
design synthesis and functionalization of self assembled
... Werner in 1893, the field of coordination chemistry have grown tremendously. It led to the understanding of the synthesis, structure and reactivity of novel complexes and materials from simple metal-ligand complexes to organometallic catalysts and extended inorganic polymers. In recent decades two b ...
... Werner in 1893, the field of coordination chemistry have grown tremendously. It led to the understanding of the synthesis, structure and reactivity of novel complexes and materials from simple metal-ligand complexes to organometallic catalysts and extended inorganic polymers. In recent decades two b ...
Week 1 -- Schedule
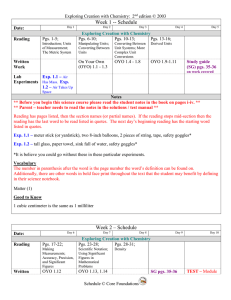
... ** Before you begin this science course please read the student notes in the book on pages i-iv. ** ** Parent – teacher needs to read the notes in the solutions / test manual ** Reading has pages listed, then the section names (or partial names). If the reading stops mid-section then the reading has ...
... ** Before you begin this science course please read the student notes in the book on pages i-iv. ** ** Parent – teacher needs to read the notes in the solutions / test manual ** Reading has pages listed, then the section names (or partial names). If the reading stops mid-section then the reading has ...
The Chemistry and Applications of Metal
... review key developments in these areas and discuss the impact of this chemistry on applications such as gas adsorption and storage, catalysis, and proton conduction. We also discuss the concept of MTV-MOFs in relation to the sequence of functionality arrangement that is influenced by the electronic ...
... review key developments in these areas and discuss the impact of this chemistry on applications such as gas adsorption and storage, catalysis, and proton conduction. We also discuss the concept of MTV-MOFs in relation to the sequence of functionality arrangement that is influenced by the electronic ...
Stoichiometry - Milton
... and light. To account for this emission, scientists proposed a theory that combustion depended on the emission of a substance called phlogiston, which appeared as a combination of energy as heat and light while the material was burning but which could not be detected beforehand. The phlogiston theor ...
... and light. To account for this emission, scientists proposed a theory that combustion depended on the emission of a substance called phlogiston, which appeared as a combination of energy as heat and light while the material was burning but which could not be detected beforehand. The phlogiston theor ...
b - Gordon State College
... 2) Find the moles of each reactant: moles = mass in gram / molar mass 3) Pick up any reactant, say A, and use the stoichiometry to calculate the required amount of the other reactant B. 4) Compare the required amount of B with the available amount of B. a) If required > available, then B is the limi ...
... 2) Find the moles of each reactant: moles = mass in gram / molar mass 3) Pick up any reactant, say A, and use the stoichiometry to calculate the required amount of the other reactant B. 4) Compare the required amount of B with the available amount of B. a) If required > available, then B is the limi ...
Chapter 10 Chemical Calculations and Chemical Equations
... To show how the concentration of solute in solution can be described with molarity, which is moles of solute per liter of solution. To show how to calculate molarity. To show how the molarity of a solution can be translated into a conversion factor that converts between moles of solute and vol ...
... To show how the concentration of solute in solution can be described with molarity, which is moles of solute per liter of solution. To show how to calculate molarity. To show how the molarity of a solution can be translated into a conversion factor that converts between moles of solute and vol ...
Cookies and Chemistry…Huh!?!?
... Limiting/Excess/ Reactant and Theoretical Yield Problems : Potassium superoxide, KO2, is used in rebreathing gas masks to generate oxygen. ...
... Limiting/Excess/ Reactant and Theoretical Yield Problems : Potassium superoxide, KO2, is used in rebreathing gas masks to generate oxygen. ...
Chapter+12
... Limiting/Excess/ Reactant and Theoretical Yield Problems : Potassium superoxide, KO2, is used in rebreathing gas masks to generate oxygen. ...
... Limiting/Excess/ Reactant and Theoretical Yield Problems : Potassium superoxide, KO2, is used in rebreathing gas masks to generate oxygen. ...
Study Guide Chapter 10: An Introduction to Chemistry
... To show how the concentration of solute in solution can be described with molarity, which is moles of solute per liter of solution. To show how to calculate molarity. To show how the molarity of a solution can be translated into a conversion factor that converts between moles of solute and vol ...
