Raman Spectroscopy: Introductory Tutorial
... Infrared and Raman Spectra of Inorganic and Coordination Compounds : Theory and Applications in Inorganic Chemistry (Volume A) by Kazuo Nakamoto Infrared and Raman Spectra of Inorganic and Coordination Compounds : Applications in Coordination, Organometallic, and Bioinorganic Chemistry (Volume B) by ...
... Infrared and Raman Spectra of Inorganic and Coordination Compounds : Theory and Applications in Inorganic Chemistry (Volume A) by Kazuo Nakamoto Infrared and Raman Spectra of Inorganic and Coordination Compounds : Applications in Coordination, Organometallic, and Bioinorganic Chemistry (Volume B) by ...
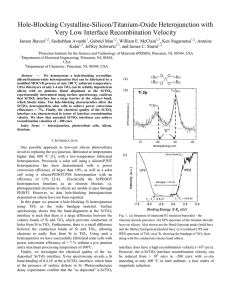
J. Jhaveri, S. Avasthi, G. Man, W. McClain, K. Nagamatsu, J. Schwartz, A. Kahn, J.C. Sturm, "Hole-blocking Crystalline Silicon/Titanium-Oxide Heterojunction with Very Low Interface Recombination Velocity", Proc. IEEE Photovoltaic Spec. Conf. (PVSC), Paper 912, Tampa, FL (JUN 2013).
... All spectroscopic analysis was done in a separate ultra-high vacuum (UHV) chamber (base pressure < 2 × 10− 10 mbar), details of which have been have been published elsewhere [67]. During transport to the UHV chamber, test samples were exposed to air for no more than 10 min. Recombination lifetimes w ...
... All spectroscopic analysis was done in a separate ultra-high vacuum (UHV) chamber (base pressure < 2 × 10− 10 mbar), details of which have been have been published elsewhere [67]. During transport to the UHV chamber, test samples were exposed to air for no more than 10 min. Recombination lifetimes w ...
support material
... such as minerals. For example, common salt, marble and limestone. Organiccompounds are those, which occur in living sources such as plants and animals. They all contain carbon. Commonorganic compounds are oils, wax, fats etc. Mixtures A mixture is a combination of two or more elements or compounds i ...
... such as minerals. For example, common salt, marble and limestone. Organiccompounds are those, which occur in living sources such as plants and animals. They all contain carbon. Commonorganic compounds are oils, wax, fats etc. Mixtures A mixture is a combination of two or more elements or compounds i ...
Barrier-free intermolecular proton transfer induced by excess
... EBE⫽1.6 –2.1 eV. The photoelectron spectrum of UA⫺ cannot be attributed to an intact U⫺ solvated by alanine. As mentioned earlier, the valence * and dipole-bound anionic states of uracil are characterized by a calculated value of VDE of 0.507 and 0.073 eV, respectively16 共see Fig. 3 for the excess ...
... EBE⫽1.6 –2.1 eV. The photoelectron spectrum of UA⫺ cannot be attributed to an intact U⫺ solvated by alanine. As mentioned earlier, the valence * and dipole-bound anionic states of uracil are characterized by a calculated value of VDE of 0.507 and 0.073 eV, respectively16 共see Fig. 3 for the excess ...
Wafer-Level Artificial Photosynthesis for CO2 Reduction into CH4
... performed. Full XPS spectra of a fresh sample of as-grown GaN nanowires and samples after CO2 reduction experiments show no significant differences. Shown in Figure S5 are the Ga2p and N1s peaks for the fresh sample and a sample after CO2 reduction experiments. The position of the Ga2p peak is ~1118 ...
... performed. Full XPS spectra of a fresh sample of as-grown GaN nanowires and samples after CO2 reduction experiments show no significant differences. Shown in Figure S5 are the Ga2p and N1s peaks for the fresh sample and a sample after CO2 reduction experiments. The position of the Ga2p peak is ~1118 ...
