04 Reactions in Aqueous Solution
... that does not change from the left side of the equation to the right. • The ions crossed out are called spectator ions, K+ and NO3−, in this example. • The remaining ions are the reactants that form the product—an insoluble salt in a precipitation reaction, as in this example. ...
... that does not change from the left side of the equation to the right. • The ions crossed out are called spectator ions, K+ and NO3−, in this example. • The remaining ions are the reactants that form the product—an insoluble salt in a precipitation reaction, as in this example. ...
chapter 7 multielectron atoms outline
... Consider the 3 electrons in a lithium atom, which has the electron configuration: 1s22s1. (a) Write the Hamiltonian for the electrons in a Lithium atom in (i) MKS (SI) units and (ii) in atomic units. (b) Use the “Independent Particle Model” (i.e. ignore interelectronic repulsions) to calculate the e ...
... Consider the 3 electrons in a lithium atom, which has the electron configuration: 1s22s1. (a) Write the Hamiltonian for the electrons in a Lithium atom in (i) MKS (SI) units and (ii) in atomic units. (b) Use the “Independent Particle Model” (i.e. ignore interelectronic repulsions) to calculate the e ...
2 - Gordon State College
... CHECK YOUR NEIGHBOR Carefully examine the following reaction sequence for the catalytic formation of ozone, O3, from molecular oxygen, O2. Which chemical compound is behaving as the catalyst? O2 + 2 NO 2 NO2 2 NO2 2 NO + 2 O 2 O + 2 O 2 2 O3 A. Nitrogen dioxide, NO2 B. Nitrogen monoxide, NO C. ...
... CHECK YOUR NEIGHBOR Carefully examine the following reaction sequence for the catalytic formation of ozone, O3, from molecular oxygen, O2. Which chemical compound is behaving as the catalyst? O2 + 2 NO 2 NO2 2 NO2 2 NO + 2 O 2 O + 2 O 2 2 O3 A. Nitrogen dioxide, NO2 B. Nitrogen monoxide, NO C. ...
Quaternary Neptunium Compounds: Syntheses and
... Syntheses. KCuNpS3 was prepared from a reaction mixture of 0.086 mmol Np, 0.086 mmol Cu, 0.043 mmol K2S, and 0.258 mmol S. KAgNpS3 was prepared from a reaction mixture of 0.084 mmol Np, 0.084 mmol Ag, 0.042 mmol K2S, and 0.253 mmol S. RbCuNpS3 was prepared from a reaction mixture of 0.0447 mmol Np, ...
... Syntheses. KCuNpS3 was prepared from a reaction mixture of 0.086 mmol Np, 0.086 mmol Cu, 0.043 mmol K2S, and 0.258 mmol S. KAgNpS3 was prepared from a reaction mixture of 0.084 mmol Np, 0.084 mmol Ag, 0.042 mmol K2S, and 0.253 mmol S. RbCuNpS3 was prepared from a reaction mixture of 0.0447 mmol Np, ...
INTERPLAY BETWEEN STRUCTURAL AND ELECTRONIC PROPERTIES OF CARBON NANOTUBES
... electrons have been considered as active. Our description of Pb nanowires within the jellium model follows Ref. 20 . We use rs = 2.30 a.u. for the WignerSeitz radius and the valence Z = 4 for Pb in the ground-state configuration [Xe]4f14 5d10 6s2 6p2 . The jellium background charge density of Bi, whi ...
... electrons have been considered as active. Our description of Pb nanowires within the jellium model follows Ref. 20 . We use rs = 2.30 a.u. for the WignerSeitz radius and the valence Z = 4 for Pb in the ground-state configuration [Xe]4f14 5d10 6s2 6p2 . The jellium background charge density of Bi, whi ...
CHAPTER
... same temperature and pressure contain equal numbers of particles, he may have been thinking of particles rearranging themselves. Individual gas particles are so small that their rearranging cannot be observed, but the volumes of gases can be measured directly. Avogadro's principle is one of the earl ...
... same temperature and pressure contain equal numbers of particles, he may have been thinking of particles rearranging themselves. Individual gas particles are so small that their rearranging cannot be observed, but the volumes of gases can be measured directly. Avogadro's principle is one of the earl ...
FEATURE ARTICLE
... interest in these techniques,53,54 and also their applicability in an ab initio context is being investigated.55,56 Although the high degree of reliability of the DFT calculations based on the present functionals, usually referred to as generalized gradient approximations (GGA), and their computatio ...
... interest in these techniques,53,54 and also their applicability in an ab initio context is being investigated.55,56 Although the high degree of reliability of the DFT calculations based on the present functionals, usually referred to as generalized gradient approximations (GGA), and their computatio ...
