RES6_chem_stretch_challenge
... or benzene rings. In each case, you must name the functional group and the reagents for the reaction. You must also write the correct equations and products. Stretch and Challenge questions often allow you to demonstrate knowledge and understanding from different parts of the specification. Again, y ...
... or benzene rings. In each case, you must name the functional group and the reagents for the reaction. You must also write the correct equations and products. Stretch and Challenge questions often allow you to demonstrate knowledge and understanding from different parts of the specification. Again, y ...
First Poly(2-oxazoline)s with Pendant Amino Groups
... the polymer. In the simplest case, the length of an attached alkyl group determines the thermal properties and solubility.[13,14] In particular, with a methyl substitution, the polymer is highly water soluble; poly(2-ethyl-2-oxazoline) is the only amorphous polymer of this class,[14] which is still ...
... the polymer. In the simplest case, the length of an attached alkyl group determines the thermal properties and solubility.[13,14] In particular, with a methyl substitution, the polymer is highly water soluble; poly(2-ethyl-2-oxazoline) is the only amorphous polymer of this class,[14] which is still ...
8 theoretical problems 2 practical problems
... excess acidic phenylhydrazine. Compound C reacts with nitric acid to give an optically inactive compound D. The Kiliani-Fischer approach is used to establish the configurational relationship between D-glyceraldehyde and C. The intermediate aldotetrose which leads to C does not give a meso compound w ...
... excess acidic phenylhydrazine. Compound C reacts with nitric acid to give an optically inactive compound D. The Kiliani-Fischer approach is used to establish the configurational relationship between D-glyceraldehyde and C. The intermediate aldotetrose which leads to C does not give a meso compound w ...
Document
... Calorimetry is an accurate technique for determining enthalpy changes, but how do chemists deal with chemical systems that cannot be analyzed using this technique? For example, the rusting of iron (Figure 1) is extremely slow and, therefore, the resulting temperature change would be too small to be ...
... Calorimetry is an accurate technique for determining enthalpy changes, but how do chemists deal with chemical systems that cannot be analyzed using this technique? For example, the rusting of iron (Figure 1) is extremely slow and, therefore, the resulting temperature change would be too small to be ...
Stoichiometry/Mass/Mole Relationships
... 10. ___ C6H12 + ___ O2 → ___ CO2 + ___ H2O 42 grams of cyclohexane burns in excess air to from carbon dioxide and water. How many grams of carbon dioxide and of water vapor are produced? ...
... 10. ___ C6H12 + ___ O2 → ___ CO2 + ___ H2O 42 grams of cyclohexane burns in excess air to from carbon dioxide and water. How many grams of carbon dioxide and of water vapor are produced? ...
IOSR Journal of Applied Chemistry (IOSR-JAC)
... CNS stimulant and recent reports suggest that compounds containing piperidin-4-one moiety elicit excellent activity when aromatic substitutions are present at 2- and/or 6-positions. Mannich reaction is one of the multi-component reactions for the carbon-carbon and carbon heteroatom sequential bond f ...
... CNS stimulant and recent reports suggest that compounds containing piperidin-4-one moiety elicit excellent activity when aromatic substitutions are present at 2- and/or 6-positions. Mannich reaction is one of the multi-component reactions for the carbon-carbon and carbon heteroatom sequential bond f ...
Can Naturalistic Evolution Explain the Origin of Life on Earth
... of traces of ‘building blocks’ is commonly equated with proving that they could have built up the required complicated molecules under natural conditions. The instability of ‘building blocks’ in non-biotic environments is usually glossed over. The RNA/DNA base cytosine is not produced in spark disch ...
... of traces of ‘building blocks’ is commonly equated with proving that they could have built up the required complicated molecules under natural conditions. The instability of ‘building blocks’ in non-biotic environments is usually glossed over. The RNA/DNA base cytosine is not produced in spark disch ...
