in Peptide Synthesis, Molecular Recognition
... To date, chemists have proposed a number of Pro analogues for constraining an amide bond in a cis conformation [16-18]. For example, bulky substituents at C(5) of the cyclic Pro system result in cis-amide bond formation; however, the need for chemical synthesis of these Prosurrogates and their incor ...
... To date, chemists have proposed a number of Pro analogues for constraining an amide bond in a cis conformation [16-18]. For example, bulky substituents at C(5) of the cyclic Pro system result in cis-amide bond formation; however, the need for chemical synthesis of these Prosurrogates and their incor ...
Types of Reactions
... When a substance gains electrons, it undergoes reduction: 2 Ca (s) + O2 (g) 2 CaO (s). • In this reaction the neutral O2 has gained electrons from the Ca to become O–2 in CaO. • We say O2 has been reduced to O–2. In all redox reactions, one species is reduced at the same time as another is oxidize ...
... When a substance gains electrons, it undergoes reduction: 2 Ca (s) + O2 (g) 2 CaO (s). • In this reaction the neutral O2 has gained electrons from the Ca to become O–2 in CaO. • We say O2 has been reduced to O–2. In all redox reactions, one species is reduced at the same time as another is oxidize ...
1984 Advanced Placement Exam
... 24. The formula for potassium hexacyanoferrate (II) 28. 2 A(g) + B(g) 2 C(g) is When the concentration of substance B in the re(A) K4[Fe(CN)6] (D) K2[Pt(CN)6] action above is doubled, all other factors being held constant, it is found that the rate of the reac(B) K3[Fe(CN)6] (E) KCN tion remains ...
... 24. The formula for potassium hexacyanoferrate (II) 28. 2 A(g) + B(g) 2 C(g) is When the concentration of substance B in the re(A) K4[Fe(CN)6] (D) K2[Pt(CN)6] action above is doubled, all other factors being held constant, it is found that the rate of the reac(B) K3[Fe(CN)6] (E) KCN tion remains ...
chemistry
... 76 Which labeled part of the digestive system has the most acidic environment? [1] 77 What is the color of thymol blue at the pH of the small intestine? [1] 78 State how the catalyst lipase increases the rate of the fat digestion. [1] ...
... 76 Which labeled part of the digestive system has the most acidic environment? [1] 77 What is the color of thymol blue at the pH of the small intestine? [1] 78 State how the catalyst lipase increases the rate of the fat digestion. [1] ...
Calculations on the equations reaction
... valences this element can have in compounds? Write the formula of highest oxide of this element. 2. An element has serial number 19 define: а) charge of nucleus atom b) number of electrons c) number of neutrons and protons. Write electronic formula of element. What valences this element can have in ...
... valences this element can have in compounds? Write the formula of highest oxide of this element. 2. An element has serial number 19 define: а) charge of nucleus atom b) number of electrons c) number of neutrons and protons. Write electronic formula of element. What valences this element can have in ...
- Solubility products -Thermochemistry
... – Chlorides, bromides and iodides are soluble except those of Ag+, Pb2+, Hg22+. – Sulfates are soluble except those of Pb2+, Hg22+, Sr2+ and Ba2+. Ag2SO4 and CaSO4 are only slightly soluble. – Carbonates, phosphates and sulphites are insoluble (except those of group 1 and NH4+) – Sulfides (S2-) are ...
... – Chlorides, bromides and iodides are soluble except those of Ag+, Pb2+, Hg22+. – Sulfates are soluble except those of Pb2+, Hg22+, Sr2+ and Ba2+. Ag2SO4 and CaSO4 are only slightly soluble. – Carbonates, phosphates and sulphites are insoluble (except those of group 1 and NH4+) – Sulfides (S2-) are ...
www.XtremePapers.com
... When the plunger is pushed in the equilibrium adjusts to produce more PCl5(g). ...
... When the plunger is pushed in the equilibrium adjusts to produce more PCl5(g). ...
Acid-Base Reactions Worksheet #2 - Mro
... Plagiarizing a lab report will result in a zero for that lab report. 2. Cutting class will result in a zero for any test or lab report expected for that class day. In addition, I will require a note from your parents informing me that they are aware you've cut class and have received a zero for any ...
... Plagiarizing a lab report will result in a zero for that lab report. 2. Cutting class will result in a zero for any test or lab report expected for that class day. In addition, I will require a note from your parents informing me that they are aware you've cut class and have received a zero for any ...
chapter 16
... Thus, as the reaction begins, an input of energy is necessary to produce the activated complex; as the reaction proceeds, and the system shifts from the activated complex to products, energy is released. In a chemical reaction, the minimum energy necessary for reaching the activated complex and proc ...
... Thus, as the reaction begins, an input of energy is necessary to produce the activated complex; as the reaction proceeds, and the system shifts from the activated complex to products, energy is released. In a chemical reaction, the minimum energy necessary for reaching the activated complex and proc ...
