Chemistry - CBSE Academic
... Group 13 Elements: General introduction, electronic configuration, occurrence, variation of properties, oxidation states, trends in chemical reactivity, anomalous properties of first element of the group, Boron - physical and chemical properties, some important compounds, Borax, Boric acid, Boron Hy ...
... Group 13 Elements: General introduction, electronic configuration, occurrence, variation of properties, oxidation states, trends in chemical reactivity, anomalous properties of first element of the group, Boron - physical and chemical properties, some important compounds, Borax, Boric acid, Boron Hy ...
Ultracold atoms in optical lattice
... are sufficiently cooled. Such optical lattice with trapped atoms resembles real crystals (fig. 1a), where neutral atoms play a role of electrons in real crystal. Yet while in real crystals typical lattice dimensions are tiny, with atoms spaced around nanometre apart, optical lattice has typical dime ...
... are sufficiently cooled. Such optical lattice with trapped atoms resembles real crystals (fig. 1a), where neutral atoms play a role of electrons in real crystal. Yet while in real crystals typical lattice dimensions are tiny, with atoms spaced around nanometre apart, optical lattice has typical dime ...
Role of Water as a Solvent
... To balance electrons we must put a 4 in front of the Ag, since each oxygen looses two electrons, and they come two at a time! That requires us to put a 4 in front of the silver complex, yielding 8 cyanide ions. 4 Ag(s) + 8 CN -(aq) + O2 (g) 4 Ag(CN)2-(aq) + OH -(aq) Add 4 OH- to balance charge. Sinc ...
... To balance electrons we must put a 4 in front of the Ag, since each oxygen looses two electrons, and they come two at a time! That requires us to put a 4 in front of the silver complex, yielding 8 cyanide ions. 4 Ag(s) + 8 CN -(aq) + O2 (g) 4 Ag(CN)2-(aq) + OH -(aq) Add 4 OH- to balance charge. Sinc ...
Introduction to Inorganic Chemistry
... order to achieve its ends. This means that a good chemist is one who not only has a mastery of chemical theory, but also a good knowledge of chemical facts. With such a knowledge, he can direct a trial and error approach to practical problems in the most promising directions. Inorganic Chemistry Org ...
... order to achieve its ends. This means that a good chemist is one who not only has a mastery of chemical theory, but also a good knowledge of chemical facts. With such a knowledge, he can direct a trial and error approach to practical problems in the most promising directions. Inorganic Chemistry Org ...
W. Pauli - Fisica Fundamental
... polarisation of the Zeeman components. It is, to be sure, not necessary according to this principle to assign in a definite stationary state to each electron an orbit uniquely determined in the sense of usual kinematics; however, it is necessary that the totality of the stationary states of an atom ...
... polarisation of the Zeeman components. It is, to be sure, not necessary according to this principle to assign in a definite stationary state to each electron an orbit uniquely determined in the sense of usual kinematics; however, it is necessary that the totality of the stationary states of an atom ...
Ex: -F, -Cl, -Br
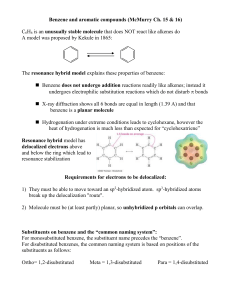
... X-ray diffraction shows all 6 bonds are equal in length (1.39 A) and that benzene is a planar molecule Hydrogenation under extreme conditions leads to cyclohexane, however the heat of hydrogenation is much less than expected for “cyclohexatriene” Resonance hybrid model has delocalized electrons ...
... X-ray diffraction shows all 6 bonds are equal in length (1.39 A) and that benzene is a planar molecule Hydrogenation under extreme conditions leads to cyclohexane, however the heat of hydrogenation is much less than expected for “cyclohexatriene” Resonance hybrid model has delocalized electrons ...
The Mole & Stoicheometry
... • Next, determine how many grams of each element are present in one mole of CO2: • 12.01 g (1 mol) of C 32.00 g (2 mole x 16.00 gram per mole) of O ...
