Camp 1 - drjosephryan.com Home Page
... exceptions are AgCl, BaSO4, and PbSO4 – most carbonates (CO32-), phosphates (PO43-), sulfides (S2-), and hydroxides (OH-) are insoluble in water; exceptions are LiOH, NaOH, KOH, and NH4OH which are soluble in water ...
... exceptions are AgCl, BaSO4, and PbSO4 – most carbonates (CO32-), phosphates (PO43-), sulfides (S2-), and hydroxides (OH-) are insoluble in water; exceptions are LiOH, NaOH, KOH, and NH4OH which are soluble in water ...
1. Formulae, equations and amounts of substance
... acid in the stomach or acid spills because they are not corrosive and will not cause a hazard if used in excess. They also have no toxicity if used for indigestion remedies but the CO 2 produced can cause wind. Magnesium hydroxide is also suitable for dealing with excess stomach acid as it has low s ...
... acid in the stomach or acid spills because they are not corrosive and will not cause a hazard if used in excess. They also have no toxicity if used for indigestion remedies but the CO 2 produced can cause wind. Magnesium hydroxide is also suitable for dealing with excess stomach acid as it has low s ...
Problem 14. MAGNESIUM DETERMINATION
... In chemical reactions molecular structure changes over time so that the electronic state of a molecule is a function of time. In some cases structure of a molecule can be presented by a superposition of the initial and final states with time-dependent coefficients. Let’s assume that a molecule oscil ...
... In chemical reactions molecular structure changes over time so that the electronic state of a molecule is a function of time. In some cases structure of a molecule can be presented by a superposition of the initial and final states with time-dependent coefficients. Let’s assume that a molecule oscil ...
CH4 Student Revision Guides pdf | GCE AS/A
... Aluminium chloride, AlCl3, is electron deficient, the aluminium having only six valency electrons around the aluminium. The reaction mechanism is thought to involve the formation of [AlCl4]- . The mechanism is electrophilic substitution. Cl Cl Cl Cl ...
... Aluminium chloride, AlCl3, is electron deficient, the aluminium having only six valency electrons around the aluminium. The reaction mechanism is thought to involve the formation of [AlCl4]- . The mechanism is electrophilic substitution. Cl Cl Cl Cl ...
Midterm Practice Exam Key
... Oxidation and reduction must occur together because the number of electrons lost must equal the number of electrons gained. This assures that charge is conserved on both sides of the equation; therefore, an oxidation-reduction reaction, or redox reaction, is a reaction in which electrons are transfe ...
... Oxidation and reduction must occur together because the number of electrons lost must equal the number of electrons gained. This assures that charge is conserved on both sides of the equation; therefore, an oxidation-reduction reaction, or redox reaction, is a reaction in which electrons are transfe ...
CHE 1400 Lab Manual - Al Akhawayn University
... another substance at a given temperature. The test substance is referred to as the solute and the substance in which the test substance is being dissolved is the solvent. A given chemical has different solubilities in different solvents, depending on the similarities in molecular composition of the ...
... another substance at a given temperature. The test substance is referred to as the solute and the substance in which the test substance is being dissolved is the solvent. A given chemical has different solubilities in different solvents, depending on the similarities in molecular composition of the ...
Additional questions
... The value of Henry’s law constant kH for oxygen in water at 24C is 1.66 x 10-6 M/torr. a. Calculate the solubility of oxygen in water at 25C when the total external pressure is 1 atm and the mole fraction of oxygen in the air is 0.20. b. Calculate the solubility at the same temperature with the sa ...
... The value of Henry’s law constant kH for oxygen in water at 24C is 1.66 x 10-6 M/torr. a. Calculate the solubility of oxygen in water at 25C when the total external pressure is 1 atm and the mole fraction of oxygen in the air is 0.20. b. Calculate the solubility at the same temperature with the sa ...
Ex: -F, -Cl, -Br
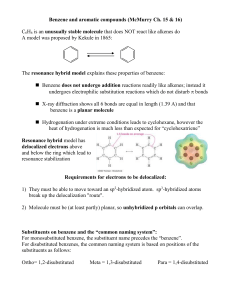
... Compounds which fit the following criteria can be considered aromatic: 1) Cyclic and planar 2) Continuous overlap of unhybridized p-orbitals forming a delocalized -cloud of e3) The number of -electrons must be equal to 4n + 2 (where n = 0, 1, 2…) Huckel’s Rule (4n + 2 rule) relates to the way el ...
... Compounds which fit the following criteria can be considered aromatic: 1) Cyclic and planar 2) Continuous overlap of unhybridized p-orbitals forming a delocalized -cloud of e3) The number of -electrons must be equal to 4n + 2 (where n = 0, 1, 2…) Huckel’s Rule (4n + 2 rule) relates to the way el ...
