The structure of a fiber optic cable
... permits transmission over longer distances and at higher bandwidths (data rates) than other forms of communications. Fibers are used instead of metal wires because signals travel along them with less loss and are also immune to electromagnetic interference. Fibers are also used for illumination, and ...
... permits transmission over longer distances and at higher bandwidths (data rates) than other forms of communications. Fibers are used instead of metal wires because signals travel along them with less loss and are also immune to electromagnetic interference. Fibers are also used for illumination, and ...
Ionic Liquids Beyond Simple Solvents: Glimpses at the State of the
... were easily and efficiently catalyzed by ionic liquids, especially by ones being derived from l-proline.[39, 40] The example shown in Scheme 3 is one in which the organocatalyst is covalently attached to an IL cation. In a recent review, Headley and Ni have called this concept ionic-liquidsupported ...
... were easily and efficiently catalyzed by ionic liquids, especially by ones being derived from l-proline.[39, 40] The example shown in Scheme 3 is one in which the organocatalyst is covalently attached to an IL cation. In a recent review, Headley and Ni have called this concept ionic-liquidsupported ...
Influence of Green, Red and Blue Light Emitting Diodes on
... environment [4,20], as the primary response of plants during photosynthesis completely depends on light conditions. Plant growth and productivity depends on the light conditions [21] and photosynthetic metabolism is detrimentally affected by light intensity. Plants have developed a sophisticated mec ...
... environment [4,20], as the primary response of plants during photosynthesis completely depends on light conditions. Plant growth and productivity depends on the light conditions [21] and photosynthetic metabolism is detrimentally affected by light intensity. Plants have developed a sophisticated mec ...
10.1 Ray Optics: Reflection and Refraction
... Light moves through air slightly more slowly than through a vacuum. The speed of light through water or glass is considerably slower than through a vacuum. The index of refraction is a property of a transparent material defined as the ratio of the speed of light in a vacuum (c) to the speed of light ...
... Light moves through air slightly more slowly than through a vacuum. The speed of light through water or glass is considerably slower than through a vacuum. The index of refraction is a property of a transparent material defined as the ratio of the speed of light in a vacuum (c) to the speed of light ...
EFFECT OF LEWIS ACID IN TiCl4/MgCl2/THF/AlCl3 CATALYST
... activity due to Fe-Al had an efficiency to remove higher amount of THF, which can be confirmed by XRD and FT-IR. Comparison for electronegativity (EN) values of Ca, Fe and Zn, they are 1.04, 1.64 and 1.66, respectively. Although EN values of Fe is as high as Zn, the radius of Fe2+ (0.61 Å) is closer ...
... activity due to Fe-Al had an efficiency to remove higher amount of THF, which can be confirmed by XRD and FT-IR. Comparison for electronegativity (EN) values of Ca, Fe and Zn, they are 1.04, 1.64 and 1.66, respectively. Although EN values of Fe is as high as Zn, the radius of Fe2+ (0.61 Å) is closer ...
Visible Light
... air, pure water, and clear glass. You can see clearly through a transparent object, such as the revolving glass doors in the figure, because all the light passes straight through it. • Translucent matter is matter that transmits but scatters light. Light passes through a translucent object but you c ...
... air, pure water, and clear glass. You can see clearly through a transparent object, such as the revolving glass doors in the figure, because all the light passes straight through it. • Translucent matter is matter that transmits but scatters light. Light passes through a translucent object but you c ...
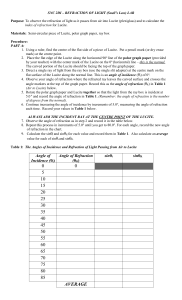
SNC 2PI – REFRACTION OF LIGHT LAB
... 3. Make a note in Table 2 of the angle of incidence when the angle of refraction is 90o. This is the critical angle of incidence for light passing from Lucite to Air. You do not have to collect any more data after this point. Table 2: The Angles of Incidence and Refraction of Light Passing from Luci ...
... 3. Make a note in Table 2 of the angle of incidence when the angle of refraction is 90o. This is the critical angle of incidence for light passing from Lucite to Air. You do not have to collect any more data after this point. Table 2: The Angles of Incidence and Refraction of Light Passing from Luci ...
Light
... Temperature. Visible light we are all familiar with and the problems of ultraviolet radiation are becoming better known with the growing concern over links between UV radiation and skin cancer. X-rays are widely used in medicine but exposure to Xrays should be strictly controlled as these rays are v ...
... Temperature. Visible light we are all familiar with and the problems of ultraviolet radiation are becoming better known with the growing concern over links between UV radiation and skin cancer. X-rays are widely used in medicine but exposure to Xrays should be strictly controlled as these rays are v ...
Elements Compounds
... Bond between two atoms that transfer electrons The bond between a metal and a non-metal Creates ions (charged atoms) to be formed ...
... Bond between two atoms that transfer electrons The bond between a metal and a non-metal Creates ions (charged atoms) to be formed ...
