Infrared spectra (cm 1) were recorded as KBr discs using a Perkin Elmer System2000 FT-IR spectrometer. UV–vis spectra were run on a Unicam UV 540 spectrometer. Melting point an ...
... d6-DMSO solutions with Me4Si reference, unless indicated otherwise, with resolutions of 0.18 Hz and 0.01 ppm, respectively. Infrared spectra (cm 1) were recorded as KBr discs using a Perkin Elmer System2000 FT-IR spectrometer. UV–vis spectra were run on a Unicam UV 540 spectrometer. Melting point an ...
Chapter
... • Structural Formula describe the kinds of elements found in the compound, the numbers of their atoms, order of atom attachment, and the kind of attachment they do not directly describe the 3-dimensional shape, but an experienced chemist can make a good guess at it use lines to represent covalen ...
... • Structural Formula describe the kinds of elements found in the compound, the numbers of their atoms, order of atom attachment, and the kind of attachment they do not directly describe the 3-dimensional shape, but an experienced chemist can make a good guess at it use lines to represent covalen ...
Chapter
... • Structural Formula describe the kinds of elements found in the compound, the numbers of their atoms, order of atom attachment, and the kind of attachment they do not directly describe the 3-dimensional shape, but an experienced chemist can make a good guess at it use lines to represent covalen ...
... • Structural Formula describe the kinds of elements found in the compound, the numbers of their atoms, order of atom attachment, and the kind of attachment they do not directly describe the 3-dimensional shape, but an experienced chemist can make a good guess at it use lines to represent covalen ...
Synthesis, Characterisation and DFT Analysis of {Ru(NO)2}8
... Fig. 2.31: Simplified model representation of static disorder. ..................................................................................................52 Fig. 2.32: Simplified model representation of a dynamic process ........................................................................ ...
... Fig. 2.31: Simplified model representation of static disorder. ..................................................................................................52 Fig. 2.32: Simplified model representation of a dynamic process ........................................................................ ...
Title Iridium, ruthenium, and palladium complexes containing a
... formation of palladium black during this process, which may point towards a limited stability of complex 4 or the anticipated transient (allyl)(carbene)palladium complex intermediate [69]. Of note, 1H NMR analysis of the soluble fractions does not indicate formation of any products by reductive elim ...
... formation of palladium black during this process, which may point towards a limited stability of complex 4 or the anticipated transient (allyl)(carbene)palladium complex intermediate [69]. Of note, 1H NMR analysis of the soluble fractions does not indicate formation of any products by reductive elim ...
INTRODUCTION
... activation have been described thoroughly in by Atkins [1], The principal method of monitoring reactions of coordination compounds is UV/visible spectrophotometery as many reactions of transition metal complexes are accompanied by changes in the electronic spectra. In coordination compounds metal at ...
... activation have been described thoroughly in by Atkins [1], The principal method of monitoring reactions of coordination compounds is UV/visible spectrophotometery as many reactions of transition metal complexes are accompanied by changes in the electronic spectra. In coordination compounds metal at ...
The synthesis and properties of solution processable red-emitting phosphorescent dendrimers Materials Chemistry
... be isolated in a 15% yield. The 1H NMR spectrum of G1ppy2BTPIr 3 was much more complicated than that observed for the simple symmetric iridium(III) complexes. However, the integral ratios of the surface alkyl groups to the aromatic region of the spectrum corresponded to the stoichiometry of the comp ...
... be isolated in a 15% yield. The 1H NMR spectrum of G1ppy2BTPIr 3 was much more complicated than that observed for the simple symmetric iridium(III) complexes. However, the integral ratios of the surface alkyl groups to the aromatic region of the spectrum corresponded to the stoichiometry of the comp ...
Chapter 5 - DORAS
... not proved as popular as its analogous palladium catalyzed counterpart. Similar as they are in mechanism, it is the palladium catalyzed reaction which has developed more rapidly over the last 35 years due to the large variety and availability of catalysts, and its more comprehensive application to a ...
... not proved as popular as its analogous palladium catalyzed counterpart. Similar as they are in mechanism, it is the palladium catalyzed reaction which has developed more rapidly over the last 35 years due to the large variety and availability of catalysts, and its more comprehensive application to a ...
Mono- and Trinuclear Nickel(II) Complexes with Sulfur-Containing Oxime Ligands:
... new dithioether-dioxime ligand 4,7-dithiadecane-2,9-dione dioxime, DtoxH2. Its homologue 3,9-dimethyl-4,8-dithiaundecane2,10-dione dioxime (DtdoH2) described previously1 forms a planar nickel(II) complex of the type Ni(Dtdo)ClO4). Nickel(II) complexes with some other polydentate thioether-oxime liga ...
