How to Assign Oxidation Numbers
... • Hydrogen has oxidation state of +1 except when it is combined with a less electronegative element • The oxidation state of oxygen is –2 except when it is bonded to fluorine (where it may be +1 or +2) and in peroxides where it has an oxidation state of –1 • The sum of the oxidation states of all th ...
... • Hydrogen has oxidation state of +1 except when it is combined with a less electronegative element • The oxidation state of oxygen is –2 except when it is bonded to fluorine (where it may be +1 or +2) and in peroxides where it has an oxidation state of –1 • The sum of the oxidation states of all th ...
Ch. 20- Electrochemistry
... 1. Rusting of iron requires both oxygen and water, and the process can be accelerated by other factors such as pH, presence of salts, contact with metals more difficult to oxidize than iron, and stress on the iron. E. Preventing Corrosion of Iron 1. Objects made of iron are often covered with a coat ...
... 1. Rusting of iron requires both oxygen and water, and the process can be accelerated by other factors such as pH, presence of salts, contact with metals more difficult to oxidize than iron, and stress on the iron. E. Preventing Corrosion of Iron 1. Objects made of iron are often covered with a coat ...
Chem163_Electrochem
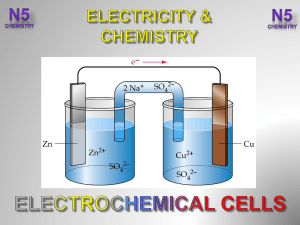
... to an external wire. The electrode at which oxidation occurs is called the anode and the electrode at which reduction occurs is called the cathode. The two half-cells are connected by a salt-bridge that allows movement of ions from one half-cell to the other. When the two electrodes are connected to ...
... to an external wire. The electrode at which oxidation occurs is called the anode and the electrode at which reduction occurs is called the cathode. The two half-cells are connected by a salt-bridge that allows movement of ions from one half-cell to the other. When the two electrodes are connected to ...
Document
... Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
... Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
(.pdf format)
... Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
... Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
Document
... 6. If necessary, equalize the number of electrons in the two halfreactions by multiplying the half-reactions by appropriate coefficients. ...
... 6. If necessary, equalize the number of electrons in the two halfreactions by multiplying the half-reactions by appropriate coefficients. ...
Chapter 18 Review 18.1 Oxidation-Reduction Reactions Oxidation
... where the agents are separated, the electrons flow through a wire, and there is a salt bridge connecting the two solutions Anode- the electrode where oxidation occurs Cathode- the electrode where reduction occurs Electrolysis- electrical energy is used to produce a chemical change - batteries uses e ...
... where the agents are separated, the electrons flow through a wire, and there is a salt bridge connecting the two solutions Anode- the electrode where oxidation occurs Cathode- the electrode where reduction occurs Electrolysis- electrical energy is used to produce a chemical change - batteries uses e ...
Electrochemistry
... 4. The oxidation number of hydrogen is +1 except when it is bonded to metals in binary compounds. In these cases, its oxidation number is –1. 5. Group IA metals are +1, IIA metals are +2 and fluorine is ...
... 4. The oxidation number of hydrogen is +1 except when it is bonded to metals in binary compounds. In these cases, its oxidation number is –1. 5. Group IA metals are +1, IIA metals are +2 and fluorine is ...
Document
... Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
... Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
Oxidation number and Electrolysis(電解)
... concentrated NaCl solution, only H + is discharged at the cathode. But if mercury electrode is used for the cathode, Na + is discharged because sodium metal forms an alloy with mercury. (This method is used in industry for the production of sodium.) ...
... concentrated NaCl solution, only H + is discharged at the cathode. But if mercury electrode is used for the cathode, Na + is discharged because sodium metal forms an alloy with mercury. (This method is used in industry for the production of sodium.) ...
E 0
... Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
... Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
Ch. 20 study questions
... A copper-zinc voltaic cell is constructed using100 mL solutions of 1M solutions of copper sulfate and zinc sulfate with a sodium sulfate salt bridge. After some time, t, had passed at 25C, the concentration of the Zn2+ ions in the anode half cell had increased to 1.50M and the concentration of the ...