BS Chemistry - Government College University Faisalabad
... Localized and delocalized bonding. Concept of hybridization leading to bond angles, bond energies and geometry of simple organic molecules; dipole moment; inductive effect; resonance, resonance energy, rules of resonance, resonance effect, steric inhibition of resonance; hypercohjugation; tautomeris ...
... Localized and delocalized bonding. Concept of hybridization leading to bond angles, bond energies and geometry of simple organic molecules; dipole moment; inductive effect; resonance, resonance energy, rules of resonance, resonance effect, steric inhibition of resonance; hypercohjugation; tautomeris ...
Using Models - Pleasant Valley School District
... this this number number equation equation following of ofatoms atoms tell tell us… or You The probably large numbers noticed in the front numbers of theus… molecules Two that the are sodium that Na chemical bonded is are in atoms required elemental together symbols will for react form. to are the ma ...
... this this number number equation equation following of ofatoms atoms tell tell us… or You The probably large numbers noticed in the front numbers of theus… molecules Two that the are sodium that Na chemical bonded is are in atoms required elemental together symbols will for react form. to are the ma ...
(+1) + - Edublogs
... shared but not equally. For electrons that are shared in these compounds, we assign the shared electrons to the most electronegative element. We are just acting as though the electronegativity difference was large enough for the transfer of electrons to occur. ...
... shared but not equally. For electrons that are shared in these compounds, we assign the shared electrons to the most electronegative element. We are just acting as though the electronegativity difference was large enough for the transfer of electrons to occur. ...
Chemical Thermodynamics presentation 1
... Chemistry and Thermodynamics • Physics Helps us understands chemistry (and biology etc.) ...
... Chemistry and Thermodynamics • Physics Helps us understands chemistry (and biology etc.) ...
Reactants Products
... – Powdered solids are more reactive than “blocks.” • More surface area for contact with other reactants – Certain types of chemicals are more reactive than others. • For example, potassium metal is more reactive than sodium – Ions react faster than molecules. • No bonds need to be broken. ...
... – Powdered solids are more reactive than “blocks.” • More surface area for contact with other reactants – Certain types of chemicals are more reactive than others. • For example, potassium metal is more reactive than sodium – Ions react faster than molecules. • No bonds need to be broken. ...
Section 3_Energetics
... hydroxide solution at the same temperature. The temperature of the calorimeter and contents rises by 7.0 oC. Calculate the standard enthalpy of neutralization. Assuming that the specific heat capacity of all the solution is 4200 J kg-1 K-1. ...
... hydroxide solution at the same temperature. The temperature of the calorimeter and contents rises by 7.0 oC. Calculate the standard enthalpy of neutralization. Assuming that the specific heat capacity of all the solution is 4200 J kg-1 K-1. ...
Proximity Effects on Reaction Rates
... to deprotonate water • The enhanced nucleophilicity of deprotonated water ...
... to deprotonate water • The enhanced nucleophilicity of deprotonated water ...
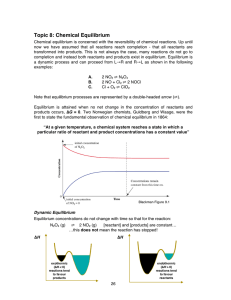
Topic 8: Chemical Equilibrium
... A catalyst lowers the activation energy, Ea for both forward and reverse reactions. The term ‘activation energy’ was first introduced in 1880 by a Swedish scientst, Svante Arrhenius, to describe the minimum amount of energy input required potential reactants to start a chemical reaction. As both for ...
... A catalyst lowers the activation energy, Ea for both forward and reverse reactions. The term ‘activation energy’ was first introduced in 1880 by a Swedish scientst, Svante Arrhenius, to describe the minimum amount of energy input required potential reactants to start a chemical reaction. As both for ...
Atomic Mass - HCC Learning Web
... 2. Write the unbalanced equation that summarizes the reaction described in Step 1. 3. Balance the equation by inspection, starting with the most complicated molecule(s). The same number of each type of atom needs to appear on both reactant and product sides. Do NOT change the formulas of any of the ...
... 2. Write the unbalanced equation that summarizes the reaction described in Step 1. 3. Balance the equation by inspection, starting with the most complicated molecule(s). The same number of each type of atom needs to appear on both reactant and product sides. Do NOT change the formulas of any of the ...
BOC-ON - Sigma
... This reagent offers a distinct advantage over t-BOC azide which can require reaction temperatures of 50-60°C (t-BOC azide is thermally unstable and decomposes with apparent detonation at temperatures above 80°C).1 The oxime by-product can be easily and completely removed from the reaction mixture by ...
... This reagent offers a distinct advantage over t-BOC azide which can require reaction temperatures of 50-60°C (t-BOC azide is thermally unstable and decomposes with apparent detonation at temperatures above 80°C).1 The oxime by-product can be easily and completely removed from the reaction mixture by ...