Higher Chemistry Specimen Question Paper
... This reaction is an example of A condensation B esterification C hydrolysis D oxidation. ...
... This reaction is an example of A condensation B esterification C hydrolysis D oxidation. ...
CHE 1031 Lab Manual
... corrosivity, reactivity or toxicity. In order to protect yourself from the chemicals you use in the chemistry laboratory, it is important to understand and be aware of these hazards. In addition, essential ...
... corrosivity, reactivity or toxicity. In order to protect yourself from the chemicals you use in the chemistry laboratory, it is important to understand and be aware of these hazards. In addition, essential ...
Selective Oxidation of Methane over Vycor Glass
... [ 301. Quartz glass from Wale Apparatus Co. was used. All the runs with catalyst beds were performed in quartz U-tubes. Fumed silica (Cab-0-Sil) made by the Cabot corporation was pelletized prior to loading into the reactor. The silicic acid was Mallinckrodt analytical reagent grade with very low im ...
... [ 301. Quartz glass from Wale Apparatus Co. was used. All the runs with catalyst beds were performed in quartz U-tubes. Fumed silica (Cab-0-Sil) made by the Cabot corporation was pelletized prior to loading into the reactor. The silicic acid was Mallinckrodt analytical reagent grade with very low im ...
Chemistry - Separation techniques
... C3.1k deduce the stoichiometry of an equation from the masses of reactants and products and explain the effect of a limiting quantity of a reactant C3.3d describe neutralisation as acid reacting with alkali or a base to form a salt plus water C4.1c recall the general properties of transition metals ...
... C3.1k deduce the stoichiometry of an equation from the masses of reactants and products and explain the effect of a limiting quantity of a reactant C3.3d describe neutralisation as acid reacting with alkali or a base to form a salt plus water C4.1c recall the general properties of transition metals ...
CHM203 - National Open University of Nigeria
... in the London forces between the larger molecules of higher molecular weight. Thus, each additional methylene (-CH2) unit contributes to the increase in melting point. In a homologous series, the higher the molecular weight, the larger will be the molecules and the greater will be the ‘area of conta ...
... in the London forces between the larger molecules of higher molecular weight. Thus, each additional methylene (-CH2) unit contributes to the increase in melting point. In a homologous series, the higher the molecular weight, the larger will be the molecules and the greater will be the ‘area of conta ...
3.5 Empirical Formulas - Mayfield City Schools
... 8. Epsom salts, a strong laxative used in veterinary medicine, is a hydrate, which means that a certain number of water molecules are included in the solid structure. The formula for Epsom salts can be written as MgSO4•xH2O, where x indicates the number of moles of H2O per mole of MgSO4. When 5.061 ...
... 8. Epsom salts, a strong laxative used in veterinary medicine, is a hydrate, which means that a certain number of water molecules are included in the solid structure. The formula for Epsom salts can be written as MgSO4•xH2O, where x indicates the number of moles of H2O per mole of MgSO4. When 5.061 ...
Neutral ionic liquid [BMIm]BF4 promoted highly selective
... 99% selectivity to tert-butyl acetate were obtained under the identical conditions (Table 1, Entry 4). This unusual observation promoted us to further investigate the effects of other ionic liquids on the esterification reaction. The rate of the formation of tert-butyl acetate in [BMIm]BF4 at 60 ◦ C ...
... 99% selectivity to tert-butyl acetate were obtained under the identical conditions (Table 1, Entry 4). This unusual observation promoted us to further investigate the effects of other ionic liquids on the esterification reaction. The rate of the formation of tert-butyl acetate in [BMIm]BF4 at 60 ◦ C ...
NIckel AdsorptIoN oNto cArboN ANode dust modIfIed by AcetIc AcId
... the specially arranged landfil. This process is rarely applied; it is expensive and requires a lot of area. Therefore, it is necessary to find its use as a secondary raw material. The adsorption capacities of the adsorbent depend on the characteristics of the individual adsorbens, its surface proper ...
... the specially arranged landfil. This process is rarely applied; it is expensive and requires a lot of area. Therefore, it is necessary to find its use as a secondary raw material. The adsorption capacities of the adsorbent depend on the characteristics of the individual adsorbens, its surface proper ...
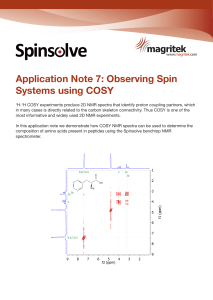
Observing Spin Systems using COSY
... rest of the molecule. Often these form ‘puzzle pieces’ in structural elucidation that can be pieced together using other 2D NMR experiments. ...
... rest of the molecule. Often these form ‘puzzle pieces’ in structural elucidation that can be pieced together using other 2D NMR experiments. ...