Quantitative methods for electron energy loss spectroscopy Alberto Eljarrat Ascunce
... optics to be configured, e.g. the scanning TEM (STEM) mode to raster a very small probe along given areas in order to form a pixel-by-pixel map. There are also many complementary characterization techniques that can be performed using specific detectors attached to the TEM column. One of these techn ...
... optics to be configured, e.g. the scanning TEM (STEM) mode to raster a very small probe along given areas in order to form a pixel-by-pixel map. There are also many complementary characterization techniques that can be performed using specific detectors attached to the TEM column. One of these techn ...
Nonradiative electron and energy transfer. Explicit
... forms explicitly consist of a weighted sum of delta functions in the frequency domain. Practically, on the other hand, they are alternatively replaced by phenomenological expressions, like Debye’s, Ohmic spectral functions and etc. Physically, it is argued that a vibrational manifold behaves like a ...
... forms explicitly consist of a weighted sum of delta functions in the frequency domain. Practically, on the other hand, they are alternatively replaced by phenomenological expressions, like Debye’s, Ohmic spectral functions and etc. Physically, it is argued that a vibrational manifold behaves like a ...
Elucidation of population and coherence dynamics using cross
... Theoretically, simulations based on exciton models are often used to iteratively fit 2D electronic spectra and provide a model description for the dynamics and electronic couplings in the system [7,9,13]. Because of the complexity of an multichromophoric systems in the condensed phase, these simulati ...
... Theoretically, simulations based on exciton models are often used to iteratively fit 2D electronic spectra and provide a model description for the dynamics and electronic couplings in the system [7,9,13]. Because of the complexity of an multichromophoric systems in the condensed phase, these simulati ...
1 - KFUPM Faculty List
... 15. Which of the following is a strong electrolyte solution? A) KMnO4(aq) KMnO4 is a normal salt, and thus this is a strong electrolyte solution B) C2H5OH(aq) This is a dissolved polar molecule (an alcohol) and thus it is a non-electrolyte solution C) NH3(aq) This is a weak base solution and thus a ...
... 15. Which of the following is a strong electrolyte solution? A) KMnO4(aq) KMnO4 is a normal salt, and thus this is a strong electrolyte solution B) C2H5OH(aq) This is a dissolved polar molecule (an alcohol) and thus it is a non-electrolyte solution C) NH3(aq) This is a weak base solution and thus a ...
Results
... Now , in 2015, a much more massive ice cloud system has now been found at an altitude of about 200 kilometers. The ice particles are made up of a variety of compounds containing hydrogen, carbon and nitrogen. ...
... Now , in 2015, a much more massive ice cloud system has now been found at an altitude of about 200 kilometers. The ice particles are made up of a variety of compounds containing hydrogen, carbon and nitrogen. ...
Physics of Projected Wavefunctions
... The frustration can arise from two sources: it can be a lattice effect; i.e., a fee or triangular lattice with only n.n. interaction is frustrated, or it can be a consequence of competing interaction between, e.g., n.n. and next-nearest-neighbor (n.n.n.) interactions. In this case, the Hamiltonian ( ...
... The frustration can arise from two sources: it can be a lattice effect; i.e., a fee or triangular lattice with only n.n. interaction is frustrated, or it can be a consequence of competing interaction between, e.g., n.n. and next-nearest-neighbor (n.n.n.) interactions. In this case, the Hamiltonian ( ...
Stoichiometry: Calculations with Chemical Formulas and
... Begin by counting each kind of atom on the two sides of the arrow. There are one Na, one O, and two H on the left side, and one Na, one O, and three H on the right. To increase the number of H atoms on the left, let’s try placing the coefficient 2 in front of H2O: Although beginning this way does no ...
... Begin by counting each kind of atom on the two sides of the arrow. There are one Na, one O, and two H on the left side, and one Na, one O, and three H on the right. To increase the number of H atoms on the left, let’s try placing the coefficient 2 in front of H2O: Although beginning this way does no ...