Chapter 4 Aqueous Reactions and Solution Stoichiometry
... • This reaction gives the predicted product, but you had better carry it out in the hood, or you will be very unpopular! • But just as in the previous examples, a gas is formed as a product of this reaction. Na2S (aq) + H2SO4 (aq) Na2SO4 (aq) + H2S (g) Aqueous Reactions © 2009, Prentice-Hall, Inc ...
... • This reaction gives the predicted product, but you had better carry it out in the hood, or you will be very unpopular! • But just as in the previous examples, a gas is formed as a product of this reaction. Na2S (aq) + H2SO4 (aq) Na2SO4 (aq) + H2S (g) Aqueous Reactions © 2009, Prentice-Hall, Inc ...
1. Given the balanced equation
... monoxide. Crushed limestone, CaCO3, is also added to the mixture to remove impurities in the ore. The carbon monoxide is formed by the oxidation of carbon(coke), as shown in the reaction below: 2 C + O2 - 2 CO + energy Liquid iron flows from the bottom of the blast furnace and is processed into diff ...
... monoxide. Crushed limestone, CaCO3, is also added to the mixture to remove impurities in the ore. The carbon monoxide is formed by the oxidation of carbon(coke), as shown in the reaction below: 2 C + O2 - 2 CO + energy Liquid iron flows from the bottom of the blast furnace and is processed into diff ...
CHEMICAL REACTIONS AND CHEMICAL EQUATIONS
... Stoichiometry is one of the most important topics in chemistry. It involves the use of the chemical formulas , mole calculations, and chemical equations. Stoichiometry is also essential in industry , there, it is used to do cost and analysis for manufacturing chemicals. In fact, manufacturing proces ...
... Stoichiometry is one of the most important topics in chemistry. It involves the use of the chemical formulas , mole calculations, and chemical equations. Stoichiometry is also essential in industry , there, it is used to do cost and analysis for manufacturing chemicals. In fact, manufacturing proces ...
chemical reaction
... • List three observations that suggest that a chemical reaction has taken place. • List three requirements for a correctly written chemical equation. • Write a word equation and a formula equation for a given chemical reaction. • Balance a formula equation by inspection. ...
... • List three observations that suggest that a chemical reaction has taken place. • List three requirements for a correctly written chemical equation. • Write a word equation and a formula equation for a given chemical reaction. • Balance a formula equation by inspection. ...
Enzymes - WordPress.com
... Enzymes Enzymes are important biological macromolecules that do work in all living things. Plants, animals, and prokaryotes all depend on enzymes to break down large molecules or build new ones. ENZYMES are proteins that act as catalysts and help chemical reactions occur. In order for these chemical ...
... Enzymes Enzymes are important biological macromolecules that do work in all living things. Plants, animals, and prokaryotes all depend on enzymes to break down large molecules or build new ones. ENZYMES are proteins that act as catalysts and help chemical reactions occur. In order for these chemical ...
chem - CBSE Guess
... 2Fe+3/2O2 +xH2O----------> Fe2O3.xH2O(Hydrated ferric oxide rust) Rancidity: The oily and fatty food oxidizes and give bad smell and test is called rancidity.Preventatioin:By adding antioxidant which slow down the process of oxidation.2. Vaccum packing,3Flusing N2 gas in chips packets.3.Refrigeratio ...
... 2Fe+3/2O2 +xH2O----------> Fe2O3.xH2O(Hydrated ferric oxide rust) Rancidity: The oily and fatty food oxidizes and give bad smell and test is called rancidity.Preventatioin:By adding antioxidant which slow down the process of oxidation.2. Vaccum packing,3Flusing N2 gas in chips packets.3.Refrigeratio ...
No Slide Title
... (b) This is a combination reaction (two reactants form a single product). The oxidation number of Li changes from 0 to +1 while that of N changes from 0 to −3. (c) This is a metal displacement reaction. The Ni metal replaces (reduces) the Pb2+ ion. The oxidation number of Ni increases from 0 to +2 w ...
