LaBrake, Fundamentals Diagnostic Questions
... 19. All of the following statements are true regarding the nuclear model of the atom, except: a) The positive charge is densely found in the center of the atom, while the negatively charged electrons exist in a diffuse cloud outside the nucleus. b) Most of the space of an atom is empty space. c) The ...
... 19. All of the following statements are true regarding the nuclear model of the atom, except: a) The positive charge is densely found in the center of the atom, while the negatively charged electrons exist in a diffuse cloud outside the nucleus. b) Most of the space of an atom is empty space. c) The ...
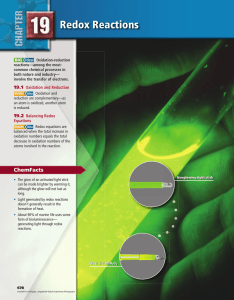
Chapter 19
... involve changes in molecular substances or polyatomic ions in which atoms are covalently bonded to other atoms. For example, the following equation represents the redox reaction used to manufacture ammonia (NH 3). N 2(g) + 3H 2(g) → 2NH 3(g) This process involves neither ions nor any obvious transfe ...
... involve changes in molecular substances or polyatomic ions in which atoms are covalently bonded to other atoms. For example, the following equation represents the redox reaction used to manufacture ammonia (NH 3). N 2(g) + 3H 2(g) → 2NH 3(g) This process involves neither ions nor any obvious transfe ...
lecture ch1-3 chem161pikul
... • Numbers in front of formulas • Indicate how many of each type of molecule reacted or formed – Equation reads methane and oxygen react to yield carbon dioxide and water Jesperson, Brady, Hyslop. Chemistry: The Molecular Nature of Matter, 6E ...
... • Numbers in front of formulas • Indicate how many of each type of molecule reacted or formed – Equation reads methane and oxygen react to yield carbon dioxide and water Jesperson, Brady, Hyslop. Chemistry: The Molecular Nature of Matter, 6E ...
Learning Outcomes
... giant molecular substances, e.g. poly(ethene); sand (silicon dioxide); diamond; graphite in order to deduce their properties (c) compare the bonding and structures of diamond and graphite in order to deduce their properties such as electrical conductivity, lubricating or cutting action (candidates w ...
... giant molecular substances, e.g. poly(ethene); sand (silicon dioxide); diamond; graphite in order to deduce their properties (c) compare the bonding and structures of diamond and graphite in order to deduce their properties such as electrical conductivity, lubricating or cutting action (candidates w ...
Word - icho39.chem.msu.ru
... Of course, these results obtained by extrapolation are approximate. Moreover, bulk properties such as melting and boiling points can be measured only for significant amounts of an element, whereas only three atoms of the 118-th element were obtained and they decayed during milliseconds. For this rea ...
... Of course, these results obtained by extrapolation are approximate. Moreover, bulk properties such as melting and boiling points can be measured only for significant amounts of an element, whereas only three atoms of the 118-th element were obtained and they decayed during milliseconds. For this rea ...
Amidine: Structure, Reactivity and Complexation Behaviour
... interactions between the benzamidines and their respective environments in the two physical states[24]. Structure and Reactivity Most fundamental aspects in chemical and biochemical studies are the concepts of structure, energetic and reactivity as well as their interrelationships. In most chemical ...
... interactions between the benzamidines and their respective environments in the two physical states[24]. Structure and Reactivity Most fundamental aspects in chemical and biochemical studies are the concepts of structure, energetic and reactivity as well as their interrelationships. In most chemical ...
Calculation of Van der Waals interaction strength between rubidium
... hydrogen, the alkali metals have an outer valence electron in a s-shell. This property makes that the alkali metals are very similar to hydrogen, as you can approximate the alkali metals in many circumstances as being a hydrogen atom with a massive core. In reality, the outer valence electron is sub ...
... hydrogen, the alkali metals have an outer valence electron in a s-shell. This property makes that the alkali metals are very similar to hydrogen, as you can approximate the alkali metals in many circumstances as being a hydrogen atom with a massive core. In reality, the outer valence electron is sub ...
Recycling and Chemical Mathematics
... release oxygen gas in the process. The animal life of Biosphere 2, through the process of breathing (respiration), takes in atmospheric oxygen and releases carbon dioxide. If everything could be arranged to come out even, a stable atmosphere with desirable levels of oxygen and carbon dioxide would b ...
... release oxygen gas in the process. The animal life of Biosphere 2, through the process of breathing (respiration), takes in atmospheric oxygen and releases carbon dioxide. If everything could be arranged to come out even, a stable atmosphere with desirable levels of oxygen and carbon dioxide would b ...
The Effect of Using Computer Simulations in Teaching Chemical
... of objects, atoms, molecules and bacteria, for example, makes learning meaningful and appealing to real life situations. Learners can test theories by developing alternative realities which may greatly facilitate the mastery of difficult concepts; a case in point is the relation between distance, mo ...
... of objects, atoms, molecules and bacteria, for example, makes learning meaningful and appealing to real life situations. Learners can test theories by developing alternative realities which may greatly facilitate the mastery of difficult concepts; a case in point is the relation between distance, mo ...
Chapter 3 Notes
... Analyze We are told that isopropyl alcohol contains C, H, and O atoms and given the quantities of CO 2 and H2O produced when a given quantity of the alcohol is combusted. We must use this information to determine the empirical formula for isopropyl alcohol, a task that requires us to calculate the n ...
... Analyze We are told that isopropyl alcohol contains C, H, and O atoms and given the quantities of CO 2 and H2O produced when a given quantity of the alcohol is combusted. We must use this information to determine the empirical formula for isopropyl alcohol, a task that requires us to calculate the n ...
