Modelling the Role of Charge in Atmospheric Particle
... was published in 2013 (IPCC, 2013) and Work Group II report was published in 2014 (IPCC, 2014), it was stated that the global mean surface temperatures for the period 2081-2100 may rise 0.3-4.8 ◦ C relative to 1986-2005, depending on the emissions. This does not mean a similar rise in temperature ev ...
... was published in 2013 (IPCC, 2013) and Work Group II report was published in 2014 (IPCC, 2014), it was stated that the global mean surface temperatures for the period 2081-2100 may rise 0.3-4.8 ◦ C relative to 1986-2005, depending on the emissions. This does not mean a similar rise in temperature ev ...
Chapter 8: Ionic Compounds
... of chemical bonds. Table 8-1 shows several examples of electron-dot structures. For example, carbon has an electron configuration of 1s22s22p2. Its valence electrons are those in the second energy level, as can be seen in the electron-dot structure for carbon in the table. Recall from Chapter 6 that ...
... of chemical bonds. Table 8-1 shows several examples of electron-dot structures. For example, carbon has an electron configuration of 1s22s22p2. Its valence electrons are those in the second energy level, as can be seen in the electron-dot structure for carbon in the table. Recall from Chapter 6 that ...
Longitudinal and Transverse Zeeman Ladders in the
... the case of Ising spins, initially described by Shiba [7], is the quantization of the transverse excitation continuum in a series of discrete lines below the Néel temperature (T N ). This effect, called Zeeman ladder, was proposed to explain the discretization of the excitations observed in the orde ...
... the case of Ising spins, initially described by Shiba [7], is the quantization of the transverse excitation continuum in a series of discrete lines below the Néel temperature (T N ). This effect, called Zeeman ladder, was proposed to explain the discretization of the excitations observed in the orde ...
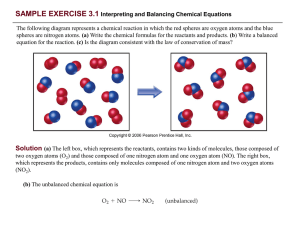
PRACTICE EXERCISE - Needham.K12.ma.us
... (c) The left box (reactants) contains four O2 molecules and eight NO molecules. Thus, the molecular ratio is one O2 for each two NO as required by the balanced equation. The right box (products) contains eight NO 2 molecules. The number of NO2 molecules on the right equals the number of NO molecules ...
... (c) The left box (reactants) contains four O2 molecules and eight NO molecules. Thus, the molecular ratio is one O2 for each two NO as required by the balanced equation. The right box (products) contains eight NO 2 molecules. The number of NO2 molecules on the right equals the number of NO molecules ...
June Exam Review Material World
... A) The element is located in period 1 and is an alkaline earth metal. B) The element is located in period 1 and is an alkali metal. C) The element is located in period 2 and is an alkali metal. D) The element is located in period 2 and is an alkaline earth metal. 15. Four elements from the periodic ...
... A) The element is located in period 1 and is an alkaline earth metal. B) The element is located in period 1 and is an alkali metal. C) The element is located in period 2 and is an alkali metal. D) The element is located in period 2 and is an alkaline earth metal. 15. Four elements from the periodic ...
International Journal of Molecular Sciences
... third-generation devices. Most of the devices on the market now are first and second generation cells, which are based on crystalline silicon and CdTe thin film, respectively. High purity requirements for the silicon crystals, high fabrication temperatures and the large amount of material which is n ...
... third-generation devices. Most of the devices on the market now are first and second generation cells, which are based on crystalline silicon and CdTe thin film, respectively. High purity requirements for the silicon crystals, high fabrication temperatures and the large amount of material which is n ...
Low-frequency conductivity of a nondegenerate two-dimensional electron liquid
... The question of observing the actual dynamics or electron scattering in the electron liquid has not been addressed previously. From analogy with single-electron scattering one may expect that an insight into this dynamics can be gained from the frequency dependence of xx ( ). Here, we develop an ...
... The question of observing the actual dynamics or electron scattering in the electron liquid has not been addressed previously. From analogy with single-electron scattering one may expect that an insight into this dynamics can be gained from the frequency dependence of xx ( ). Here, we develop an ...
Magnetic excitations of stripes and checkerboards in the cuprates
... and spin modulations. Since the neutron scattering shows satellite peaks around antiferromagnetism, rather than a peak at the antiferromagnetic wave vector !! , !", any universal spin texture which is consistent with the proposed checkerboard pattern must also incorporate antiphase domain walls in t ...
... and spin modulations. Since the neutron scattering shows satellite peaks around antiferromagnetism, rather than a peak at the antiferromagnetic wave vector !! , !", any universal spin texture which is consistent with the proposed checkerboard pattern must also incorporate antiphase domain walls in t ...
Chapter 2 Elements and Compounds 2.1 The Structure of the Atom
... 2.1b Atomic Number, Mass Number, and Atomic Symbols Atoms of each element can be distinguished by the number of protons in the nucleus. The atomic number (Z) of an element is equal to the number of protons in the nucleus. For example, a carbon atom has six protons in its nucleus, and therefore carbo ...
... 2.1b Atomic Number, Mass Number, and Atomic Symbols Atoms of each element can be distinguished by the number of protons in the nucleus. The atomic number (Z) of an element is equal to the number of protons in the nucleus. For example, a carbon atom has six protons in its nucleus, and therefore carbo ...
Model for ultrashort laser pulse-induced ionization dynamics in
... [10,12]. These models of free-carrier dynamics to date are also monochromatic. In most pulse propagation simulations, the time-evolved carrier density, calculated from a rate-equation model [9], is exported to a Drude model for the free current density that assumes averaged values for material const ...