Toward Greener Chemistry Methods for Preparation of
... and strain to fracture, there have been numerous efforts to obtain hybrid materials with im‐ proved mechanical properties [2]. Within the structural configurations for this specific applica‐ tion, the “chopped fibers” configuration, as seen in Figure 1(d), has been the most desired. On the other han ...
... and strain to fracture, there have been numerous efforts to obtain hybrid materials with im‐ proved mechanical properties [2]. Within the structural configurations for this specific applica‐ tion, the “chopped fibers” configuration, as seen in Figure 1(d), has been the most desired. On the other han ...
End-of-Chapter Exercises
... spectrum, light is generally sent through a diffraction grating, splitting the light into the various wavelengths that make it up. (a) The first step in the process is to calibrate the grating, so we know the grating spacing. Sodium has two yellow lines that are very close together in wavelength at ...
... spectrum, light is generally sent through a diffraction grating, splitting the light into the various wavelengths that make it up. (a) The first step in the process is to calibrate the grating, so we know the grating spacing. Sodium has two yellow lines that are very close together in wavelength at ...
Light
... The History of the Photoelectric effect In late 19th century it was discovered that electrons were emitted from a zinc plate when it was exposed to ultraviolet (UV) radiation. As visible light and UV are electromagnetic (EM) waves of similar nature, the result suggests that EM radiation carries ener ...
... The History of the Photoelectric effect In late 19th century it was discovered that electrons were emitted from a zinc plate when it was exposed to ultraviolet (UV) radiation. As visible light and UV are electromagnetic (EM) waves of similar nature, the result suggests that EM radiation carries ener ...
Surface-Mediated Visible-Light Photo-oxidation
... Usually only ultraviolet light must be used for photochemical reactions on TiO2 because of its bulk band gap (3.0-3.2 eV). However, we used scanning tunneling microscopy (STM) to observe visible light photo-oxidation reactions of formic acid on the new ordered lattice-work structure of a TiO2(001) s ...
... Usually only ultraviolet light must be used for photochemical reactions on TiO2 because of its bulk band gap (3.0-3.2 eV). However, we used scanning tunneling microscopy (STM) to observe visible light photo-oxidation reactions of formic acid on the new ordered lattice-work structure of a TiO2(001) s ...
Retinal illuminance from vertical daylight openings in office
... The measurements for the different positions and viewing angles are given in Table 3, for the combination daylight - electric lighting. Like the situation with daylight only or electric lighting only all measured results for Efacial and Eretinal are divided by the horizontal illuminance on the desk ...
... The measurements for the different positions and viewing angles are given in Table 3, for the combination daylight - electric lighting. Like the situation with daylight only or electric lighting only all measured results for Efacial and Eretinal are divided by the horizontal illuminance on the desk ...
Photopolymer
A photopolymer is a polymer that changes its properties when exposed to light, often in the ultraviolet or visible region of the electromagnetic spectrum. These changes are often manifested structurally, for example hardening of the material occurs as a result of cross-linking when exposed to light. An example is shown below depicting a mixture of monomers, oligomers, and photoinitiators that conform into a hardened polymeric material through a process called curing,.A wide variety of technologically useful applications rely on photopolymers, for example some enamels and varnishes depend on photopolymer formulation for proper hardening upon exposure to light. In some instances, an enamel can cure in a fraction of a second when exposed to light, as opposed to thermally cured enamels which can require half an hour or longer. Curable materials are widely used for medical, printing, and photoresist technologies. Changes in structural and chemical properties can be induced internally by chromophores that the polymer subunit already possesses, or externally by addition of photosensitive molecules. Typically a photopolymer consists of a mixture of multifunctional monomers and oligomers in order to achieve the desired physical properties, and therefore a wide variety of monomers and oligomers have been developed that can polymerize in the presence of light either through internal or external initiation. Photopolymers undergo a process called curing, where oligomers are cross-linked upon exposure to light, forming what is known as a network polymer. The result of photo curing is the formation of a thermoset network of polymers. One of the advantages of photo-curing is that it can be done selectively using high energy light sources, for example lasers, however, most systems are not readily activated by light, and in this case a photoinitiator is required. Photoinitiators are compounds that upon radiation of light decompose into reactive species that activate polymerization of specific functional groups on the oligomers. An example of a mixture that undergoes cross-linking when exposed to light is shown below. The mixture consists of monomeric styrene and oligomeric acrylates.Most commonly, photopolymerized systems are typically cured through UV radiation, since ultraviolet light is more energetic; however, the development of dye-based photoinitiator systems have allowed for the use of visible light, having potential advantages of processes that are more simple and safe to handle. UV curing in industrial processes has greatly expanded over the past several decades. Many traditional thermally cured and solvent-based technologies can be replaced by photopolymerization technologies. The advantages of photopolymerization over thermally cured polymerization include high rates of polymerization and environmental benefits from elimination of volatile organic solvents.There are two general routes for photoinitiation: free radical and ionic. The general process involves doping a batch of neat polymer with small amounts of photoinitiator, followed by selective radiation of light, resulting a highly cross-linked product. Many of these reactions do not require solvent which eliminates termination path via reaction of initiators with solvent and impurities, in addition to decreasing the overall cost.