... new dithioether-dioxime ligand 4,7-dithiadecane-2,9-dione dioxime, DtoxH2. Its homologue 3,9-dimethyl-4,8-dithiaundecane2,10-dione dioxime (DtdoH2) described previously1 forms a planar nickel(II) complex of the type Ni(Dtdo)ClO4). Nickel(II) complexes with some other polydentate thioether-oxime liga ...
Chemistry of the d
... e.g.: i) all Co(III) Complexes are diamagnetic; except [CoF6]3-, [CoF3(H2O)3] ii) Cr(III) in Oh: 3 unpaired e−; Cr(II) in Oh: predicted: 2e−, exp.: 4 unpaired e− 2) Ni(II) planar vs. octahedral is supposed to introduce change from covalent to ionic e.g.: [Ni(en)2]2+ 2NH3 Æ [Ni(NH3)2(en)2] 3) Two Ato ...
... e.g.: i) all Co(III) Complexes are diamagnetic; except [CoF6]3-, [CoF3(H2O)3] ii) Cr(III) in Oh: 3 unpaired e−; Cr(II) in Oh: predicted: 2e−, exp.: 4 unpaired e− 2) Ni(II) planar vs. octahedral is supposed to introduce change from covalent to ionic e.g.: [Ni(en)2]2+ 2NH3 Æ [Ni(NH3)2(en)2] 3) Two Ato ...
Thermally Driven Crossover from Indirect toward Direct Bandgap in
... coupling, <ℏω> is the average acoustic phonon energy involving in the electron−phonon interaction, and last the cosh term is related to the density of phonons at the specific temperature. We find that this model fits the temperature dependence of the bandgap well as shown in Figure 3f with E0g = 1.64 e ...
... coupling, <ℏω> is the average acoustic phonon energy involving in the electron−phonon interaction, and last the cosh term is related to the density of phonons at the specific temperature. We find that this model fits the temperature dependence of the bandgap well as shown in Figure 3f with E0g = 1.64 e ...
Activation of Carbon-Hydrogen Bonds via 1,2-Addition across M
... below) and is distinguished from the latter by more significant interaction between the metal and the hydrogen of the carbonhydrogen bond being activated. The distinction between OA, OHM, and SBM is reminiscent of many debates in chemical bonding; while technical distinctions exist and demarcation i ...
... below) and is distinguished from the latter by more significant interaction between the metal and the hydrogen of the carbonhydrogen bond being activated. The distinction between OA, OHM, and SBM is reminiscent of many debates in chemical bonding; while technical distinctions exist and demarcation i ...
Phosphorescent Organic Light-Emitting Devices: Working Principle and Iridium Based Emitter Materials
... states) in suitable device architectures. Thus, uncorrelated electrons and holes form triplet states with a threefold higher probability than singlet states [9]. Here it should be noted, that recent studies on spin statistics suggest variations in the singlet-to-triplet-ratios. These findings have a ...
... states) in suitable device architectures. Thus, uncorrelated electrons and holes form triplet states with a threefold higher probability than singlet states [9]. Here it should be noted, that recent studies on spin statistics suggest variations in the singlet-to-triplet-ratios. These findings have a ...
$doc.title
... Oithienothiophenes (OTT) are polycondensed heteroaromatic molecules consisting of three condensed thiophene rings. New interest in compounds of this type arose since dithienothiophenes possess three different types of n-conjugation which play an important role as a spacer and donor1 and these compou ...
... Oithienothiophenes (OTT) are polycondensed heteroaromatic molecules consisting of three condensed thiophene rings. New interest in compounds of this type arose since dithienothiophenes possess three different types of n-conjugation which play an important role as a spacer and donor1 and these compou ...
Jahn–Teller effect
-3D-balls.png?width=300)
The Jahn–Teller effect, sometimes also known as Jahn–Teller distortion, describes the geometrical distortion of molecules and ions that is associated with certain electron configurations. This electronic effect is named after Hermann Arthur Jahn and Edward Teller, who proved, using group theory, that orbital nonlinear spatially degenerate molecules cannot be stable. The Jahn–Teller theorem essentially states that any nonlinear molecule with a spatially degenerate electronic ground state will undergo a geometrical distortion that removes that degeneracy, because the distortion lowers the overall energy of the species. For a description of another type of geometrical distortion that occurs in crystals with substitutional impurities see article off-center ions.