1 Unit 11-12: Equilibrium and Acid/Bases Notes Colligative
... ‐ Decrease Temp – Shift Right, toward removal ...
... ‐ Decrease Temp – Shift Right, toward removal ...
Chapter 4 Aqueous Reactions and Solution Stoichiometry
... 2. Dissociate all strong electrolytes. 3. Cross out anything that remains unchanged from the left side to the right side of the equation. 4. Write the net ionic equation with the species that remain. Aqueous Reactions ...
... 2. Dissociate all strong electrolytes. 3. Cross out anything that remains unchanged from the left side to the right side of the equation. 4. Write the net ionic equation with the species that remain. Aqueous Reactions ...
The Uptake of Methyl Vinyl Ketone
... was observed, and the reaction was tentatively identified as Pinacol rearrangement. Similar rearrangements could be at the origin of some substituted oxygenated species found in atmospheric aerosols. ...
... was observed, and the reaction was tentatively identified as Pinacol rearrangement. Similar rearrangements could be at the origin of some substituted oxygenated species found in atmospheric aerosols. ...
CHEMISTRY 11 SOLUTION WORKSHEET #4
... Precipitation Problems In the following questions, the two solutions are mixed together and a precipitate is formed. The precipitate is allowed to fall out of solution. We will assume that ALL of one of the ions involved in the reaction is completely used up (i.e. none of it still remains in solutio ...
... Precipitation Problems In the following questions, the two solutions are mixed together and a precipitate is formed. The precipitate is allowed to fall out of solution. We will assume that ALL of one of the ions involved in the reaction is completely used up (i.e. none of it still remains in solutio ...
The Hydroxylation of Aromatic Nitro Compounds by Alkalies
... reacting 8urface. Chemical purity of the potassium hydroxide had no observable effect, but the presence of more than two percent ot water in It lowered the yield markedly. Intensive drying was therefore tried, but neither precautions taken In grinding, nor preliminary fusion of the alkali, nor the a ...
... reacting 8urface. Chemical purity of the potassium hydroxide had no observable effect, but the presence of more than two percent ot water in It lowered the yield markedly. Intensive drying was therefore tried, but neither precautions taken In grinding, nor preliminary fusion of the alkali, nor the a ...
Test - Regents
... nucleus has a positive charge. (2) An atom is mainly empty space, and nucleus has a negative charge. (3) An atom has hardly any empty space, the nucleus has a positive charge. (4) An atom has hardly any empty space, the nucleus has a negative charge. ...
... nucleus has a positive charge. (2) An atom is mainly empty space, and nucleus has a negative charge. (3) An atom has hardly any empty space, the nucleus has a positive charge. (4) An atom has hardly any empty space, the nucleus has a negative charge. ...
- Department of Chemistry, York University
... (NH2CH2COOH)H+ +CH3COOH(CH3CONHCH2COOH)H++H2O protonated N-acetyl-glycine (CH3CONHCH2COOH)H+ + NH2OH no (clusters) (NH2CH2CONHCH2COOH)H+ + H2O Fe+CH3CONHCH2COOH + NH2OH ? (too complicated) Fe+NH2CH2CONHCH2COOH + H2O diglycine, a dipeptide M+(Gly)n + CH3COOH + NH2OH M+(Gly)n+1 + H2O (M+ assemb ...
... (NH2CH2COOH)H+ +CH3COOH(CH3CONHCH2COOH)H++H2O protonated N-acetyl-glycine (CH3CONHCH2COOH)H+ + NH2OH no (clusters) (NH2CH2CONHCH2COOH)H+ + H2O Fe+CH3CONHCH2COOH + NH2OH ? (too complicated) Fe+NH2CH2CONHCH2COOH + H2O diglycine, a dipeptide M+(Gly)n + CH3COOH + NH2OH M+(Gly)n+1 + H2O (M+ assemb ...
Experimental determination of hydromagnesite precipitation rates
... It is surprising that such a simple way to solve this crucial problem has not been described in the geothermal scientific literature, as far as we know. For example, the classical textbook of Ellis and Mahon (1977) reports that “if sulfide or sulfur dioxide are present, the silicomolybdic acid turns ...
... It is surprising that such a simple way to solve this crucial problem has not been described in the geothermal scientific literature, as far as we know. For example, the classical textbook of Ellis and Mahon (1977) reports that “if sulfide or sulfur dioxide are present, the silicomolybdic acid turns ...
Loeblein chemistry clicker questions2013
... A. The ratio would be the same, but the balls would be connected B. The ratio would change to 1 magnesium for every oxide and balls would be ...
... A. The ratio would be the same, but the balls would be connected B. The ratio would change to 1 magnesium for every oxide and balls would be ...