... (b) This is a combination reaction (two reactants form a single product). The oxidation number of Li changes from 0 to +1 while that of N changes from 0 to −3. (c) This is a metal displacement reaction. The Ni metal replaces (reduces) the Pb2+ ion. The oxidation number of Ni increases from 0 to +2 w ...
thermodynamics
... gases into each other in a closed container is a spontaneous process or not? 28. Heat has randomising influence on a system and temperature is the measure of average chaotic motion of particles in the system. Write the mathematical relation which relates these three parameters. 29. Increase in entha ...
... gases into each other in a closed container is a spontaneous process or not? 28. Heat has randomising influence on a system and temperature is the measure of average chaotic motion of particles in the system. Write the mathematical relation which relates these three parameters. 29. Increase in entha ...
Thermochemistry Exam Review Questions
... 11. A student found that orange IV indicator turned yellow and methyl orange turned red in samples of an unknown solution. What is the pH for the unknown solution likely to be? A. 1.2 B. 3.0 C. 5.3 D. 9.0 12. What is the name of the ion when a positively charged proton combines with a water molecul ...
... 11. A student found that orange IV indicator turned yellow and methyl orange turned red in samples of an unknown solution. What is the pH for the unknown solution likely to be? A. 1.2 B. 3.0 C. 5.3 D. 9.0 12. What is the name of the ion when a positively charged proton combines with a water molecul ...
Physical chemistry and transition elements 5.1 Rates, equilibrium
... The copper half cell would have been set up so that copper metal was in contact with its ions: a strip of copper would have been placed in a solution such as CuSO4(aq), at a concentration of 1 mol dm−3 and a temperature of 298 K. The copper metal would have been the electrode and would have been con ...
... The copper half cell would have been set up so that copper metal was in contact with its ions: a strip of copper would have been placed in a solution such as CuSO4(aq), at a concentration of 1 mol dm−3 and a temperature of 298 K. The copper metal would have been the electrode and would have been con ...
Inorganometallic Chemistry
... TM complexes containing borylene ligands (BR) can adopt two different coordination modes: either terminal (BR group terminally bonded to one metal atom and boron assuming a coordination number 2) or bridging (borylene group adopting bridging position between two metal centers with a threefold-coordi ...
... TM complexes containing borylene ligands (BR) can adopt two different coordination modes: either terminal (BR group terminally bonded to one metal atom and boron assuming a coordination number 2) or bridging (borylene group adopting bridging position between two metal centers with a threefold-coordi ...
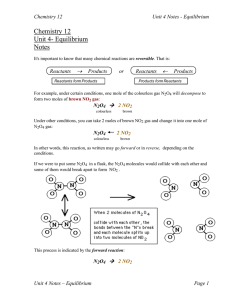
unit-4-notes-1_enthalpy-and-entropy
... and no NO2. The forward reaction rate was high at the start, but the reverse reaction rate eventually "caught up", the rates became equal and equilibrium was established. Can you guess what would happen if we had started with pure NO2 instead (no N2O4 )? The reverse rate would start out high and the ...
... and no NO2. The forward reaction rate was high at the start, but the reverse reaction rate eventually "caught up", the rates became equal and equilibrium was established. Can you guess what would happen if we had started with pure NO2 instead (no N2O4 )? The reverse rate would start out high and the ...
Stoichiometric Problems III: Sto c o et c ob e s
... In a limiting reactant problem you are given either g gram or mole amounts of starting g reactants, and asked to determine which reactant runs out first, and ‘limits’ the amount of product that can be made made. I have found that the method that the book uses for f these th problems bl tends t d to ...
... In a limiting reactant problem you are given either g gram or mole amounts of starting g reactants, and asked to determine which reactant runs out first, and ‘limits’ the amount of product that can be made made. I have found that the method that the book uses for f these th problems bl tends t d to ...