College Chemistry I PHS 1025 Fall 2012 Practice Exam 3A
... Cr(OH)4-(aq) → CrO42-(aq) + 4 H+(aq) + 3eD) Cr(OH)4-(aq) + 4 OH-(aq) → CrO42-(aq) + 4 H2O(l) + 3e46) If 100. mL of 0.100 M Na2SO4 is added to 200. mL of 0.150 M NaCl, what is the concentration of Na+ ions in the final solution? Assume that the volumes are additive. A) 0.167 M ...
... Cr(OH)4-(aq) → CrO42-(aq) + 4 H+(aq) + 3eD) Cr(OH)4-(aq) + 4 OH-(aq) → CrO42-(aq) + 4 H2O(l) + 3e46) If 100. mL of 0.100 M Na2SO4 is added to 200. mL of 0.150 M NaCl, what is the concentration of Na+ ions in the final solution? Assume that the volumes are additive. A) 0.167 M ...
Equilibrium STUDY GUIDE by Keshara Senanayake ---
... The equation for this is AgCl(s) <----- Ag+ (aq) + Cl - (aq) All of this is factored back into solubility (which is the maximum quantity of that substance that call dissolve in a given solvent) at some point no additional AgCl can dissolve because the solution is saturated. Although ions continue to ...
... The equation for this is AgCl(s) <----- Ag+ (aq) + Cl - (aq) All of this is factored back into solubility (which is the maximum quantity of that substance that call dissolve in a given solvent) at some point no additional AgCl can dissolve because the solution is saturated. Although ions continue to ...
GCE Chemistry SAMs 2009 onwards pdf
... Reliable resources of energy need to be available in the future. A UK report anticipates the differing quantities of fuels needed in 50 years time. In this report three predictions are made based on different assumptions about future energy supply and demand. Among the assumptions the following were ...
... Reliable resources of energy need to be available in the future. A UK report anticipates the differing quantities of fuels needed in 50 years time. In this report three predictions are made based on different assumptions about future energy supply and demand. Among the assumptions the following were ...
STUDY GUIDE
... multiple bonds, and they are more reactive than alkanes. They may participate in addition reactions, including hydrogenation, halogenation, hydrohalogenation, and hydration. A naming scheme has been established by the International Union of Pure and Applied Chemistry (IUPAC) to name the organic comp ...
... multiple bonds, and they are more reactive than alkanes. They may participate in addition reactions, including hydrogenation, halogenation, hydrohalogenation, and hydration. A naming scheme has been established by the International Union of Pure and Applied Chemistry (IUPAC) to name the organic comp ...
Chapter 2 Matter and Components F11 110
... 3. For Type II metals with only two common oxidation states an older, Latin system was once used; while it is not employed very often it is useful to know some simple rules regarding it. It is sometimes called the “-ous/-ic” system, where the LOWER charged cation will be denoted by the latin root na ...
... 3. For Type II metals with only two common oxidation states an older, Latin system was once used; while it is not employed very often it is useful to know some simple rules regarding it. It is sometimes called the “-ous/-ic” system, where the LOWER charged cation will be denoted by the latin root na ...
Chapter 2 Matter and Components F11 110pt
... LOWER charged cation will be denoted by the latin root name for that element plus -ous and the HIGHER charged cation will be denoted by the latin root name for that element ...
... LOWER charged cation will be denoted by the latin root name for that element plus -ous and the HIGHER charged cation will be denoted by the latin root name for that element ...
chemistry writing team
... fraction of ethanol is 0.040. Assume the density of water to be 1.0 g mL−1. [Ans. : 2.31 Μ] How many grams of HCl react with 5.0 g MnO2 according to the equation : 4HCl (aq) + MnO2 (s) → 2H2O (l) + MnCl2 (aq) + Cl2 (g) [ Ans. : 8.40 g] How are 0.5 mol Na2CO3 and 0.5 M Na2CO3 are different from each ...
... fraction of ethanol is 0.040. Assume the density of water to be 1.0 g mL−1. [Ans. : 2.31 Μ] How many grams of HCl react with 5.0 g MnO2 according to the equation : 4HCl (aq) + MnO2 (s) → 2H2O (l) + MnCl2 (aq) + Cl2 (g) [ Ans. : 8.40 g] How are 0.5 mol Na2CO3 and 0.5 M Na2CO3 are different from each ...
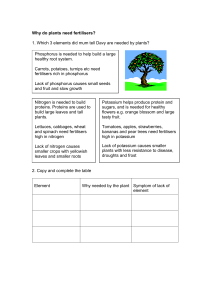
Fertilisers
... nitrate ( NH4 NO3 ), a nitrogen only fertiliser would be suitable. Wheat requires all three essential elements so a NPK compound fertiliser is used. NPK fertilisers are usually a mixture of ammonium nitrate (NH4 NO3 ) ammonium phosphate(( NH4)3 PO4 ) and potassium chloride (KCl). Some examples of br ...
... nitrate ( NH4 NO3 ), a nitrogen only fertiliser would be suitable. Wheat requires all three essential elements so a NPK compound fertiliser is used. NPK fertilisers are usually a mixture of ammonium nitrate (NH4 NO3 ) ammonium phosphate(( NH4)3 PO4 ) and potassium chloride (KCl). Some examples of br ...
On the Networking Mechanisms of Additives
... also presented. q 1997 John Wiley & Sons, Inc. J Appl Polym Sci 66: 255–266, 1997 ...
... also presented. q 1997 John Wiley & Sons, Inc. J Appl Polym Sci 66: 255–266, 1997 ...