... [10,12]. These models of free-carrier dynamics to date are also monochromatic. In most pulse propagation simulations, the time-evolved carrier density, calculated from a rate-equation model [9], is exported to a Drude model for the free current density that assumes averaged values for material const ...
Sructural and chemisorption properties of metallic surfaces and metallic overlayers
... Al(110) and Ni on W(110)) and the hydrogen interaction with two clean metal surfaces (Nb(110) and W(110)) have been studied using various surface analytical techniques. The results for the Pd/Al system show that the Pd film grows on Al(110) in a layer-by-layer mode. The binding energies of the Pd 4d ...
... Al(110) and Ni on W(110)) and the hydrogen interaction with two clean metal surfaces (Nb(110) and W(110)) have been studied using various surface analytical techniques. The results for the Pd/Al system show that the Pd film grows on Al(110) in a layer-by-layer mode. The binding energies of the Pd 4d ...
Honors Chemistry Curr
... Differentiated Learning Activities - Using IBooks, reference books and texts, students will work in lab groups (2 or 3 students per group) to complete a “Periodic Table Scavenger Hunt” where they will use clues given to identify unknown elements and place them in their appropriate location on the pe ...
... Differentiated Learning Activities - Using IBooks, reference books and texts, students will work in lab groups (2 or 3 students per group) to complete a “Periodic Table Scavenger Hunt” where they will use clues given to identify unknown elements and place them in their appropriate location on the pe ...
Honors Chemistry
... Differentiated Learning Activities - Using IBooks, reference books and texts, students will work in lab groups (2 or 3 students per group) to complete a “Periodic Table Scavenger Hunt” where they will use clues given to identify unknown elements and place them in their appropriate location on the pe ...
... Differentiated Learning Activities - Using IBooks, reference books and texts, students will work in lab groups (2 or 3 students per group) to complete a “Periodic Table Scavenger Hunt” where they will use clues given to identify unknown elements and place them in their appropriate location on the pe ...
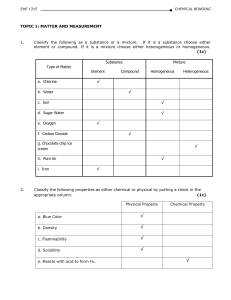
DEPARTMENT OF CHEMISTRY, CFS, IIUM
... variety of matter is recognized is called a property. A characteristic that depends upon the amount of matter in the sample is called an extensive property. A characteristic that does not depend upon the amount of matter is called an intensive property. A characteristic that can be observed without ...
... variety of matter is recognized is called a property. A characteristic that depends upon the amount of matter in the sample is called an extensive property. A characteristic that does not depend upon the amount of matter is called an intensive property. A characteristic that can be observed without ...
From a few to many electrons in quantum dots under strong
... long conjectured16–18 for solid 4He under appropriate conditions. The recent experimental discovery19 that solid 4He exhibits a nonclassical 共nonrigid兲 rotational inertia 共NCRI18兲 has revived an intense interest20–24 in the existence and properties of the supersolid phase in this system, as well as ...
... long conjectured16–18 for solid 4He under appropriate conditions. The recent experimental discovery19 that solid 4He exhibits a nonclassical 共nonrigid兲 rotational inertia 共NCRI18兲 has revived an intense interest20–24 in the existence and properties of the supersolid phase in this system, as well as ...
X-ray photoelectron spectroscopy

X-ray photoelectron spectroscopy (XPS) is a surface-sensitive quantitative spectroscopic technique that measures the elemental composition at the parts per thousand range, empirical formula, chemical state and electronic state of the elements that exist within a material. XPS spectra are obtained by irradiating a material with a beam of X-rays while simultaneously measuring the kinetic energy and number of electrons that escape from the top 0 to 10 nm of the material being analyzed. XPS requires high vacuum (P ~ 10−8 millibar) or ultra-high vacuum (UHV; P < 10−9 millibar) conditions, although a current area of development is ambient-pressure XPS, in which samples are analyzed at pressures of a few tens of millibar.XPS is a surface chemical analysis technique that can be used to analyze the surface chemistry of a material in its as-received state, or after some treatment, for example: fracturing, cutting or scraping in air or UHV to expose the bulk chemistry, ion beam etching to clean off some or all of the surface contamination (with mild ion etching) or to intentionally expose deeper layers of the sample (with more extensive ion etching) in depth-profiling XPS, exposure to heat to study the changes due to heating, exposure to reactive gases or solutions, exposure to ion beam implant, exposure to ultraviolet light.XPS is also known as ESCA (Electron Spectroscopy for Chemical Analysis), an abbreviation introduced by Kai Siegbahn's research group to emphasize the chemical (rather than merely elemental) information that the technique provides.In principle XPS detects all elements. In practice, using typical laboratory-scale X-ray sources, XPS detects all elements with an atomic number (Z) of 3 (lithium) and above. It cannot easily detect hydrogen (Z = 1) or helium (Z = 2).Detection limits for most of the elements (on a modern instrument) are in the parts per thousand range. Detection limits of parts per million (ppm) are possible, but require special conditions: concentration at top surface or very long collection time (overnight).XPS is routinely used to analyze inorganic compounds, metal alloys, semiconductors, polymers, elements, catalysts, glasses, ceramics, paints, papers, inks, woods, plant parts, make-up, teeth, bones, medical implants, bio-materials, viscous oils, glues, ion-modified materials and many others.XPS is less routinely used to analyze the hydrated forms of some of the above materials by freezing the samples in their hydrated state in an ultra pure environment, and allowing or causing multilayers of ice to sublime away prior to analysis. Such hydrated XPS analysis allows hydrated sample structures, which may be different from vacuum-dehydrated sample structures, to be studied in their more relevant as-used hydrated structure. Many bio-materials such as hydrogels are examples of such samples.