AP Reactions - Georgetown ISD
... In its covalent compounds with nonmetals, hydrogen is assigned an oxidation state of +1. Metal hydrides are an exception; H is at the end of the chemical formula since it has an oxidation state of 1-. The sum of the oxidation states must be zero for an electrically neutral compound. For a polyatomic ...
... In its covalent compounds with nonmetals, hydrogen is assigned an oxidation state of +1. Metal hydrides are an exception; H is at the end of the chemical formula since it has an oxidation state of 1-. The sum of the oxidation states must be zero for an electrically neutral compound. For a polyatomic ...
Section 2 Types of Chemical Reactions Chapter 8
... CH4(g) + O2(g) CO2(g) + 2H2O(g) (partially balanced) • Now consider the number of oxygen atoms. • Increase the number of oxygen atoms on the left side to four by placing the coefficient 2 in front of the molecular formula ...
... CH4(g) + O2(g) CO2(g) + 2H2O(g) (partially balanced) • Now consider the number of oxygen atoms. • Increase the number of oxygen atoms on the left side to four by placing the coefficient 2 in front of the molecular formula ...
SAT Practice Test 3
... Benzene has delocalized pi electrons that stabilize its structure The element with an electron configuration of [He]2s 1 has a greater nuclear charge than fluorine 1 mole of NaCl yields 3 moles of ions in solution Neutrons and protons are both located in the principal energy levels of the atom HCl i ...
... Benzene has delocalized pi electrons that stabilize its structure The element with an electron configuration of [He]2s 1 has a greater nuclear charge than fluorine 1 mole of NaCl yields 3 moles of ions in solution Neutrons and protons are both located in the principal energy levels of the atom HCl i ...
No Slide Title
... 2. Write the ionic equation showing the strong electrolytes completely dissociated into cations and anions. 3. Cancel the spectator ions on both sides of the ionic equation 4. Check that charges and number of atoms are balanced in the net ionic equation ...
... 2. Write the ionic equation showing the strong electrolytes completely dissociated into cations and anions. 3. Cancel the spectator ions on both sides of the ionic equation 4. Check that charges and number of atoms are balanced in the net ionic equation ...
Oxidation-Reduction (Redox) Reactions
... Cl– is –1 You already know what types of ions many of the elements tend to form! ...
... Cl– is –1 You already know what types of ions many of the elements tend to form! ...
Chapter 4
... We assign oxidation states to individual atoms in a reaction to observe the change in electrons. ...
... We assign oxidation states to individual atoms in a reaction to observe the change in electrons. ...
Kinetics in the Study of Organic Reaction Mechanisms
... all mechanisms from which the observed rate equation cannot he derived. I n other words, the chief t~lility of reaction kinetics i n mechanisms studies lies not in establishing the correct mechanism, but in ruling out all of the possible mechanisms which are ineonswtent with the kineticdata. I n the ...
... all mechanisms from which the observed rate equation cannot he derived. I n other words, the chief t~lility of reaction kinetics i n mechanisms studies lies not in establishing the correct mechanism, but in ruling out all of the possible mechanisms which are ineonswtent with the kineticdata. I n the ...
Chapter 7 Lecture
... show what is happening, spectator ions can be omitted. • Equations such as this one, which show only the species that actually participate in the reaction, are called net ionic equations. Ag+(aq) + Cl− (aq) AgCl(s) ...
... show what is happening, spectator ions can be omitted. • Equations such as this one, which show only the species that actually participate in the reaction, are called net ionic equations. Ag+(aq) + Cl− (aq) AgCl(s) ...
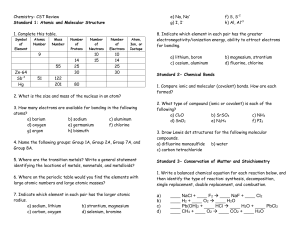
Chemistry- CST Review
... volume at 25 °C if the pressure does not change? 9. A gas at 790 mm Hg and 25 °C occupies a container with an initial volume of 1.20 L. By changing the volume, the pressure of the gas increases to 1500 mm Hg as the temperature is raised to 125 °C. What is the new volume? 10. A 500 mL air sample at a ...
