KEY + + - UIC Department of Chemistry
... (d) salt + CO2(g) + H 2 O 3. A compound was found to contain 54.5% carbon, 9.2% hydrogen, and 36.3% oxygen by mass. What is the empirical formula of the compound? (a) C6 HO4 (b) C4 H8 O2 (c) C2 H4 O (d) C3 H4 O (e) C3 H4 O3 Questions 4 – 6 refer to the following unbalanced equation: Fe(s) ...
... (d) salt + CO2(g) + H 2 O 3. A compound was found to contain 54.5% carbon, 9.2% hydrogen, and 36.3% oxygen by mass. What is the empirical formula of the compound? (a) C6 HO4 (b) C4 H8 O2 (c) C2 H4 O (d) C3 H4 O (e) C3 H4 O3 Questions 4 – 6 refer to the following unbalanced equation: Fe(s) ...
Mechanistic Studies of the Reactions of Silicon
... latter solvent compared to that in acetonitrile and hexane, consistent with the formation of a solvent complex. Steady-state competition experiments have been carried out with various pairs of alcohols and water. The product ratios agree with the corresponding relative rate constants for water, meth ...
... latter solvent compared to that in acetonitrile and hexane, consistent with the formation of a solvent complex. Steady-state competition experiments have been carried out with various pairs of alcohols and water. The product ratios agree with the corresponding relative rate constants for water, meth ...
Chemistry: An Introduction for Medical and Health Sciences - E
... of aspirin-like medicines in Britain. It cured the pains from various complaints. Herbal concoctions have been the basis of healing and also poisoning for centuries. Curare was used on the tips of poison darts to kill opponents, but in smaller quantities it was used as a muscle relaxant in surgery u ...
... of aspirin-like medicines in Britain. It cured the pains from various complaints. Herbal concoctions have been the basis of healing and also poisoning for centuries. Curare was used on the tips of poison darts to kill opponents, but in smaller quantities it was used as a muscle relaxant in surgery u ...
Chemistry - An Introduction for Medical and Hea..
... of aspirin-like medicines in Britain. It cured the pains from various complaints. Herbal concoctions have been the basis of healing and also poisoning for centuries. Curare was used on the tips of poison darts to kill opponents, but in smaller quantities it was used as a muscle relaxant in surgery u ...
... of aspirin-like medicines in Britain. It cured the pains from various complaints. Herbal concoctions have been the basis of healing and also poisoning for centuries. Curare was used on the tips of poison darts to kill opponents, but in smaller quantities it was used as a muscle relaxant in surgery u ...
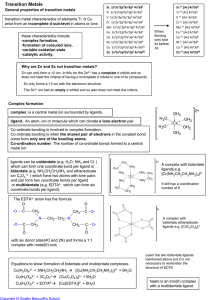
Transition Metals
... ligands can be unidentate (e.g. H2O, NH3 and Cl- ) which can form one coordinate bond per ligand or bidentate (e.g. NH2CH2CH2NH2 and ethanedioate ion C2O4 2- ) which have two atoms with lone pairs and can form two coordinate bonds per ligand or multidentate (e.g. EDTA4- which can form six coordinate ...
... ligands can be unidentate (e.g. H2O, NH3 and Cl- ) which can form one coordinate bond per ligand or bidentate (e.g. NH2CH2CH2NH2 and ethanedioate ion C2O4 2- ) which have two atoms with lone pairs and can form two coordinate bonds per ligand or multidentate (e.g. EDTA4- which can form six coordinate ...
Chem 171-2-3: Final Exam Review Multiple Choice Problems 1
... A gas mixture contains 3.00 atm of H2 and 1.00 atm of O2 in a 1.00 L vessel at 400K. If the mixture burns to form water while the temperature is held at 400K, what is the partial pressure of H2O? a. 5.00 atm ...
... A gas mixture contains 3.00 atm of H2 and 1.00 atm of O2 in a 1.00 L vessel at 400K. If the mixture burns to form water while the temperature is held at 400K, what is the partial pressure of H2O? a. 5.00 atm ...
