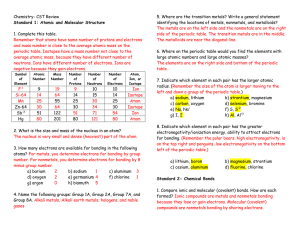
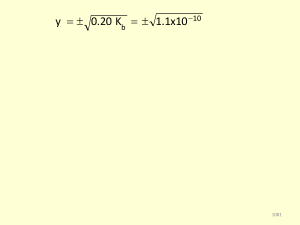Test 2 - Northwest Florida State College
... ketones, carboxylic acid, esters, amines, mole-to-mole ratio (mole ratio), limiting reactant, excess reactant, actual yield, theoretical yield, percent yield, solute, solvent, solution, Molarity (M), concentrated solution, diluted solution, concentration, making a solution by dilution method, electr ...
... ketones, carboxylic acid, esters, amines, mole-to-mole ratio (mole ratio), limiting reactant, excess reactant, actual yield, theoretical yield, percent yield, solute, solvent, solution, Molarity (M), concentrated solution, diluted solution, concentration, making a solution by dilution method, electr ...
PowerPoint
... Writing Balanced Equations for Neutralization Reactions Problem: Write balanced molecular and net ionic equations for the following chemical ...
... Writing Balanced Equations for Neutralization Reactions Problem: Write balanced molecular and net ionic equations for the following chemical ...
10th Carbon and Its Compounds Solved Paper-3
... 10. An organic compound A is widely used as a preservative in pickles and has a molecular formula C2H4O2. This compound reacts with ethanol to form a sweet smelling compound B. i) Identify the compound A. ii) Write the chemical equation for its reaction with Ethanol to form compound B. iii) How can ...
... 10. An organic compound A is widely used as a preservative in pickles and has a molecular formula C2H4O2. This compound reacts with ethanol to form a sweet smelling compound B. i) Identify the compound A. ii) Write the chemical equation for its reaction with Ethanol to form compound B. iii) How can ...
ouble Replacement or (Metathesis) Reactions
... Since all these happens on the negative electrode. So, H2 gas forms from the negative electrode and that's exactly what happens when water reduces at the cathode. 8. (Trick #2) When CuSO4(aq) is electrolyzed, you know that Cu° metal is going to form because copper's potential is higher than water. S ...
... Since all these happens on the negative electrode. So, H2 gas forms from the negative electrode and that's exactly what happens when water reduces at the cathode. 8. (Trick #2) When CuSO4(aq) is electrolyzed, you know that Cu° metal is going to form because copper's potential is higher than water. S ...
Introduction(s)
... convert back into Na2CO3(s). Other carbonates follow this same pattern, as seen in the reaction of carbonic acid; H2CO3(aq) CO2(g) + H2O. ...
... convert back into Na2CO3(s). Other carbonates follow this same pattern, as seen in the reaction of carbonic acid; H2CO3(aq) CO2(g) + H2O. ...
Memorization?
... convert back into Na2CO3(s). Other carbonates follow this same pattern, as seen in the reaction of carbonic acid; H2CO3(aq) CO2(g) + H2O. ...
... convert back into Na2CO3(s). Other carbonates follow this same pattern, as seen in the reaction of carbonic acid; H2CO3(aq) CO2(g) + H2O. ...
Chemistry- CST Review
... Standard 4 – Gases and their Properties 1. What causes gas pressure in terms of kinetic theory? Gas pressure is caused by the random motion of the gas molecules. 2. If someone sprays perfume at the front of the room, will the people in the back of the room eventually be able to smell it? Why? Expla ...
... Standard 4 – Gases and their Properties 1. What causes gas pressure in terms of kinetic theory? Gas pressure is caused by the random motion of the gas molecules. 2. If someone sprays perfume at the front of the room, will the people in the back of the room eventually be able to smell it? Why? Expla ...
factors affecting strength of acids
... - Bases are proton acceptors - Not restricted to aqueous solutions NH3, dimethyl sulfoxide (DMSO) - Proton donation cannot occur unless an acceptor is present ...
... - Bases are proton acceptors - Not restricted to aqueous solutions NH3, dimethyl sulfoxide (DMSO) - Proton donation cannot occur unless an acceptor is present ...