... volume at 25 °C if the pressure does not change? 9. A gas at 790 mm Hg and 25 °C occupies a container with an initial volume of 1.20 L. By changing the volume, the pressure of the gas increases to 1500 mm Hg as the temperature is raised to 125 °C. What is the new volume? 10. A 500 mL air sample at a ...
Paper - Edexcel
... (iii) The student writes this word equation for one of the experiments in which a reaction occurs. bromine + potassium iodide ĺ potassium bromine + iodine The name of one of the substances is incorrect. Write the correct name of this substance. ...
... (iii) The student writes this word equation for one of the experiments in which a reaction occurs. bromine + potassium iodide ĺ potassium bromine + iodine The name of one of the substances is incorrect. Write the correct name of this substance. ...
"Introduction" Kinetics in Process Chemistry: Case Studies Baran Group Meeting Mike DeMartino
... crystallization as the HCl salt was 89-94% overall yield, 350:1 dr, and 99.7 area % purity (HPLC). This corresponds to each step (one nucleophilic addition and two reductions) occurring at > 95% efficiency. When first performed on multi-kilo scale, up to 0.5 area % defluoro was observed. The Grignar ...
... crystallization as the HCl salt was 89-94% overall yield, 350:1 dr, and 99.7 area % purity (HPLC). This corresponds to each step (one nucleophilic addition and two reductions) occurring at > 95% efficiency. When first performed on multi-kilo scale, up to 0.5 area % defluoro was observed. The Grignar ...
Oregon State University, Summer 2009 Chemistry 121 Midterm
... Which of the answers correctly best describes the following list of compounds? Nitric acid (HNO3), ammonia (NH3), sucrose (C12H22O11), KCl, acetic acid (CH3COOH) a. Strong electrolyte, weak acid, non-electrolyte, strong acid, weak electrolyte b. Strong acid, weak base, non-electrolyte, strong electr ...
... Which of the answers correctly best describes the following list of compounds? Nitric acid (HNO3), ammonia (NH3), sucrose (C12H22O11), KCl, acetic acid (CH3COOH) a. Strong electrolyte, weak acid, non-electrolyte, strong acid, weak electrolyte b. Strong acid, weak base, non-electrolyte, strong electr ...
ouble Replacement or (Metathesis) Reactions
... These are usually formed from a transition metal surrounded by ligands (polar molecules or negative ions). As a "rule of thumb" you place twice the number of ligands around an ion as the charge on the ion... example: the dark blue Cu(NH 3)42+ (ammonia is used as a test for Cu2+ ions), and Ag(NH3)2+. ...
... These are usually formed from a transition metal surrounded by ligands (polar molecules or negative ions). As a "rule of thumb" you place twice the number of ligands around an ion as the charge on the ion... example: the dark blue Cu(NH 3)42+ (ammonia is used as a test for Cu2+ ions), and Ag(NH3)2+. ...
Are You suprised ?
... For the following reaction 2 NH3(g) ⇌ N2(g) + 3 H2(g) if, Kc = 2.6×10-5 mol2 L-2 at 127oC. Then Kp at the same temperature is: A) 2.8×10-2 atm2 C) 8.2×10-4 atm2 ...
... For the following reaction 2 NH3(g) ⇌ N2(g) + 3 H2(g) if, Kc = 2.6×10-5 mol2 L-2 at 127oC. Then Kp at the same temperature is: A) 2.8×10-2 atm2 C) 8.2×10-4 atm2 ...
+ 2 HCL(aq) CaCl2(aq) + H2O(l) + CO2(g)
... KEY Vocabulary Cation: An ion with a positive charge Anion: An ion with a negative charge Covalent Bond: A bond between two non-metals where a pair of electrons are shared. Ionic Bond: A bond between a non-metal and a metal where electrons are lost or gained. Subscript: A number that represents how ...