... A copper-zinc voltaic cell is constructed using100 mL solutions of 1M solutions of copper sulfate and zinc sulfate with a sodium sulfate salt bridge. After some time, t, had passed at 25C, the concentration of the Zn2+ ions in the anode half cell had increased to 1.50M and the concentration of the ...
Galvanic Cell Lab
... are harder to reduce are easier to oxidize. They are strong reducing agents. Therefore metals are generally easier to oxidize than non-metal ions. A redox reaction is spontaneous when the stronger oxidizing agent is being reduced and the stronger reducing agent is being oxidized. Consequently the ab ...
... are harder to reduce are easier to oxidize. They are strong reducing agents. Therefore metals are generally easier to oxidize than non-metal ions. A redox reaction is spontaneous when the stronger oxidizing agent is being reduced and the stronger reducing agent is being oxidized. Consequently the ab ...
3.-Electrochemical-Cells-V2-
... More advanced rechargeable batteries such as lithium-ion batteries have been developed. The main advantage of these batteries is they are much lighter. ...
... More advanced rechargeable batteries such as lithium-ion batteries have been developed. The main advantage of these batteries is they are much lighter. ...
Unit 5: Electrochemistry
... 3. More about standard reduction potential and Cell Potential Cell potential, also called electromotive force, or emf, is measured in volts and sometimes referred to as the cell voltage. For Eocell, the circle denotes that this is the cell potential at standard conditions. Standard conditions ...
... 3. More about standard reduction potential and Cell Potential Cell potential, also called electromotive force, or emf, is measured in volts and sometimes referred to as the cell voltage. For Eocell, the circle denotes that this is the cell potential at standard conditions. Standard conditions ...
EXAM 007534RR Acids, Bases, and Redox Reactions
... 8. B. Cohesion is exhibited as water molecules are attracted to the molecules in the glass. 9. C. the salt breaks into positive chlorine ions and negative sodium ions. 10. D. high specific heat. 11. C. atoms must not be able to closely approach the hydrogen. 12. B. Three 13. B. None 14. C. Lithium i ...
... 8. B. Cohesion is exhibited as water molecules are attracted to the molecules in the glass. 9. C. the salt breaks into positive chlorine ions and negative sodium ions. 10. D. high specific heat. 11. C. atoms must not be able to closely approach the hydrogen. 12. B. Three 13. B. None 14. C. Lithium i ...
Unit 15 Electrochemistry
... The more positive the Eored value for a half reaction, the greater the tendency for the reactant of the half reaction to be reduced and, therefore, to oxidize another species The half reaction with the smallest reduction potential is most easily reversed as an oxidation The Eored table acts as an a ...
... The more positive the Eored value for a half reaction, the greater the tendency for the reactant of the half reaction to be reduced and, therefore, to oxidize another species The half reaction with the smallest reduction potential is most easily reversed as an oxidation The Eored table acts as an a ...
Answers for Review Questions Exam 3
... Lead Storage – High mass to power ratio, high mass but little power. 9. Electrolysis is the use of an electric current to bring about a chemical change. Reduction and oxidation both occur at the same place as in a galvanic cell, but they have different polarities, - and + respectively. It differs fr ...
... Lead Storage – High mass to power ratio, high mass but little power. 9. Electrolysis is the use of an electric current to bring about a chemical change. Reduction and oxidation both occur at the same place as in a galvanic cell, but they have different polarities, - and + respectively. It differs fr ...
Electrochemistry
Electrochemistry is the branch of physical chemistry that studies chemical reactions which take place at the interface of an electrode, usually a solid metal or a semiconductor, and an ionic conductor, the electrolyte. These reactions involve electric charges moving between the electrodes and the electrolyte (or ionic species in a solution). Thus electrochemistry deals with the interaction between electrical energy and chemical change.When a chemical reaction is caused by an externally supplied current, as in electrolysis, or if an electric current is produced by a spontaneous chemical reaction as in a battery, it is called an electrochemical reaction. Chemical reactions where electrons are transferred directly between molecules and/or atoms are called oxidation-reduction or (redox) reactions. In general, electrochemistry describes the overall reactions when individual redox reactions are separate but connected by an external electric circuit and an intervening electrolyte.