Document
... WHY ARE THERE CHEMICAL REACTIONS? CHEMICAL REACTIONS HAPPEN WHEN MOLECULES BUMP INTO EACH OTHER CAUSING THE STARTING BONDS TO BREAK APART, THE ATOMS REARRANGE, AND NEW BONDS ARE FORMED ...
... WHY ARE THERE CHEMICAL REACTIONS? CHEMICAL REACTIONS HAPPEN WHEN MOLECULES BUMP INTO EACH OTHER CAUSING THE STARTING BONDS TO BREAK APART, THE ATOMS REARRANGE, AND NEW BONDS ARE FORMED ...
Bacteria and Virus Research Jigsaw
... WHY ARE THERE CHEMICAL REACTIONS? CHEMICAL REACTIONS HAPPEN WHEN MOLECULES BUMP INTO EACH OTHER CAUSING THE STARTING BONDS TO BREAK APART, THE ATOMS REARRANGE, AND NEW BONDS ARE FORMED ...
... WHY ARE THERE CHEMICAL REACTIONS? CHEMICAL REACTIONS HAPPEN WHEN MOLECULES BUMP INTO EACH OTHER CAUSING THE STARTING BONDS TO BREAK APART, THE ATOMS REARRANGE, AND NEW BONDS ARE FORMED ...
6.5 Main Group
... From antiquity up to ca.1910, “lead white” was used as a white pigment (lead white = PbCO3 · x Pb(OH)2 ). Rembrandt used lead white in his paintings. The typical dark brown appearance of the old Dutch masters is an artifact of the formation of brown lead sulfide PbS: ...
... From antiquity up to ca.1910, “lead white” was used as a white pigment (lead white = PbCO3 · x Pb(OH)2 ). Rembrandt used lead white in his paintings. The typical dark brown appearance of the old Dutch masters is an artifact of the formation of brown lead sulfide PbS: ...
Describing Chemical Reactions
... The principle called conservation of mass was first demonstrated in the late 1700s. The principle of conservation of mass states that in a chemical reaction, the total mass of the reactants must equal the total mass of the products. In an open system, matter can enter from or escape to the surroundi ...
... The principle called conservation of mass was first demonstrated in the late 1700s. The principle of conservation of mass states that in a chemical reaction, the total mass of the reactants must equal the total mass of the products. In an open system, matter can enter from or escape to the surroundi ...
Chapter 12 Packet
... Write and/or balance the following equations (remember the diatomic elements and to criss-cross charges for ionic compounds!!!) Use the mole ratios from the balanced equations to solve the following stoichiometry problems. Use units and labels in all conversions, and round your answer to sig figs. 1 ...
... Write and/or balance the following equations (remember the diatomic elements and to criss-cross charges for ionic compounds!!!) Use the mole ratios from the balanced equations to solve the following stoichiometry problems. Use units and labels in all conversions, and round your answer to sig figs. 1 ...
document
... Short-hand way of describing a reaction. Provides information about the reaction. Formulas of reactants and products. States of reactants and products. Relative numbers of reactant and product molecules that are required. Can be used to determine masses of reactants used and products tha ...
... Short-hand way of describing a reaction. Provides information about the reaction. Formulas of reactants and products. States of reactants and products. Relative numbers of reactant and product molecules that are required. Can be used to determine masses of reactants used and products tha ...
Fundamental Knowledge for Analysis of Chemical Reactor
... equivolumetric mixture of P.O. and methanol 2.5 times volume of 0.1%H2SO4 water solution uniform concentration and temperature inside whole reactor products is continuously discharged from reactor with the same concentration and temperature as inside reactor what we want to know: production ability, ...
... equivolumetric mixture of P.O. and methanol 2.5 times volume of 0.1%H2SO4 water solution uniform concentration and temperature inside whole reactor products is continuously discharged from reactor with the same concentration and temperature as inside reactor what we want to know: production ability, ...
Module 3 -- Lesson 4
... replace the lost product. Consider the example of an organic acid reacting with an alcohol to produce an ester and water. An ester is a compound with a pleasant odor that can be synthesized in the laboratory by reacting an alcohol and an organic acid (chemistry 30S students do this as part of their ...
... replace the lost product. Consider the example of an organic acid reacting with an alcohol to produce an ester and water. An ester is a compound with a pleasant odor that can be synthesized in the laboratory by reacting an alcohol and an organic acid (chemistry 30S students do this as part of their ...
Discussion 9, Mahaffy et al., Chapter 15
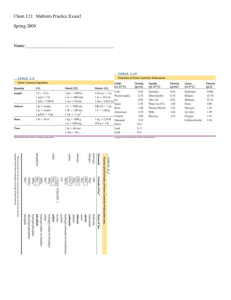
... b. With regards to CaF2, circle the correct choice Precipitation will occur Precipitation will not occur More information needed 5. The number of moles of a solid that dissolves in 1 liter of water is called the molar solubility. For Ag2SO4, the molar solubility is 1.4∙10-2 M. What is the Ksp ...