Electrochemistry
... A galvanic cell is composed of aqueous Cr2O72-, Cr3+, Fe3+ and Fe2+ ions. At 25°C it runs at a pH of 2.75. a. Write the balanced spontaneous reaction for the galvanic cell. b. Draw a diagram of the galvanic cell. Identify the anode and the cathode. Indicate the direction of electron flow. c. Determi ...
... A galvanic cell is composed of aqueous Cr2O72-, Cr3+, Fe3+ and Fe2+ ions. At 25°C it runs at a pH of 2.75. a. Write the balanced spontaneous reaction for the galvanic cell. b. Draw a diagram of the galvanic cell. Identify the anode and the cathode. Indicate the direction of electron flow. c. Determi ...
Application Note 7: Observing Spin Systems using COSY
... rest of the molecule. Often these form ‘puzzle pieces’ in structural elucidation that can be pieced together using other 2D NMR experiments. ...
... rest of the molecule. Often these form ‘puzzle pieces’ in structural elucidation that can be pieced together using other 2D NMR experiments. ...
Stains for Developing TLC Plates
... Once a TLC has been developed, it is frequently necessary to aid in the visualization of the components of a reaction mixture. This is true primarily because most organic compounds are colorless. Frequently, the organic compounds of interest contain a chromophore which may be visualized by employing ...
... Once a TLC has been developed, it is frequently necessary to aid in the visualization of the components of a reaction mixture. This is true primarily because most organic compounds are colorless. Frequently, the organic compounds of interest contain a chromophore which may be visualized by employing ...
Calculations on the equations reaction
... Determine the degree of oxidation of elements. Write the oxidant and reductant. 7. To equalize the oxidation-reduction reaction by electron balance method Cl2 + KOH → KCl + KClO3 + H2O Determine the degree of oxidation of elements. Write the oxidant and reductant. 8. To equalize the oxidation-reduct ...
... Determine the degree of oxidation of elements. Write the oxidant and reductant. 7. To equalize the oxidation-reduction reaction by electron balance method Cl2 + KOH → KCl + KClO3 + H2O Determine the degree of oxidation of elements. Write the oxidant and reductant. 8. To equalize the oxidation-reduct ...
Review Answers - cloudfront.net
... (d) Describe how the value of the acid-dissociation constant, Ka, for the weak acid HX could be determined from the titration curve in part (c). At ½ the equivalence point (12.5mL) the pH=pKa.. Then use the equation 10-pKa = Ka (e) The graph to the right shows the results obtained by titrating a dif ...
... (d) Describe how the value of the acid-dissociation constant, Ka, for the weak acid HX could be determined from the titration curve in part (c). At ½ the equivalence point (12.5mL) the pH=pKa.. Then use the equation 10-pKa = Ka (e) The graph to the right shows the results obtained by titrating a dif ...
College Chemistry 1 Note Guide(free download)
... All college chemistry courses are not organized in the same manner. The American Chemical Society provided recommended content information to members regarding these undergraduate courses, but did not specify the order in which topics were to be addressed. Although Professor Etheridge has included t ...
... All college chemistry courses are not organized in the same manner. The American Chemical Society provided recommended content information to members regarding these undergraduate courses, but did not specify the order in which topics were to be addressed. Although Professor Etheridge has included t ...
1. Atomic Structure and Periodic Table THE MASS SPECTROMETER
... reducing agents are electron donors oxidising agents are electron acceptors When naming oxidising and reducing agents always refer to full name of substance and not just name of element ...
... reducing agents are electron donors oxidising agents are electron acceptors When naming oxidising and reducing agents always refer to full name of substance and not just name of element ...
chemistry - Rwanda Education Board
... 9. Define oxidation and reduction in terms of loss or gain of oxygen or hydrogen and in terms of loss or gain of electrons. 10. Identify the substances that are oxidized and those that are reduced in a redox reaction. 11. Distinguish between oxidizing agent and reducing agent in a redox reaction. 12 ...
... 9. Define oxidation and reduction in terms of loss or gain of oxygen or hydrogen and in terms of loss or gain of electrons. 10. Identify the substances that are oxidized and those that are reduced in a redox reaction. 11. Distinguish between oxidizing agent and reducing agent in a redox reaction. 12 ...
WRITING CHEMICAL FORMULAE
... What mass of magnesium chloride can be produced by the reaction between 3.6 g of magnesium and excess hydrochloric acid? Mg + 2 HCl ...
... What mass of magnesium chloride can be produced by the reaction between 3.6 g of magnesium and excess hydrochloric acid? Mg + 2 HCl ...






![Neutral ionic liquid [BMIm]BF4 promoted highly selective](http://s1.studyres.com/store/data/017897985_1-047f9869d5604c115b21339541ccfffe-300x300.png)