The p orbital paradox
... electron is bound to the atom, we will not be able to say much more about its position than that it is in the atom. Certainly all models of the atom which describe the electron as a particle following a definite trajectory or orbit must be discarded. We can obtain an energy and one or more wave func ...
... electron is bound to the atom, we will not be able to say much more about its position than that it is in the atom. Certainly all models of the atom which describe the electron as a particle following a definite trajectory or orbit must be discarded. We can obtain an energy and one or more wave func ...
Molecular diffusion at surfaces
... close to C move 0. 13 A. Twelve more surrounding Fe atoms relax < 0.07 A. The change in energy for C moving from the O site through the T site is shown in the fig. The 0 and T sites are comparable in energy to each other but both of them are predicted to be less stable than the O-M site. It was foun ...
... close to C move 0. 13 A. Twelve more surrounding Fe atoms relax < 0.07 A. The change in energy for C moving from the O site through the T site is shown in the fig. The 0 and T sites are comparable in energy to each other but both of them are predicted to be less stable than the O-M site. It was foun ...
File
... Using the tripeptide in (ii), state two types of bonding that can be formed and the groups in the tripeptide that are involved in this bonding. bond .................................................... groups ........................................................ bond ............................. ...
... Using the tripeptide in (ii), state two types of bonding that can be formed and the groups in the tripeptide that are involved in this bonding. bond .................................................... groups ........................................................ bond ............................. ...
Photoassociation of ultracold LiRb molecules: Observation of high efficiency
... Heteronuclear polar molecules have recently attracted enormous attention [1–17] owing to their ground state having a large electric dipole moment [16]. The long range anisotropic dipole-dipole interaction in such systems is the basis for a variety of applications including quantum computing [13], pr ...
... Heteronuclear polar molecules have recently attracted enormous attention [1–17] owing to their ground state having a large electric dipole moment [16]. The long range anisotropic dipole-dipole interaction in such systems is the basis for a variety of applications including quantum computing [13], pr ...
Answers/solutions
... The reminder can be proved exactly the same way. 11.14 (a) Calculate the angle in Fig. 11.3 between the z axis and S for the spin function α(1) α(2).(b) Calculate the angle between S1 and S2 for each of the functions (11.57) to (11.60). 〔Hint :One approach is to use the law of cosines. A second appr ...
... The reminder can be proved exactly the same way. 11.14 (a) Calculate the angle in Fig. 11.3 between the z axis and S for the spin function α(1) α(2).(b) Calculate the angle between S1 and S2 for each of the functions (11.57) to (11.60). 〔Hint :One approach is to use the law of cosines. A second appr ...
Bond Dissociation Energies of Organic Molecules
... integrals, ∫[Cp(R) - Cp(R-) + Cp(H) - Cp(H+)] dT, which may be evaluated as previously described, but their value is generally less than 0.3 kcal mol-1. By rearranging eq 16, one can estimate typical values for ∆acidH298(RH); since DH298(RH) for many organics is approximately 100 kcal mol-1, the IE0 ...
... integrals, ∫[Cp(R) - Cp(R-) + Cp(H) - Cp(H+)] dT, which may be evaluated as previously described, but their value is generally less than 0.3 kcal mol-1. By rearranging eq 16, one can estimate typical values for ∆acidH298(RH); since DH298(RH) for many organics is approximately 100 kcal mol-1, the IE0 ...
Formation of Phosphorus-Nitrogen Bonds by Reduction of a
... dinitrogen moiety coordinated to the dititanium core. In fact, the dinuclear metal complex has only the [NPN] bound but with one important change: the phosphine donor has been transformed into a phosphinimide unit.8 Formally, the [N(PN)N] ligand set (where [N(PN)N] ) (PhNSiMe2CH2)2P(dN)Ph) is a tria ...
... dinitrogen moiety coordinated to the dititanium core. In fact, the dinuclear metal complex has only the [NPN] bound but with one important change: the phosphine donor has been transformed into a phosphinimide unit.8 Formally, the [N(PN)N] ligand set (where [N(PN)N] ) (PhNSiMe2CH2)2P(dN)Ph) is a tria ...
Chapter 7: Chemical Formulas and Chemical Compounds
... Oxidation numbers do not have an exact physical meaning. However they do come in handy when naming compounds, writing formulas, and balancing chemical equations. As a general rule in assigning oxidation numbers, shared electrons are assumed to belong to the more-electronegative atom in each bond. He ...