... • Next, determine how many grams of each element are present in one mole of CO2: • 12.01 g (1 mol) of C 32.00 g (2 mole x 16.00 gram per mole) of O ...
2 Atoms and Molecules
... that a minimum of 115 different kinds of atoms exist. Eighty-eight of the elements are naturally occurring and therefore are found in Earth’s crust, oceans, or atmosphere. The others are synthetic elements produced in the laboratory. Each element can be characterized and identified by its unique set ...
... that a minimum of 115 different kinds of atoms exist. Eighty-eight of the elements are naturally occurring and therefore are found in Earth’s crust, oceans, or atmosphere. The others are synthetic elements produced in the laboratory. Each element can be characterized and identified by its unique set ...
1AMQ, Part II Quantum Mechanics
... The Rydberg-Ritz Combination Principle is an empirical relationship which states that if n1 and n2 are any 2 lines in one series, then |n1n2| is a line in another series. ...
... The Rydberg-Ritz Combination Principle is an empirical relationship which states that if n1 and n2 are any 2 lines in one series, then |n1n2| is a line in another series. ...
A Guide to Molecular Mechanics and Quantum Chemical Calculations
... promoting reaction are taken into account by way of the “A factor” multiplying the exponential. This is generally assumed to be constant for reactions involving a single set of reactants going to different products, or for reactions involving closely-related reactants. In general, the lower the acti ...
... promoting reaction are taken into account by way of the “A factor” multiplying the exponential. This is generally assumed to be constant for reactions involving a single set of reactants going to different products, or for reactions involving closely-related reactants. In general, the lower the acti ...
On inelastic hydrogen atom collisions in stellar atmospheres
... The Drawin formula is the result of a number of modifications and extensions of the classical formula for ionization of atoms by electron impact due to Thomson (1912). In Thomson’s theory, the bound electron in the target atom is considered as a stationary free classical electron. The Coulomb intera ...
... The Drawin formula is the result of a number of modifications and extensions of the classical formula for ionization of atoms by electron impact due to Thomson (1912). In Thomson’s theory, the bound electron in the target atom is considered as a stationary free classical electron. The Coulomb intera ...
Scanning Tunneling Microscopy and Quartz Crystal Microbalance
... obtained many images like this one in air.19 After exposure to an oxygen-saturated (1 part 02/32 parts H20 by volume at 25 "C) 10-5 M solution of Na2Sfor 30 min, atomic resolution images of the surface showed a new pattern (Figure 2A). The surface was composed of a series of squares, each of which c ...
... obtained many images like this one in air.19 After exposure to an oxygen-saturated (1 part 02/32 parts H20 by volume at 25 "C) 10-5 M solution of Na2Sfor 30 min, atomic resolution images of the surface showed a new pattern (Figure 2A). The surface was composed of a series of squares, each of which c ...
Lewis Acids and Bases - Screenshot for timg.co.il
... There are reactions in nonaqueous solvents, in the gaseous state, and even in the solid state that can be considered acid–base reactions in which Brønsted–Lowry theory is not adequate to explain. ...
... There are reactions in nonaqueous solvents, in the gaseous state, and even in the solid state that can be considered acid–base reactions in which Brønsted–Lowry theory is not adequate to explain. ...
Chapter 5 - CARSON`S CHEMISTRY CLASS
... the quest to understand the atom and its structure had really just begun. That quest continues in this chapter, as scientists pursued an understanding of how electrons were arranged within atoms. Perform the DISCOVERY LAB on this page to better understand the difficulties scientists faced in researc ...
... the quest to understand the atom and its structure had really just begun. That quest continues in this chapter, as scientists pursued an understanding of how electrons were arranged within atoms. Perform the DISCOVERY LAB on this page to better understand the difficulties scientists faced in researc ...