Tech Tip 0013 - Hydrates and Salts
... Confusion is not uncommon when reporting the final values calculated with using such standards. The analyst must first determine if the end result will be reported as the hydrate or salt, or as the compound without the hydrate or salt. This determines how the initial calibration curve is generated. ...
... Confusion is not uncommon when reporting the final values calculated with using such standards. The analyst must first determine if the end result will be reported as the hydrate or salt, or as the compound without the hydrate or salt. This determines how the initial calibration curve is generated. ...
Chemical Reactions and Equations - 2012 Book Archive
... two solid rocket boosters, which use a solid fuel mixture that contains mainly ammonium perchlorate and powdered aluminum. The chemical reaction between these substances produces aluminum oxide, water, nitrogen gas, and hydrogen chloride. Although the solid rocket boosters each have a significantly ...
... two solid rocket boosters, which use a solid fuel mixture that contains mainly ammonium perchlorate and powdered aluminum. The chemical reaction between these substances produces aluminum oxide, water, nitrogen gas, and hydrogen chloride. Although the solid rocket boosters each have a significantly ...
- Mendeley Data
... +4 and +5 are the most stable ones under aerobic conditions), (c) the Lewis acid character of the vanadium centers and (d) the high affinity of the metal toward oxygen [22-24]. Since epoxides are the key starting materials for a wide variety of products, the epoxidation of alkenes is considered to ...
... +4 and +5 are the most stable ones under aerobic conditions), (c) the Lewis acid character of the vanadium centers and (d) the high affinity of the metal toward oxygen [22-24]. Since epoxides are the key starting materials for a wide variety of products, the epoxidation of alkenes is considered to ...
Year 11 C2 Mock Exam Revision Questions
... Oxygen atoms have 8 electrons. Complete the diagram to represent the arrangement of electrons in an oxygen atom. Use crosses (×) to represent the electrons. ...
... Oxygen atoms have 8 electrons. Complete the diagram to represent the arrangement of electrons in an oxygen atom. Use crosses (×) to represent the electrons. ...
St. Xavier`s College – Autonomous Mumbai Syllabus for 3 Semester
... 1. To understand some more concepts of thermodynamics from a chemist’s viewpoint. 2. To predict the feasibility of a reaction. 3. To understand concepts involved in electrolytic cells and their applications. 4. To motivate students to solve numerical problems with different systems of units which il ...
... 1. To understand some more concepts of thermodynamics from a chemist’s viewpoint. 2. To predict the feasibility of a reaction. 3. To understand concepts involved in electrolytic cells and their applications. 4. To motivate students to solve numerical problems with different systems of units which il ...
Stereochemistry - Kantipur Engineering College
... enantiomers and diastereomers. The students should understand basic concepts like chirality, plane polarized light, specific rotation, stereocenters, maximum number of stereoisomers related to the number of chiral centers, racemic mixture, meso compound, and ways to separate enantiomers from their r ...
... enantiomers and diastereomers. The students should understand basic concepts like chirality, plane polarized light, specific rotation, stereocenters, maximum number of stereoisomers related to the number of chiral centers, racemic mixture, meso compound, and ways to separate enantiomers from their r ...
Solids Chemistry XII - The Gurukul Institute
... SOLUTIONS 1 MARK QUESTIONS Give two examples of gaseous solution. When would dissolving of solute in a solvent leads to liberation of heat energy? How is it that NaCl is soluble in water but not in benzene? What is the weight percent of a solution? State the unit of it in which it is expressed. ...
... SOLUTIONS 1 MARK QUESTIONS Give two examples of gaseous solution. When would dissolving of solute in a solvent leads to liberation of heat energy? How is it that NaCl is soluble in water but not in benzene? What is the weight percent of a solution? State the unit of it in which it is expressed. ...
Molecules, Moles and Chemical Equations File
... probably know that gases have much lower densities than solids. So the gaseous products tend to expand rapidly to reach their normal densities. This expansion typically results in the formation of a shock wave, which helps to deliver the blow of the explosion to its surroundings as shown in Figure 3 ...
... probably know that gases have much lower densities than solids. So the gaseous products tend to expand rapidly to reach their normal densities. This expansion typically results in the formation of a shock wave, which helps to deliver the blow of the explosion to its surroundings as shown in Figure 3 ...