... Oxidation numbers do not have an exact physical meaning. However they do come in handy when naming compounds, writing formulas, and balancing chemical equations. As a general rule in assigning oxidation numbers, shared electrons are assumed to belong to the more-electronegative atom in each bond. He ...
Cooling and Trapping Neutral Atoms
... However, this behavior requires a commensurable ratio between electrons and sites. For neutral bosonic particles, the equivalent phenomenon is the transition from a superfluid to an insulator for commensurable densities. In inhomogeneous systems, as in atom traps, the condition of commensurability n ...
... However, this behavior requires a commensurable ratio between electrons and sites. For neutral bosonic particles, the equivalent phenomenon is the transition from a superfluid to an insulator for commensurable densities. In inhomogeneous systems, as in atom traps, the condition of commensurability n ...
... as those arising from the Breit interaction and from quantum electrodynamics (QED), are sometimes incorporated using hydrogenic expressions with screened charges [7]. In the screened hydrogenic model (SHM), it is assumed that the wavefunction of an electron in subshell n, l, j can be described by me ...
1. Structure of Matter
... • As the electron moves to orbits of increasing radius, it does so in opposition to the restoring force due to the positive nucleus, and its potential energy is thereby raised. This is entirely analogous to the increase in potential energy that occurs when any mechanical system moves against a rest ...
... • As the electron moves to orbits of increasing radius, it does so in opposition to the restoring force due to the positive nucleus, and its potential energy is thereby raised. This is entirely analogous to the increase in potential energy that occurs when any mechanical system moves against a rest ...
chemistry
... Statement B: Atoms of one element cannot be changed into atoms of another element. Statement C: All atoms of one element have the same mass. 66 Explain, in terms of particles, why statement A is no longer accepted. [1] 67 The decay of N-16 is represented by the balanced equation below. ...
... Statement B: Atoms of one element cannot be changed into atoms of another element. Statement C: All atoms of one element have the same mass. 66 Explain, in terms of particles, why statement A is no longer accepted. [1] 67 The decay of N-16 is represented by the balanced equation below. ...
Chapter 20
... Because magnesium is a better reducing agent than iron and is more easily oxidized the magnesium immediately transfers electrons to the iron, preventing their oxidation to iron ...
... Because magnesium is a better reducing agent than iron and is more easily oxidized the magnesium immediately transfers electrons to the iron, preventing their oxidation to iron ...
Chapter 4 Student Notes
... Acids are substances that are able to ionize in aqueous solution to form H 1+ (Arrhenius definition). H1+ lacks an electron, so it is a bare proton. Acids are often called proton donors in acid-base reactions. An example is HC2H3O2 (acetic acid). Since H1+ is a naked proton, we refer to acids as pro ...
... Acids are substances that are able to ionize in aqueous solution to form H 1+ (Arrhenius definition). H1+ lacks an electron, so it is a bare proton. Acids are often called proton donors in acid-base reactions. An example is HC2H3O2 (acetic acid). Since H1+ is a naked proton, we refer to acids as pro ...
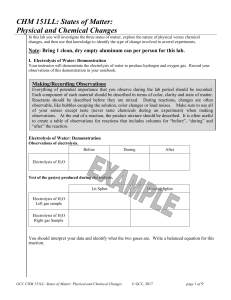
CHM 151LL: States of Matter: Physical and Chemical Changes
... Substances can exist in three physical states: solid, liquid, and gas. Some of the differences between these states of matter are 1) the atoms’ or molecules’ freedom of movement and 2) the amount of space between the atoms or molecules. The physical state of a substance at a specific temperature dep ...
... Substances can exist in three physical states: solid, liquid, and gas. Some of the differences between these states of matter are 1) the atoms’ or molecules’ freedom of movement and 2) the amount of space between the atoms or molecules. The physical state of a substance at a specific temperature dep ...
Chapter 3 MAGNETISM OF THE ELECTRON
... The angular frequency ! c = 2 fc :Any component of the electron velocity parallel to the magnetic …eld is unin‡uenced by the Lorentz force, so the trajectory or the electron is a helix along the …eld direction. Electrons which follow cyclotron orbits radiate energy of frequency fc . The cyclotron fr ...
... The angular frequency ! c = 2 fc :Any component of the electron velocity parallel to the magnetic …eld is unin‡uenced by the Lorentz force, so the trajectory or the electron is a helix along the …eld direction. Electrons which follow cyclotron orbits radiate energy of frequency fc . The cyclotron fr ...