... To show how the concentration of solute in solution can be described with molarity, which is moles of solute per liter of solution. To show how to calculate molarity. To show how the molarity of a solution can be translated into a conversion factor that converts between moles of solute and vol ...
COMPARATIVE EVALUATION OF TCF BLEACHED
... distributions confirms the broader distribution and the higher amount of low molecular weight carbohydrate fractions of the sulfite pulps. Figure 1 cornpares sulfite and PHK pulps at different levels of purity. The low molecular weight fraction tan be reduced to a comparable level as in the correspo ...
... distributions confirms the broader distribution and the higher amount of low molecular weight carbohydrate fractions of the sulfite pulps. Figure 1 cornpares sulfite and PHK pulps at different levels of purity. The low molecular weight fraction tan be reduced to a comparable level as in the correspo ...
The science of chemistry is concerned
... a mixture of two or more products containing the same element is formed. For example, when octane (or gasoline in general) burns in an excess of air, the reaction is 2C8H18 + 25O2 → 16CO2 + 18H2O If oxygen is the limiting reagent, however, the reaction does not necessarily stop short of consuming al ...
... a mixture of two or more products containing the same element is formed. For example, when octane (or gasoline in general) burns in an excess of air, the reaction is 2C8H18 + 25O2 → 16CO2 + 18H2O If oxygen is the limiting reagent, however, the reaction does not necessarily stop short of consuming al ...
UNIT 1. SOME BASIC CONCEPTS OF CHEMISTRY Concept
... Ans. Matter can neither be created nor destroyed in the course of a Physical or chemical process although it may change from one form to another. Q4. Which of the following statement about a compound is incorrect?(L2) (I) A molecule of a compound has atom of different elements. (II) A compound canno ...
... Ans. Matter can neither be created nor destroyed in the course of a Physical or chemical process although it may change from one form to another. Q4. Which of the following statement about a compound is incorrect?(L2) (I) A molecule of a compound has atom of different elements. (II) A compound canno ...
AQA Science GCSE Chemistry
... AQA recognizes the importance of good-quality teaching, learning and assessment resources to accompany their specification. That's why they've chosen to work exclusively with nelson Thornes. With AQA examiners providing content and quality control, you can be confident that this course is as closely ...
... AQA recognizes the importance of good-quality teaching, learning and assessment resources to accompany their specification. That's why they've chosen to work exclusively with nelson Thornes. With AQA examiners providing content and quality control, you can be confident that this course is as closely ...
Part 3-ICHO-31-35
... Solutions I, II and III contain a pH indicator HIn (KIn = 4.19×10 ) and other reagents as indicated in the table. The absorbance values at 400 nm of the solutions measured in ...
... Solutions I, II and III contain a pH indicator HIn (KIn = 4.19×10 ) and other reagents as indicated in the table. The absorbance values at 400 nm of the solutions measured in ...
Stoichiometry Chapter 3 CHEMA1301 [Compatibility Mode]
... 1. Combination Reactions In combination reactions two or more substances react to form one product. For example, magnesium metal burns brilliantly in air to produce magnesium oxide: 2Mg(s) + O2(g) g 2 MgO(s) This reaction is used to produce the bright flame generated by flares and some fireworks. A ...
... 1. Combination Reactions In combination reactions two or more substances react to form one product. For example, magnesium metal burns brilliantly in air to produce magnesium oxide: 2Mg(s) + O2(g) g 2 MgO(s) This reaction is used to produce the bright flame generated by flares and some fireworks. A ...
X Science Practice Paper - Brilliant Public School Sitamarhi
... (i) Name the substance which is reduced and which is oxidised? (ii) Name the oxidising agent. Q 23 When magnesium burns in chlorine, it forms Magnesium chloride. On the basis of electron transfer process, state the following (i) Which element is oxidised? (ii) Which one is reduced? Q 24 Oily and fat ...
... (i) Name the substance which is reduced and which is oxidised? (ii) Name the oxidising agent. Q 23 When magnesium burns in chlorine, it forms Magnesium chloride. On the basis of electron transfer process, state the following (i) Which element is oxidised? (ii) Which one is reduced? Q 24 Oily and fat ...