ENERGIES OF RARE-EARTH ION STATES RELATIVE TO HOST
... 4.7 CIS spectrum of the combined 4f electron and valence band photoemission for YbAG in the region of the 4d to 4f giant resonance ........189 4.8 CIS spectra of the combined 4f electron and valence band photoemission for EuGG and GdAG in the region of the 4d to 4f giant ...
... 4.7 CIS spectrum of the combined 4f electron and valence band photoemission for YbAG in the region of the 4d to 4f giant resonance ........189 4.8 CIS spectra of the combined 4f electron and valence band photoemission for EuGG and GdAG in the region of the 4d to 4f giant ...
Many-body van der Waals interactions in molecules and condensed
... dispersion interactions in DFT [14–23]. Despite such significant progress in the field of modeling vdW interactions, many questions still remain and further development is required before a universally applicable method emerges. For instance, pairwise interatomic vdW methods are frequently employed ...
... dispersion interactions in DFT [14–23]. Despite such significant progress in the field of modeling vdW interactions, many questions still remain and further development is required before a universally applicable method emerges. For instance, pairwise interatomic vdW methods are frequently employed ...
Schroedinger`s Model of Hydrogen Atom
... The hydrogen atom is the simplest atom. It has only one electron and the nucleus is a proton. It is therefore not surprising that it has been the test-bed for new theories. In this module, we will look at the attempts that have been made to understand the structure of the hydrogen atom1—1a structure ...
... The hydrogen atom is the simplest atom. It has only one electron and the nucleus is a proton. It is therefore not surprising that it has been the test-bed for new theories. In this module, we will look at the attempts that have been made to understand the structure of the hydrogen atom1—1a structure ...
X-ray photoelectron spectroscopy

X-ray photoelectron spectroscopy (XPS) is a surface-sensitive quantitative spectroscopic technique that measures the elemental composition at the parts per thousand range, empirical formula, chemical state and electronic state of the elements that exist within a material. XPS spectra are obtained by irradiating a material with a beam of X-rays while simultaneously measuring the kinetic energy and number of electrons that escape from the top 0 to 10 nm of the material being analyzed. XPS requires high vacuum (P ~ 10−8 millibar) or ultra-high vacuum (UHV; P < 10−9 millibar) conditions, although a current area of development is ambient-pressure XPS, in which samples are analyzed at pressures of a few tens of millibar.XPS is a surface chemical analysis technique that can be used to analyze the surface chemistry of a material in its as-received state, or after some treatment, for example: fracturing, cutting or scraping in air or UHV to expose the bulk chemistry, ion beam etching to clean off some or all of the surface contamination (with mild ion etching) or to intentionally expose deeper layers of the sample (with more extensive ion etching) in depth-profiling XPS, exposure to heat to study the changes due to heating, exposure to reactive gases or solutions, exposure to ion beam implant, exposure to ultraviolet light.XPS is also known as ESCA (Electron Spectroscopy for Chemical Analysis), an abbreviation introduced by Kai Siegbahn's research group to emphasize the chemical (rather than merely elemental) information that the technique provides.In principle XPS detects all elements. In practice, using typical laboratory-scale X-ray sources, XPS detects all elements with an atomic number (Z) of 3 (lithium) and above. It cannot easily detect hydrogen (Z = 1) or helium (Z = 2).Detection limits for most of the elements (on a modern instrument) are in the parts per thousand range. Detection limits of parts per million (ppm) are possible, but require special conditions: concentration at top surface or very long collection time (overnight).XPS is routinely used to analyze inorganic compounds, metal alloys, semiconductors, polymers, elements, catalysts, glasses, ceramics, paints, papers, inks, woods, plant parts, make-up, teeth, bones, medical implants, bio-materials, viscous oils, glues, ion-modified materials and many others.XPS is less routinely used to analyze the hydrated forms of some of the above materials by freezing the samples in their hydrated state in an ultra pure environment, and allowing or causing multilayers of ice to sublime away prior to analysis. Such hydrated XPS analysis allows hydrated sample structures, which may be different from vacuum-dehydrated sample structures, to be studied in their more relevant as-used hydrated structure. Many bio-materials such as hydrogels are examples of such samples.