... KEY Vocabulary Cation: An ion with a positive charge Anion: An ion with a negative charge Covalent Bond: A bond between two non-metals where a pair of electrons are shared. Ionic Bond: A bond between a non-metal and a metal where electrons are lost or gained. Subscript: A number that represents how ...
General Chemistry Unit 11
... Chemical reactions in which energy is absorbed are endothermic. Energy is required for the reaction to occur. The energy absorbed is often heat energy or electrical energy. Adding electrical energy to metal oxides can separate them into the pure metal and oxygen. Adding electrical energy to sodium c ...
... Chemical reactions in which energy is absorbed are endothermic. Energy is required for the reaction to occur. The energy absorbed is often heat energy or electrical energy. Adding electrical energy to metal oxides can separate them into the pure metal and oxygen. Adding electrical energy to sodium c ...
Time
... - explain the effect of temperature and pressure on the solubility of gases and solids in liquids - express the concentrations of solutions using: Molarity (M, mol/L) , %weight by volume, ppm and ppb; use c = n/v to solve problems regarding solutions ...
... - explain the effect of temperature and pressure on the solubility of gases and solids in liquids - express the concentrations of solutions using: Molarity (M, mol/L) , %weight by volume, ppm and ppb; use c = n/v to solve problems regarding solutions ...
3_2: More Chemical Changes
... the classroom to help you during labs and quizzes • See Ms. B if you need paper, markers, etc. to take home for the night. • DUE MONDAY/TUESDAY ...
... the classroom to help you during labs and quizzes • See Ms. B if you need paper, markers, etc. to take home for the night. • DUE MONDAY/TUESDAY ...
General Chemistry Questions
... 7. Calculate the pH of a solution necessary to just begin the precipitation of Mg(OH)2 when [Mg2+] = 0.001 M. (Ksp for Mg(OH)2 = 1.2 x 10-11) a. b. c. d. e. ...
... 7. Calculate the pH of a solution necessary to just begin the precipitation of Mg(OH)2 when [Mg2+] = 0.001 M. (Ksp for Mg(OH)2 = 1.2 x 10-11) a. b. c. d. e. ...
Lewis acid catalysis
In Lewis acid catalysis of organic reactions, a metal-based Lewis acid acts as an electron pair acceptor to increase the reactivity of a substrate. Common Lewis acid catalysts are based on main group metals such as aluminum, boron, silicon, and tin, as well as many early (titanium, zirconium) and late (iron, copper, zinc) d-block metals. The metal atom forms an adduct with a lone-pair bearing electronegative atom in the substrate, such as oxygen (both sp2 or sp3), nitrogen, sulfur, and halogens. The complexation has partial charge-transfer character and makes the lone-pair donor effectively more electronegative, activating the substrate toward nucleophilic attack, heterolytic bond cleavage, or cycloaddition with 1,3-dienes and 1,3-dipoles.Many classical reactions involving carbon–carbon or carbon–heteroatom bond formation can be catalyzed by Lewis acids. Examples include the Friedel-Crafts reaction, the aldol reaction, and various pericyclic processes that proceed slowly at room temperature, such as the Diels-Alder reaction and the ene reaction. In addition to accelerating the reactions, Lewis acid catalysts are able to impose regioselectivity and stereoselectivity in many cases.Early developments in Lewis acid reagents focused on easily available compounds such as TiCl4, BF3, SnCl4, and AlCl3. The relative strengths of these (and other) Lewis acids may be estimated from NMR spectroscopy by the Childs method or the Gutmann-Beckett method. Over the years, versatile catalysts bearing ligands designed for specific applications have facilitated improvement in both reactivity and selectivity of Lewis acid-catalyzed reactions. More recently, Lewis acid catalysts with chiral ligands have become an important class of tools for asymmetric catalysis.Challenges in the development of Lewis acid catalysis include inefficient catalyst turnover (caused by catalyst affinity for the product) and the frequent requirement of two-point binding for stereoselectivity, which often necessitates the use of auxiliary groups.