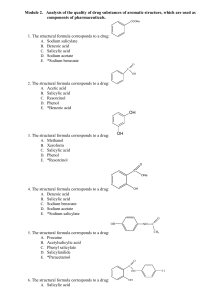
3. Chemical changes and Structure Unit Questions
... o The state symbols are: o (s)-solid o (l)- liquid o (g)- gas o (aq)- in solution (dissolved) o The first 20 elements can be separated into the following: o Metallic-left of the thick black line o Monatomic- noble gases o covalent network- B,C and Si o covalent molecular (including diatomic)- everyt ...
... o The state symbols are: o (s)-solid o (l)- liquid o (g)- gas o (aq)- in solution (dissolved) o The first 20 elements can be separated into the following: o Metallic-left of the thick black line o Monatomic- noble gases o covalent network- B,C and Si o covalent molecular (including diatomic)- everyt ...
AP® Chemistry 2009 Free-Response Questions - AP Central
... NO CALCULATORS MAY BE USED FOR PART B. Answer Question 4 below. The Section II score weighting for this question is 10 percent. 4. For each of the following three reactions, write a balanced equation in part (i) and answer the question in part (ii). In part (i), coefficients should be in terms of lo ...
... NO CALCULATORS MAY BE USED FOR PART B. Answer Question 4 below. The Section II score weighting for this question is 10 percent. 4. For each of the following three reactions, write a balanced equation in part (i) and answer the question in part (ii). In part (i), coefficients should be in terms of lo ...
Theoretical problems - Scheikundeolympiade
... iv) The concentrations at pH 7.5 of HOCl and ClO– in a solution where the total concentration of hypochlorite (chlorate (I)) is 0.20 mmol dm–3, and the electrode potential for the reduction of this system to chlorine at this pH with unit activity of chlorine. These conditions are typical of a swimmi ...
... iv) The concentrations at pH 7.5 of HOCl and ClO– in a solution where the total concentration of hypochlorite (chlorate (I)) is 0.20 mmol dm–3, and the electrode potential for the reduction of this system to chlorine at this pH with unit activity of chlorine. These conditions are typical of a swimmi ...
Identify the following properties as either - Teach-n-Learn-Chem
... 1. Explain what is wrong with the statement “My friend burned a piece of paper (a hydrocarbon) that had the final exam on it and it disappeared”. (Be sure to use a chemical equation, identify reactants and product(s) and include energy). ANSWER: The paper (CxHy) was burned with oxygen and the atoms ...
... 1. Explain what is wrong with the statement “My friend burned a piece of paper (a hydrocarbon) that had the final exam on it and it disappeared”. (Be sure to use a chemical equation, identify reactants and product(s) and include energy). ANSWER: The paper (CxHy) was burned with oxygen and the atoms ...
Acetic acid-water system thermodynamical correlation of vapor
... Following the same procedure, the results of ARICH and TAGLIAVINI[25] were reported at 25°C; in Fig. 4 are reported the one obtained from the experimental runs at 79*9”C. logy r$ agree very well, while log yj: is sensibly different from the other plotted values. Arich and Tagliavini suppose that it ...
... Following the same procedure, the results of ARICH and TAGLIAVINI[25] were reported at 25°C; in Fig. 4 are reported the one obtained from the experimental runs at 79*9”C. logy r$ agree very well, while log yj: is sensibly different from the other plotted values. Arich and Tagliavini suppose that it ...
Praktikum in Allgemeiner Chemie für Biologen und Pharmazeuten
... toxic as KCN. Solutions of toxic compounds are aspirated into pipettes with the aid of a balloon, never orally. For work with gases and vapours like Br2, NO2, HCN etc. a ventilated hood must be used, also with chlorinated solvents and benzene. Organic solvents are often flammable, and their vapours, ...
... toxic as KCN. Solutions of toxic compounds are aspirated into pipettes with the aid of a balloon, never orally. For work with gases and vapours like Br2, NO2, HCN etc. a ventilated hood must be used, also with chlorinated solvents and benzene. Organic solvents are often flammable, and their vapours, ...
An Overview of Organic Reactions
... The energy is mostly determined by the type of bond, independent of the molecule § The C-H bond in methane requires a net energy input of 106 kcal/mol to be broken at 25 ºC. § Table 6.3 lists energies for many bond types Changes in bonds can be used to calculate net changes in heat ...