... b. With regards to CaF2, circle the correct choice Precipitation will occur Precipitation will not occur More information needed 5. The number of moles of a solid that dissolves in 1 liter of water is called the molar solubility. For Ag2SO4, the molar solubility is 1.4∙10-2 M. What is the Ksp ...
chem10chp7spr08
... in water; they are called aqueous solutions Dissolving chemicals in water helps them to react together faster - water separates chemicals into individual molecules or ions, called dissociation - the particles come in contact more frequently speeding the reaction up ...
... in water; they are called aqueous solutions Dissolving chemicals in water helps them to react together faster - water separates chemicals into individual molecules or ions, called dissociation - the particles come in contact more frequently speeding the reaction up ...
practice test2(Answers)
... A) The temperature of steam cannot exceed 100°C. B) The temperature of ice remains at 0°C as it melts. C) The temperature of liquid water increases linearly as it is heated D) The temperature of liquid water remains at 100°C as it boils E) Both liquid water and ice are present at 0°C. ...
... A) The temperature of steam cannot exceed 100°C. B) The temperature of ice remains at 0°C as it melts. C) The temperature of liquid water increases linearly as it is heated D) The temperature of liquid water remains at 100°C as it boils E) Both liquid water and ice are present at 0°C. ...
Chemistry Final - Practice Test I
... What was the contribution to chemistry by each of these individuals? Neils Bohr Developed the Planetary Model of the atom based on Quantum energy levels Henry Moseley Arranged the Periodic Table – Increasing atomic number using x-rays and wavelengths Rutherford Discovered that most of the atoms mass ...
... What was the contribution to chemistry by each of these individuals? Neils Bohr Developed the Planetary Model of the atom based on Quantum energy levels Henry Moseley Arranged the Periodic Table – Increasing atomic number using x-rays and wavelengths Rutherford Discovered that most of the atoms mass ...
11 BALANCING CHEMICAL EQUATIONS 1. 2 K + 1
... The large numbers in front of chemical formulas. Coefficients represent the number of molecules of the substance in the reaction. ...
... The large numbers in front of chemical formulas. Coefficients represent the number of molecules of the substance in the reaction. ...
Revised Syllabus - M. Sc. First Year - Chemistry
... a) Types of Aliphatic nucleophilic substitution Reactions-SN1, SN2, SET mechanism, b) NGP by pi and sigma bonds, classical and non-classical carbocations, phenonium ions, norbornyl system, carbocation rearrangement in NGP, SNi mechanism, c) effect of structure, nucleophile, leaving group, solvent on ...
... a) Types of Aliphatic nucleophilic substitution Reactions-SN1, SN2, SET mechanism, b) NGP by pi and sigma bonds, classical and non-classical carbocations, phenonium ions, norbornyl system, carbocation rearrangement in NGP, SNi mechanism, c) effect of structure, nucleophile, leaving group, solvent on ...
4. chemical kinetics
... If concentration is higher, the number of molecules per unit volume is, more and the number of active collisions increases. According to collision theory, rate of reaction increases with increase in the number of collisions. ...
... If concentration is higher, the number of molecules per unit volume is, more and the number of active collisions increases. According to collision theory, rate of reaction increases with increase in the number of collisions. ...
Exam 1 Review
... 7. The place on a phase diagram where solid and liquid are in equilibrium is called a. the triple point b. the normal melting point c. the solid-liquid boundary d. the supercritical fluid 8. A chemical reaction taking place at 24 atm of pressure results in the volume of the system decreasing from 14 ...
... 7. The place on a phase diagram where solid and liquid are in equilibrium is called a. the triple point b. the normal melting point c. the solid-liquid boundary d. the supercritical fluid 8. A chemical reaction taking place at 24 atm of pressure results in the volume of the system decreasing from 14 ...
Bonding 1. Which one of the following is most likely to be an ionic
... b. In [A] plotted against time gives a straight line of negative slope. c. 1/[A] plotted against time gives a straight line of positive slope. d. [A] plotted against time gives a straight line of negative slope. e. [A] plotted against time gives a curved line of negative slope, decreasing in magnitu ...
... b. In [A] plotted against time gives a straight line of negative slope. c. 1/[A] plotted against time gives a straight line of positive slope. d. [A] plotted against time gives a straight line of negative slope. e. [A] plotted against time gives a curved line of negative slope, decreasing in magnitu ...
Conservation of Mass Lab
... during a chemical reaction. This means that all chemical reactions must be balanced—the number of atoms, moles, and ultimately the total mass must be conserved during a chemical process. Here are the rules to follow when balancing equations: ...
... during a chemical reaction. This means that all chemical reactions must be balanced—the number of atoms, moles, and ultimately the total mass must be conserved during a chemical process. Here are the rules to follow when balancing equations: ...
unit 4 practice
... 2. Equal volumes and concentrations of hydrochloric acid and ethanoic acid are titrated with sodium hydroxide solutions of the same concentration. Which statement is correct? A. The initial pH values of ...
... 2. Equal volumes and concentrations of hydrochloric acid and ethanoic acid are titrated with sodium hydroxide solutions of the same concentration. Which statement is correct? A. The initial pH values of ...
Pb2+ +2I- → PbI2 (s)
... What is happened to the molecule when you are changing the state of matter? Breaking intermolecular attractions, NOT the bonds. ...
... What is happened to the molecule when you are changing the state of matter? Breaking intermolecular attractions, NOT the bonds. ...