Paper
... quantum systems depends on the relative phase of the two systems. These effects are direct evidence for the existence of the phase of a macroscopic quantum system [2] and observed in quantum systems such as superconductors [3], superfluid 3 He [4], and Bose condensed gases [5,6]. Josephson coupling ...
... quantum systems depends on the relative phase of the two systems. These effects are direct evidence for the existence of the phase of a macroscopic quantum system [2] and observed in quantum systems such as superconductors [3], superfluid 3 He [4], and Bose condensed gases [5,6]. Josephson coupling ...
Comprehensive analysis of electron correlations in
... those of a two-electron atom? In what way is the description to be modified for doubly excited states of a many-electron atom? Second, can one make the next step to understand the correlation of triply excited states? Both questions can be addressed by studying the doubly and triply excited states o ...
... those of a two-electron atom? In what way is the description to be modified for doubly excited states of a many-electron atom? Second, can one make the next step to understand the correlation of triply excited states? Both questions can be addressed by studying the doubly and triply excited states o ...
Structure and Properties of Polymers
... the Institute of Chemical Technology in Prague and received his PhD and DSc degrees in macromolecular chemistry from the then Czechoslovak Academy of Sciences in 1960 and 1968, respectively. For most of his professional career, he has been affiliated as a Chief Research Fellow with the Institute of ...
... the Institute of Chemical Technology in Prague and received his PhD and DSc degrees in macromolecular chemistry from the then Czechoslovak Academy of Sciences in 1960 and 1968, respectively. For most of his professional career, he has been affiliated as a Chief Research Fellow with the Institute of ...
chapter 1 - Louisiana Tech University
... (1) Temperature and pressure are the major factors that determine the state of matter (solid, liquid, gas) observed for a particular substance. (2) The components of a mixture maintain their identity if the mixture is heterogeneous but lose their identity if the mixture is homogeneous. (3) No more t ...
... (1) Temperature and pressure are the major factors that determine the state of matter (solid, liquid, gas) observed for a particular substance. (2) The components of a mixture maintain their identity if the mixture is heterogeneous but lose their identity if the mixture is homogeneous. (3) No more t ...
Improvement of the Sorption Performance of Nanosilica
... metal removal due to their good physicochemical properties such as porosity and high surface area. However, they do not lead satisfactory efficiency with respect to weak binding properties (7). Consequently, the modification of porous sorbents by immobilization or incorporation of the functional gro ...
... metal removal due to their good physicochemical properties such as porosity and high surface area. However, they do not lead satisfactory efficiency with respect to weak binding properties (7). Consequently, the modification of porous sorbents by immobilization or incorporation of the functional gro ...
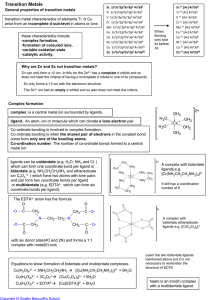
Transition Metals
... Zn can only form a +2 ion. In this ion the Zn2+ has a complete d orbital and so does not meet the criteria of having a incomplete d orbital in one of its compounds. Sc only forms a +3 ion with the electronic structure The Sc3+ ion had an empty d orbital and so also does not meet the criteria ...
... Zn can only form a +2 ion. In this ion the Zn2+ has a complete d orbital and so does not meet the criteria of having a incomplete d orbital in one of its compounds. Sc only forms a +3 ion with the electronic structure The Sc3+ ion had an empty d orbital and so also does not meet the criteria ...
Molecules, Moles and Chemical Equations File
... that releases the same amount of energy. Careful examination of the progress of explosive chemical reactions reveals that they accelerate as they proceed. As a result, all of the available explosive is consumed in a very short period of time. As that happens, the energy from the explosion is also re ...
... that releases the same amount of energy. Careful examination of the progress of explosive chemical reactions reveals that they accelerate as they proceed. As a result, all of the available explosive is consumed in a very short period of time. As that happens, the energy from the explosion is also re ...
1 AM SYLLABUS (2015) CHEMISTRY AM 06 SYLLABUS
... deduce the relative atomic mass of an element from the isotopic masses and relative abundances; describe the use of isotopes as tracers in mechanistic studies using the example of of 18O labelling in ...
... deduce the relative atomic mass of an element from the isotopic masses and relative abundances; describe the use of isotopes as tracers in mechanistic studies using the example of of 18O labelling in ...
Print this article - Bangladesh Journals Online
... assignable for protons Hd and Ha respectively. The two doublets of doublet at δ 6.5 (JHa-Hb = JHb-Hc = J = 8.0 Hz) and 6.9 (JHb-Hc= JHc-Hd = J = 8.0 Hz) accounts for the Ha and Hd respectively, while the relatively downfield signal at δ 8.5 has been assigned for the imine (=N-H) proton of 2-mercapto ...
... assignable for protons Hd and Ha respectively. The two doublets of doublet at δ 6.5 (JHa-Hb = JHb-Hc = J = 8.0 Hz) and 6.9 (JHb-Hc= JHc-Hd = J = 8.0 Hz) accounts for the Ha and Hd respectively, while the relatively downfield signal at δ 8.5 has been assigned for the imine (=N-H) proton of 2-mercapto ...
Theoretical Modeling of Magnesium Ion Imprints in the Raman
... clusters. Minor Raman spectral features could also be explained. Ab initio and Fourier transform (FT) techniques coupled with the Car-Parrinello molecular dynamics were adapted to provide the spectra from dynamical trajectories. The results suggest that even in concentrated solutions magnesium prefe ...
... clusters. Minor Raman spectral features could also be explained. Ab initio and Fourier transform (FT) techniques coupled with the Car-Parrinello molecular dynamics were adapted to provide the spectra from dynamical trajectories. The results suggest that even in concentrated solutions magnesium prefe ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.