... The energy is mostly determined by the type of bond, independent of the molecule § The C-H bond in methane requires a net energy input of 106 kcal/mol to be broken at 25 ºC. § Table 6.3 lists energies for many bond types Changes in bonds can be used to calculate net changes in heat ...
- Catalyst
... Possible reaction products are KCl and NH4NO3, or NH4Cl and KNO3. All are soluble, so there is no precipitate. KCl(aq) + NH4NO3 (aq) = No Reaction! Example: If a solution containing sodium sulfate is added to a solution containing barium nitrate, will a precipitate form? ...
... Possible reaction products are KCl and NH4NO3, or NH4Cl and KNO3. All are soluble, so there is no precipitate. KCl(aq) + NH4NO3 (aq) = No Reaction! Example: If a solution containing sodium sulfate is added to a solution containing barium nitrate, will a precipitate form? ...
2014_S4_CHM_NORMAL (ALL)
... 53. Element X (atomic number 11) reacts with element Y (atomic number 16) to form an ionic compound. Each atom of X loses one electron and each atom of Y accepts two electrons to form a compound with formula X2Y. 54. Consider the following information: ...
... 53. Element X (atomic number 11) reacts with element Y (atomic number 16) to form an ionic compound. Each atom of X loses one electron and each atom of Y accepts two electrons to form a compound with formula X2Y. 54. Consider the following information: ...
Complexing properties of bivalent and trivalent iron in the system
... GDR). The Potentiometrie measurements were carried out with a compensation pH-meter pHm 4d, titrigraph SBR 4c (Radiometer, Kopenhagen, Denmark), and a potentiograph E 336 A (Metrohm). The error of Potentiometrie measurements was ± 1 mV. In accord with the theory of redox potential [10], the composit ...
... GDR). The Potentiometrie measurements were carried out with a compensation pH-meter pHm 4d, titrigraph SBR 4c (Radiometer, Kopenhagen, Denmark), and a potentiograph E 336 A (Metrohm). The error of Potentiometrie measurements was ± 1 mV. In accord with the theory of redox potential [10], the composit ...
Otto F. Meyerhof - Nobel Lecture
... the heat occurs in the recovery phase. For if out of six molecules, for instance, only one is burnt then there must arose from the conversion of 1 g of sugar 3772/6 = 630 cal, of which 385 are in the fatigue phase, so that 245 must be in the recovery phase. In this case 60% must already be released ...
... the heat occurs in the recovery phase. For if out of six molecules, for instance, only one is burnt then there must arose from the conversion of 1 g of sugar 3772/6 = 630 cal, of which 385 are in the fatigue phase, so that 245 must be in the recovery phase. In this case 60% must already be released ...
avogadro exam 1994 - University of Waterloo
... 33. Sodium hypochlorite is prepared by the reaction 2 NaOH + Cl2 → NaCl + NaClO + H2O If 71 g of chlorine, Cl2, is bubbled into a solution containing 50 g of sodium hydroxide, which component limits the quantity of sodium hypochlorite formed? ...
... 33. Sodium hypochlorite is prepared by the reaction 2 NaOH + Cl2 → NaCl + NaClO + H2O If 71 g of chlorine, Cl2, is bubbled into a solution containing 50 g of sodium hydroxide, which component limits the quantity of sodium hypochlorite formed? ...
2003
... Substance C conducts electricity in solid and molten states due to free (delocalised) electrons which can move freely through the lattice. Substance D does not conduct electricity in the solid state because ions are fixed. However, when molten, the ions are mobile, free to move and conduct electrici ...
... Substance C conducts electricity in solid and molten states due to free (delocalised) electrons which can move freely through the lattice. Substance D does not conduct electricity in the solid state because ions are fixed. However, when molten, the ions are mobile, free to move and conduct electrici ...
final-H-2006-07-v1
... 65. In general, conditions which increase the number of collisions taking place between reactant molecules have no effect on the rate of a reaction. a. true b. false 66. Which will react faster with water, a lump of calcium, or finely powdered calcium? a. The lump of calcium will react faster. b. Th ...
... 65. In general, conditions which increase the number of collisions taking place between reactant molecules have no effect on the rate of a reaction. a. true b. false 66. Which will react faster with water, a lump of calcium, or finely powdered calcium? a. The lump of calcium will react faster. b. Th ...