3.2 MB - KFUPM Resources v3
... 100% - % mass C - % mass H = % mass O = 53.2% O (c) Assuming having 100 g of CxHyOz, there will be 40.1g C , 6.74g H, and 53.2g O. # mol of C = 40.1g C × [1 mol C / 12.01 g C] = 3.34 mol C In the same way: we get 6.67 mol H and 3.33 mol O. (d) Finding the smallest whole number ratio by dividing by 3 ...
... 100% - % mass C - % mass H = % mass O = 53.2% O (c) Assuming having 100 g of CxHyOz, there will be 40.1g C , 6.74g H, and 53.2g O. # mol of C = 40.1g C × [1 mol C / 12.01 g C] = 3.34 mol C In the same way: we get 6.67 mol H and 3.33 mol O. (d) Finding the smallest whole number ratio by dividing by 3 ...
Theoretical Study of Tautomeric Forms of Uracil and Their
... each strand - base pairing and by stacking interactions between the base pairs. Hydrophilic and hydrophobic interactions are also important for the structure and stability of DNA. Charged and highly polar phosphate-deoxyribose chain is exposed to the solvent while less polar nitrogenous bases are he ...
... each strand - base pairing and by stacking interactions between the base pairs. Hydrophilic and hydrophobic interactions are also important for the structure and stability of DNA. Charged and highly polar phosphate-deoxyribose chain is exposed to the solvent while less polar nitrogenous bases are he ...
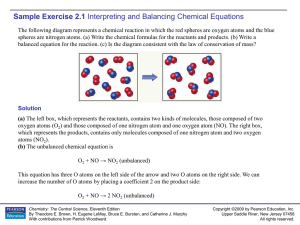
Sample Exercise 2.1
... Write balanced equations for the following reactions: (a) The combination reaction that occurs when lithium metal and fluorine gas react. (b) The decomposition reaction that occurs when solid barium carbonate is heated. (Two products form: a solid and a gas.) Solution (a) The symbol for lithium is L ...
... Write balanced equations for the following reactions: (a) The combination reaction that occurs when lithium metal and fluorine gas react. (b) The decomposition reaction that occurs when solid barium carbonate is heated. (Two products form: a solid and a gas.) Solution (a) The symbol for lithium is L ...
Deuterium fractionation of methylamine through atomic grain
... Deuterium fractionation of methylamine through atomic grain-surface reactions at low temperatures: implication for the possible D/H ratio in molecular clouds Y. Oba, T. Chigai, N. Watanabe, and A. Kouchi Institute of Low Temperature Science, Hokkaido University, Japan Interstellar methylamine (CH3NH ...
... Deuterium fractionation of methylamine through atomic grain-surface reactions at low temperatures: implication for the possible D/H ratio in molecular clouds Y. Oba, T. Chigai, N. Watanabe, and A. Kouchi Institute of Low Temperature Science, Hokkaido University, Japan Interstellar methylamine (CH3NH ...
Mole
... Mole Ratio In a balanced equation, the ration between the numbers of moles of any two substances. ...
... Mole Ratio In a balanced equation, the ration between the numbers of moles of any two substances. ...
File
... Determine the empirical formula of Ascorbic acid (vitamin C: cures/prevents scurvy). It is composed of 40.92% C, 4.58% H, and 54.50% O by mass. To determine the empirical formula, we will first assume a 100 g sample of Ascorbic Acid ...
... Determine the empirical formula of Ascorbic acid (vitamin C: cures/prevents scurvy). It is composed of 40.92% C, 4.58% H, and 54.50% O by mass. To determine the empirical formula, we will first assume a 100 g sample of Ascorbic Acid ...
Quantum transport through STM-lifted single PTCDA molecules
... simulated from first principles. (4) Various density functionals, including GGA and LDA, have been evaluated for the PTCDA/Ag(111) adsorption system. In particular, it must be noted here that according to the bonding model of Fig. 2 the carboxylic oxygen atoms are involved in local Ag–O bonds. ...
... simulated from first principles. (4) Various density functionals, including GGA and LDA, have been evaluated for the PTCDA/Ag(111) adsorption system. In particular, it must be noted here that according to the bonding model of Fig. 2 the carboxylic oxygen atoms are involved in local Ag–O bonds. ...
Ground-state stability and criticality of two
... accurate enough to discard a small 2e− − 0e− line. We discard the existence of an infinite 2e− − 0e− line. We have shown in previous works that FSS combined with different basis functions (Hylleraas, Gaussian, Slater) is a powerful method to obtain quantum critical parameters for few-body systems [2 ...
... accurate enough to discard a small 2e− − 0e− line. We discard the existence of an infinite 2e− − 0e− line. We have shown in previous works that FSS combined with different basis functions (Hylleraas, Gaussian, Slater) is a powerful method to obtain quantum critical parameters for few-body systems [2 ...