1 Indentifying Unknown #M20 via Infrared Spectroscopy, Mass
... de calculated and will shed light on the number of double and/or triple bonds and the possibility of a ringed structure. Once the molecular formula is determined based on the information presented in both the IR and mass spectra, the 13C NMR contributes by determining symmetry and carbon bonding env ...
... de calculated and will shed light on the number of double and/or triple bonds and the possibility of a ringed structure. Once the molecular formula is determined based on the information presented in both the IR and mass spectra, the 13C NMR contributes by determining symmetry and carbon bonding env ...
Audit Schedule
... 1. To be familiar with the basic assumptions of Dalton's Atomic Theory. 2. To state the Law of Conservation of Matter. 3. To know the relationships among atomic number, mass number, number of protons, number of neutrons, and number of electrons. [C1] 4. To calculate the average atomic mass of an ele ...
... 1. To be familiar with the basic assumptions of Dalton's Atomic Theory. 2. To state the Law of Conservation of Matter. 3. To know the relationships among atomic number, mass number, number of protons, number of neutrons, and number of electrons. [C1] 4. To calculate the average atomic mass of an ele ...
AP Chemistry Review Preparing for the AP
... Give examples and solve calculation problems related to each of the three theories. Sketch a cathode ray tube as demonstrated in class and state how J.J. Thomson’s experiments led to the idea that atoms have positive and negative parts, the negative parts are all the same, and the negative parts ...
... Give examples and solve calculation problems related to each of the three theories. Sketch a cathode ray tube as demonstrated in class and state how J.J. Thomson’s experiments led to the idea that atoms have positive and negative parts, the negative parts are all the same, and the negative parts ...
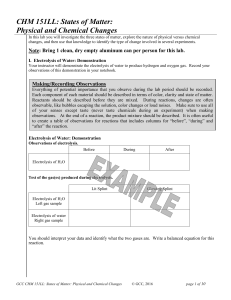
CHM 151LL: States of Matter: Physical and Chemical Changes
... and that have not reacted with each other to form a pure substance. Mixtures can be separated into their individual compounds by physical methods, that is, by methods that don’t require the breaking or making of chemical bonds. Common separation methods include filtration, decanting, distillation, e ...
... and that have not reacted with each other to form a pure substance. Mixtures can be separated into their individual compounds by physical methods, that is, by methods that don’t require the breaking or making of chemical bonds. Common separation methods include filtration, decanting, distillation, e ...
- Kendriya Vidyalaya No. 2 Raipur
... irrespective of the method of preparation or the source from where it is taken. 3. Write the empirical formula of the following: (a) N2O4 (b) C6H12O6 (c) H2O (d) H2O2 Ans. (a)NO2 (b) CH2O (c) H2O ...
... irrespective of the method of preparation or the source from where it is taken. 3. Write the empirical formula of the following: (a) N2O4 (b) C6H12O6 (c) H2O (d) H2O2 Ans. (a)NO2 (b) CH2O (c) H2O ...
class XI CHEMISTRY - Kendriya Vidyalaya No.1 Harni Road
... irrespective of the method of preparation or the source from where it is taken. 3. Write the empirical formula of the following: (a) N2O4 (b) C6H12O6 (c) H2O (d) H2O2 Ans. (a)NO2 (b) CH2O (c) H2O ...
... irrespective of the method of preparation or the source from where it is taken. 3. Write the empirical formula of the following: (a) N2O4 (b) C6H12O6 (c) H2O (d) H2O2 Ans. (a)NO2 (b) CH2O (c) H2O ...
class XI CHEMISTRY - Kendriya Vidyalaya No.1 Ichhanath Surat
... irrespective of the method of preparation or the source from where it is taken. 3. Write the empirical formula of the following: (a) N2O4 (b) C6H12O6 (c) H2O (d) H2O2 Ans. (a)NO2 (b) CH2O (c) H2O ...
... irrespective of the method of preparation or the source from where it is taken. 3. Write the empirical formula of the following: (a) N2O4 (b) C6H12O6 (c) H2O (d) H2O2 Ans. (a)NO2 (b) CH2O (c) H2O ...
Teaching with SCIGRESS - Photochemical Dynamics Group
... provides much of the information upon which these predictions are made. From lectures you have learned to use several models to describe molecules. For example Lewis dot structures delineate connectivity and bonding relationships between atoms of a molecule or ion and can predict bond energies and b ...
... provides much of the information upon which these predictions are made. From lectures you have learned to use several models to describe molecules. For example Lewis dot structures delineate connectivity and bonding relationships between atoms of a molecule or ion and can predict bond energies and b ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.