Redox speciation analysis of antimony in soil extracts by hydride
... 3. Results and discussion 3.1. Optimization of the redox speciation analysis of antimony The determination of the redox species of antimony by HG-AFS in soil extracts was based on the selective generation of stibine from Sb(III) by reaction with borohydride in citric acid–HCl medium. As was describe ...
... 3. Results and discussion 3.1. Optimization of the redox speciation analysis of antimony The determination of the redox species of antimony by HG-AFS in soil extracts was based on the selective generation of stibine from Sb(III) by reaction with borohydride in citric acid–HCl medium. As was describe ...
Soft X-Ray-Induced Decomposition of Amino Acids: An XPS, Mass
... spectra have complicated asymmetric shapes due to contributions of several functional groups and shake-up satellites.3 For the assignment of the spectral features, it must be taken into account that the form of amino acids that is most stable in the solid state is a zwitterion with a protonated amin ...
... spectra have complicated asymmetric shapes due to contributions of several functional groups and shake-up satellites.3 For the assignment of the spectral features, it must be taken into account that the form of amino acids that is most stable in the solid state is a zwitterion with a protonated amin ...
1.2 Calculations
... number of particles as there are atoms in 12 grams of carbon-12. DEFINITION: Relative atomic mass is the average mass of one atom compared to one twelfth of the mass of one atom of carbon-12 DEFINITION: Relative molecular mass is the average mass of a molecule compared to one twelfth of the mass of ...
... number of particles as there are atoms in 12 grams of carbon-12. DEFINITION: Relative atomic mass is the average mass of one atom compared to one twelfth of the mass of one atom of carbon-12 DEFINITION: Relative molecular mass is the average mass of a molecule compared to one twelfth of the mass of ...
Exam Review Packet Table of Contents
... An incorrect statement in an otherwise correct 2 pt response will result in a score of 1 pt The answers labeled (i) below received two points; (ii) received one point. a) two points -‐ The ...
... An incorrect statement in an otherwise correct 2 pt response will result in a score of 1 pt The answers labeled (i) below received two points; (ii) received one point. a) two points -‐ The ...
teaching and learning materials - UNESDOC
... questions about these important missing areas. This workbook responds to these questions, by offering a limited repertoire of basic organic chemistry experiences that can be provided with low-cost small scale equipment. They have been developed at the RADMASTE Centre, University of the Witwatersrand ...
... questions about these important missing areas. This workbook responds to these questions, by offering a limited repertoire of basic organic chemistry experiences that can be provided with low-cost small scale equipment. They have been developed at the RADMASTE Centre, University of the Witwatersrand ...
Question Bank - Edudel.nic.in
... separated by a semi-permeable membrane will there be appearance of a white ppt. in the side ‘X’ due to osmosis? 0.1 M AgNO3 X ...
... separated by a semi-permeable membrane will there be appearance of a white ppt. in the side ‘X’ due to osmosis? 0.1 M AgNO3 X ...
Reactions in Aqueous Solution
... Reactions in Aqueous Solution In Chapter 3 "Chemical Reactions", we described chemical reactions in general and introduced some techniques that are used to characterize them quantitatively. For the sake of simplicity, we discussed situations in which the reactants and the products of a given reactio ...
... Reactions in Aqueous Solution In Chapter 3 "Chemical Reactions", we described chemical reactions in general and introduced some techniques that are used to characterize them quantitatively. For the sake of simplicity, we discussed situations in which the reactants and the products of a given reactio ...
Chapter 4 "Reactions in Aqueous Solution"
... Reactions in Aqueous Solution In Chapter 3 "Chemical Reactions", we described chemical reactions in general and introduced some techniques that are used to characterize them quantitatively. For the sake of simplicity, we discussed situations in which the reactants and the products of a given reactio ...
... Reactions in Aqueous Solution In Chapter 3 "Chemical Reactions", we described chemical reactions in general and introduced some techniques that are used to characterize them quantitatively. For the sake of simplicity, we discussed situations in which the reactants and the products of a given reactio ...
Problem Authors - PianetaChimica
... Preparatory Problems, Theoretical contaminations. The reaction of sulfuric acid with colemanite takes place in two steps: In the first step colemanite is dissolved in sulfuric acid forming the calcium(II) ion and boric acid. In the second step, calcium sulfate, formed from Ca2+ and SO42− ions, prec ...
... Preparatory Problems, Theoretical contaminations. The reaction of sulfuric acid with colemanite takes place in two steps: In the first step colemanite is dissolved in sulfuric acid forming the calcium(II) ion and boric acid. In the second step, calcium sulfate, formed from Ca2+ and SO42− ions, prec ...
ap 2005 chemistry_b scoring guidelines - AP Central
... Write the formulas to show the reactants and the products for any FIVE of the laboratory situations described below. Answers to more than five choices will not be graded. In all cases, a reaction occurs. Assume that solutions are aqueous unless otherwise indicated. Represent substances in solution a ...
... Write the formulas to show the reactants and the products for any FIVE of the laboratory situations described below. Answers to more than five choices will not be graded. In all cases, a reaction occurs. Assume that solutions are aqueous unless otherwise indicated. Represent substances in solution a ...
selection of chelating agents for remediation of radionuclide
... functions separated by two or three carbon atoms. Compounds having more than one kind of heterotom in the ring are called mixed-donor macrocycles. Three-dimensional, polymacrocyclic chelating agents are formed by joining bridgehead structures with chains that contain properly spaced donor atoms. Suc ...
... functions separated by two or three carbon atoms. Compounds having more than one kind of heterotom in the ring are called mixed-donor macrocycles. Three-dimensional, polymacrocyclic chelating agents are formed by joining bridgehead structures with chains that contain properly spaced donor atoms. Suc ...