A Spectroscopic Determination of Scattering Lengths for Sodium
... ,fa between the initial ground electronic state labeled by ,fa and the excited electronic state labeled by b . Dipole selection rules require that p' = 2 p , and DF = F' 2 F = {0, 6 1}, except that DF Þ 0 for F = 0. F'p'b The VFp ,fa are calculated from the known atomic transition dipole moment betw ...
... ,fa between the initial ground electronic state labeled by ,fa and the excited electronic state labeled by b . Dipole selection rules require that p' = 2 p , and DF = F' 2 F = {0, 6 1}, except that DF Þ 0 for F = 0. F'p'b The VFp ,fa are calculated from the known atomic transition dipole moment betw ...
ATOMISTIC INTERACTIONS IN STM ATOM MANIPULATION A
... different values of manipulation voltage and current. The threshold distance to pull an atom out from the cluster was determined. The tip-cluster distance proved to be the governing factor for the atom extraction mechanism. Lateral manipulation of a metal atom, silver, and a halogen atom, bromine, w ...
... different values of manipulation voltage and current. The threshold distance to pull an atom out from the cluster was determined. The tip-cluster distance proved to be the governing factor for the atom extraction mechanism. Lateral manipulation of a metal atom, silver, and a halogen atom, bromine, w ...
Phenomenological description of the transition state, and the bond
... information about the stationary points of the energy surface. Considering the Born–Oppenheimer approximation, minima on the N-dimensional potential energy surface for the nuclei can be identified with the classical picture of equilibrium structures of molecules and saddle points can be related to t ...
... information about the stationary points of the energy surface. Considering the Born–Oppenheimer approximation, minima on the N-dimensional potential energy surface for the nuclei can be identified with the classical picture of equilibrium structures of molecules and saddle points can be related to t ...
Fluctuation-Induced Forces Between Atoms and
... with temperature is actually a free energy of interaction and is also known as the Keesom potential between polar molecules: there, the dipoles are rotating freely under the influence of thermal fluctuations [26]. In this case (Rydberg atoms provide another example), the particle resonances overlap ...
... with temperature is actually a free energy of interaction and is also known as the Keesom potential between polar molecules: there, the dipoles are rotating freely under the influence of thermal fluctuations [26]. In this case (Rydberg atoms provide another example), the particle resonances overlap ...
An experimental and theoretical guide to strongly interacting
... as long as there is no deterministic loading scheme available [33, 34]. An alternative way to achieve a large quantum register with individually resolved sites is a Mottinsulator state with single site resolution [35, 36] or a switchable local electric field, e.g. produced by an electron beam [37] w ...
... as long as there is no deterministic loading scheme available [33, 34]. An alternative way to achieve a large quantum register with individually resolved sites is a Mottinsulator state with single site resolution [35, 36] or a switchable local electric field, e.g. produced by an electron beam [37] w ...
intestazione repositorydell`ateneo Layer charge and heavy metals
... in the octahedral sheet, whereas it behaves like a hard base when the charge is located in the tetrahedral sites. Hard bases bond preferentially to hard acids and soft bases to soft acids, thus also confirming the fundamental role of layer charge location on exchange processes, since different heavy ...
... in the octahedral sheet, whereas it behaves like a hard base when the charge is located in the tetrahedral sites. Hard bases bond preferentially to hard acids and soft bases to soft acids, thus also confirming the fundamental role of layer charge location on exchange processes, since different heavy ...
Proton Chemical Shift Tensors and Hydrogen Bond Geometry: A 1H
... Proton CS Tensors of the Stationary Water Molecules. It is clear from above discussion that the rapid flipping motion of the water molecule results in aVeraged 1H CS tensors to be measured through experiments. However, it is more desirable to obtain 1H CS tensors for the stationary water molecules, ...
... Proton CS Tensors of the Stationary Water Molecules. It is clear from above discussion that the rapid flipping motion of the water molecule results in aVeraged 1H CS tensors to be measured through experiments. However, it is more desirable to obtain 1H CS tensors for the stationary water molecules, ...
Η - Knockhardy
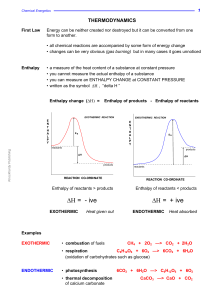
... Imagine that, during a reaction, all the bonds of reacting species are broken and the individual atoms join up again but in the form of products. The overall energy change will depend on the difference between the energy required to break the bonds and that released as bonds are made. energy release ...
... Imagine that, during a reaction, all the bonds of reacting species are broken and the individual atoms join up again but in the form of products. The overall energy change will depend on the difference between the energy required to break the bonds and that released as bonds are made. energy release ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.