The Formation of Solvated Electrons in the Photochemistry of the
... radiation chemicalI2 and photochemical1@data is approximately il.9. One therefore gets k~~ + e a e - j k ~+z ~ e4Q- = 0.4 to 0.6, in fair agreement with the value obtained in our treatment. These results yield a further independent support to the identification of the species formed from the excited ...
... radiation chemicalI2 and photochemical1@data is approximately il.9. One therefore gets k~~ + e a e - j k ~+z ~ e4Q- = 0.4 to 0.6, in fair agreement with the value obtained in our treatment. These results yield a further independent support to the identification of the species formed from the excited ...
PowerPoint
... To balance electrons we must put a 4 in front of the Ag, since each oxygen looses two electrons, and they come two at a time! That requires us to put a 4 in front of the silver complex, yielding 8 cyanide ions. 4 Ag(s) + 8 CN -(aq) + O2 (g) 4 Ag(CN)2-(aq) + OH -(aq) Add 4 OH- to balance charge. Sinc ...
... To balance electrons we must put a 4 in front of the Ag, since each oxygen looses two electrons, and they come two at a time! That requires us to put a 4 in front of the silver complex, yielding 8 cyanide ions. 4 Ag(s) + 8 CN -(aq) + O2 (g) 4 Ag(CN)2-(aq) + OH -(aq) Add 4 OH- to balance charge. Sinc ...
Note Sheets and Sample Problems
... Chemical Foundations Chemistry: An Overview • Matter – takes up space, has mass, exhibits inertia - composed of atoms only 100 or so different types - Water made up of one oxygen and two hydrogen atoms - Pass an electric current through it to separate the two types of atoms and they rearrange to bec ...
... Chemical Foundations Chemistry: An Overview • Matter – takes up space, has mass, exhibits inertia - composed of atoms only 100 or so different types - Water made up of one oxygen and two hydrogen atoms - Pass an electric current through it to separate the two types of atoms and they rearrange to bec ...
Angewandte - School of Physics
... in two main ways. First, at finite temperature, the model catalyst (and in particular the cluster component) will form an equilibrium of coexisting structural configurations, with various isomers exhibiting different chemical reactivities. This situation is illustrated here by the properties of two ...
... in two main ways. First, at finite temperature, the model catalyst (and in particular the cluster component) will form an equilibrium of coexisting structural configurations, with various isomers exhibiting different chemical reactivities. This situation is illustrated here by the properties of two ...
A Primer to Electronic Structure Computation
... of electrons are certainly correlated because they experience a mutual coulomb repulsion and therefore the presence of an electron decreases the probability of nding another electron nearby. Secondly, real electrons are indistinguishable and obey the antisymmetry principle (1, p.47). Because of the ...
... of electrons are certainly correlated because they experience a mutual coulomb repulsion and therefore the presence of an electron decreases the probability of nding another electron nearby. Secondly, real electrons are indistinguishable and obey the antisymmetry principle (1, p.47). Because of the ...
Atomic Structure
... An unwanted side effect of this medicine is that it can cause the patient to have ‘wind’ (too much gas in the intestine). The equation below represents the reaction between calcium carbonate and hydrochloric acid (the acid present in the stomach). CaCO3 (s) + 2HCl (aq) →CaCl2 (aq) + H2O (l) + CO2 (g ...
... An unwanted side effect of this medicine is that it can cause the patient to have ‘wind’ (too much gas in the intestine). The equation below represents the reaction between calcium carbonate and hydrochloric acid (the acid present in the stomach). CaCO3 (s) + 2HCl (aq) →CaCl2 (aq) + H2O (l) + CO2 (g ...
Chemistry Unit Outcomes
... Describe what is known as a physical change. List 7 types of physical changes. Outline what it is possible to do with most physical changes. Describe what happens to the original substance or substances during a chemical change. Outline one thing that chemical changes always involve. Explain one thi ...
... Describe what is known as a physical change. List 7 types of physical changes. Outline what it is possible to do with most physical changes. Describe what happens to the original substance or substances during a chemical change. Outline one thing that chemical changes always involve. Explain one thi ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.