Crosslinking Technical Handbook
... Imidoester crosslinkers react rapidly with amines at alkaline pH to form amidine bonds but have short half-lives. As the pH becomes more alkaline, the half-life and reactivity with amines increases, making crosslinking more efficient when performed at pH 10 than at pH 8. Reaction conditions below pH ...
... Imidoester crosslinkers react rapidly with amines at alkaline pH to form amidine bonds but have short half-lives. As the pH becomes more alkaline, the half-life and reactivity with amines increases, making crosslinking more efficient when performed at pH 10 than at pH 8. Reaction conditions below pH ...
... Considerable advances have been made towards quantifying time-integrated and timeaveraged metamorphic fluid fluxes from the propagation and broadening of reaction and isotope fronts (e.g. Bickle and Baker, 1990; Skelton et al., 1995; Skelton, 2011). However, flux estimates for climate-forcing metamo ...
Late Transition Metal Amido Complexes: Electronic
... 1 Late transition metal amido complexes: metal-ligand bonding and reactivity In organometallic chemistry considerable effort is being expended on the stabilization and versatile adjustment of the reactivity of the containing metal center, e.g. for the design of homogeneous catalysts. In accordance w ...
... 1 Late transition metal amido complexes: metal-ligand bonding and reactivity In organometallic chemistry considerable effort is being expended on the stabilization and versatile adjustment of the reactivity of the containing metal center, e.g. for the design of homogeneous catalysts. In accordance w ...
IB Chemistry Online SAQ_Ans
... g In a continuous spectrum the radiations corresponding to all the wavelengths (within a certain range) are present. In an emission spectrum only a limited number of radiations corresponding to a small number of wavelengths (within a certain range) are present. 10 This is a thought experiment illu ...
... g In a continuous spectrum the radiations corresponding to all the wavelengths (within a certain range) are present. In an emission spectrum only a limited number of radiations corresponding to a small number of wavelengths (within a certain range) are present. 10 This is a thought experiment illu ...
Lewis acid catalysis
In Lewis acid catalysis of organic reactions, a metal-based Lewis acid acts as an electron pair acceptor to increase the reactivity of a substrate. Common Lewis acid catalysts are based on main group metals such as aluminum, boron, silicon, and tin, as well as many early (titanium, zirconium) and late (iron, copper, zinc) d-block metals. The metal atom forms an adduct with a lone-pair bearing electronegative atom in the substrate, such as oxygen (both sp2 or sp3), nitrogen, sulfur, and halogens. The complexation has partial charge-transfer character and makes the lone-pair donor effectively more electronegative, activating the substrate toward nucleophilic attack, heterolytic bond cleavage, or cycloaddition with 1,3-dienes and 1,3-dipoles.Many classical reactions involving carbon–carbon or carbon–heteroatom bond formation can be catalyzed by Lewis acids. Examples include the Friedel-Crafts reaction, the aldol reaction, and various pericyclic processes that proceed slowly at room temperature, such as the Diels-Alder reaction and the ene reaction. In addition to accelerating the reactions, Lewis acid catalysts are able to impose regioselectivity and stereoselectivity in many cases.Early developments in Lewis acid reagents focused on easily available compounds such as TiCl4, BF3, SnCl4, and AlCl3. The relative strengths of these (and other) Lewis acids may be estimated from NMR spectroscopy by the Childs method or the Gutmann-Beckett method. Over the years, versatile catalysts bearing ligands designed for specific applications have facilitated improvement in both reactivity and selectivity of Lewis acid-catalyzed reactions. More recently, Lewis acid catalysts with chiral ligands have become an important class of tools for asymmetric catalysis.Challenges in the development of Lewis acid catalysis include inefficient catalyst turnover (caused by catalyst affinity for the product) and the frequent requirement of two-point binding for stereoselectivity, which often necessitates the use of auxiliary groups.
















![Stoichiometry Chapter 3 CHEMA1301 [Compatibility Mode]](http://s1.studyres.com/store/data/014247793_1-84b4b6fe6fa37d77afbf7eb657ee347a-300x300.png)