Glossary (PDF file)
... table was developed by the Russian chemist Dmitri Mendeleev. The chart shows all the known elements on Earth. The elements are arranged according to their atomic numbers and their properties. pH A measure of the amount of acid in a solution. A low pH means a solution is acidic. Stomach acid has a pH ...
... table was developed by the Russian chemist Dmitri Mendeleev. The chart shows all the known elements on Earth. The elements are arranged according to their atomic numbers and their properties. pH A measure of the amount of acid in a solution. A low pH means a solution is acidic. Stomach acid has a pH ...
American-Journal-of-Oil-and-Chemical-Technologies
... In recent years, pyridinedicarboxylic acid derivatives have attracted much interest in designing and synthesis of novel compounds with desirable properties. Also they are a class of organic compounds with variety roles in biology and medicine and they are present in many natural products, such as al ...
... In recent years, pyridinedicarboxylic acid derivatives have attracted much interest in designing and synthesis of novel compounds with desirable properties. Also they are a class of organic compounds with variety roles in biology and medicine and they are present in many natural products, such as al ...
chemistry 103 - chem.uwec.edu
... Example: Calculate the pH of a buffer system containing 1.0 M CH3CO2H and 1.0 M NaCH3CO2. What is the pH of the buffer after the addition of 0.10 moles of gaseous HCl to 1.00 liter of the buffer solution? The Ka for acetic acid is 1.8 x 10-5. Because acetic acid is a weak acid, we can ignore the sm ...
... Example: Calculate the pH of a buffer system containing 1.0 M CH3CO2H and 1.0 M NaCH3CO2. What is the pH of the buffer after the addition of 0.10 moles of gaseous HCl to 1.00 liter of the buffer solution? The Ka for acetic acid is 1.8 x 10-5. Because acetic acid is a weak acid, we can ignore the sm ...
Chemical Nomenclature, Formulas, and Equations
... (b) There are two silicon atoms and six bromine atoms present, so the formula is Si2Br6. ...
... (b) There are two silicon atoms and six bromine atoms present, so the formula is Si2Br6. ...
Elements, Compounds and Mixtures Elements are the simplest type
... A solution is made when a solid (solute) is dissolved in a liquid (solvent). Dilute solution = lots of solvent : little solute. Concentrated solution = little solvent : lots of solute Saturated solution = no more solute can be dissolved in the solvent. A little more can be ...
... A solution is made when a solid (solute) is dissolved in a liquid (solvent). Dilute solution = lots of solvent : little solute. Concentrated solution = little solvent : lots of solute Saturated solution = no more solute can be dissolved in the solvent. A little more can be ...
Chemistry Project for School Students by iCBSE
... bases. A base is any substance that can neutralize an acid. The pH of a base is 7.1-14(above 7). All antacids have chemical in them called a buffer. When an antacid is mixed with an acid the buffer tries to even out the acidity and that is how stomach acid gets neutralized. In an antacid it is not t ...
... bases. A base is any substance that can neutralize an acid. The pH of a base is 7.1-14(above 7). All antacids have chemical in them called a buffer. When an antacid is mixed with an acid the buffer tries to even out the acidity and that is how stomach acid gets neutralized. In an antacid it is not t ...
Ion Exchange
... Ion-exchange chromatography retains analyte molecules on the column based on coulombic (ionic) interactions. The stationary phase surface displays ionic functional groups (R-X) that interact with analyte ions of opposite charge. An ion-exchange resin or ion-exchange polymer is an insoluble matrix no ...
... Ion-exchange chromatography retains analyte molecules on the column based on coulombic (ionic) interactions. The stationary phase surface displays ionic functional groups (R-X) that interact with analyte ions of opposite charge. An ion-exchange resin or ion-exchange polymer is an insoluble matrix no ...
LECTURE_Solutions2013(1)
... What happens when compounds dissolve? Dissolving Covalent Compounds • C12H22O11 (s) C12H22O11 (aq) • NO dissociation because NO ions • Sucrose dissolves in water because sugar is polar (-OH group), but dissociation does not occur. Sucrose molecules are simply separated from each other. No ions ar ...
... What happens when compounds dissolve? Dissolving Covalent Compounds • C12H22O11 (s) C12H22O11 (aq) • NO dissociation because NO ions • Sucrose dissolves in water because sugar is polar (-OH group), but dissociation does not occur. Sucrose molecules are simply separated from each other. No ions ar ...