Nano-transistors Sensitive to Vibrations in a Single Molecule
... Nanoparticles of many materials have been prepared in the laboratory and all are very fine powders in pure form. We have discovered that nano-particles below a certain size can be transformed into pure liquids by attaching the right molecules to the surface of each particle. The addition of a second ...
... Nanoparticles of many materials have been prepared in the laboratory and all are very fine powders in pure form. We have discovered that nano-particles below a certain size can be transformed into pure liquids by attaching the right molecules to the surface of each particle. The addition of a second ...
Physics Higher Level Radiation and Matter
... NOTE: This is a trial paper and contains questions of the type that will be encountered in the actual unit assessment. The threshold of attainment of the unit assessment (pass mark) is 18 marks. ...
... NOTE: This is a trial paper and contains questions of the type that will be encountered in the actual unit assessment. The threshold of attainment of the unit assessment (pass mark) is 18 marks. ...
Chapter 5 – Organic Analysis
... Coherent light Light that has all its waves pulsating in unison ...
... Coherent light Light that has all its waves pulsating in unison ...
Lighting the Way
... produce heat. • Thing about the Science Mind Molder….. • The battery gave off an electrical charge due to the chemical reaction taken place inside of it. That electrical charge runs through the light bulb creating light and heat energy! ...
... produce heat. • Thing about the Science Mind Molder….. • The battery gave off an electrical charge due to the chemical reaction taken place inside of it. That electrical charge runs through the light bulb creating light and heat energy! ...
Brought to you by: Jonathan E. Mace
... Light from the decay of one excited atom interacts with another similarly excited atom causing that element to emit light This light produced is: in phase, directional, and ...
... Light from the decay of one excited atom interacts with another similarly excited atom causing that element to emit light This light produced is: in phase, directional, and ...
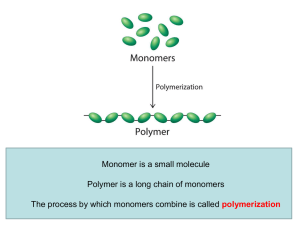
Linear Polymer
... Appearance of real linear polymer chains as recorded using an atomic force microscope on surface under liquid medium. Chain contour length for this polymer is ~204 nm; thickness is ~0.4 nm. (The contour length of a polymer chain is its length at maximum ...
... Appearance of real linear polymer chains as recorded using an atomic force microscope on surface under liquid medium. Chain contour length for this polymer is ~204 nm; thickness is ~0.4 nm. (The contour length of a polymer chain is its length at maximum ...
SSERC Support for practical science (Secondary school pupils)
... rates of reactions or the calculation of concentrations of a compound in an unknown sample. It does this by measuring the absorbance or percentage transmittance of solutions. It measures how much a chemical substance absorbs light by measuring the intensity of light as a beam of light passes through ...
... rates of reactions or the calculation of concentrations of a compound in an unknown sample. It does this by measuring the absorbance or percentage transmittance of solutions. It measures how much a chemical substance absorbs light by measuring the intensity of light as a beam of light passes through ...
Sources and Nature of Light worksheet File
... Main Types of Light Emissions: 1. Incandescence: __________________________________________ _________________________________________________________ 2. Luminescence: __________________________________________________________________________________ Questions: (Ref. P. 403-409) 1) Define fluorescenc ...
... Main Types of Light Emissions: 1. Incandescence: __________________________________________ _________________________________________________________ 2. Luminescence: __________________________________________________________________________________ Questions: (Ref. P. 403-409) 1) Define fluorescenc ...
eprint_5_24935_775
... Because amino acids have both amino and carboxylic groups, they can be ionized. The net ionic charge of an amino acid varies with changes of solution pH. At certain pH an amino acid can be electrically neutral and this pH is called isoelectric point. For simple amino acids which contain only one aci ...
... Because amino acids have both amino and carboxylic groups, they can be ionized. The net ionic charge of an amino acid varies with changes of solution pH. At certain pH an amino acid can be electrically neutral and this pH is called isoelectric point. For simple amino acids which contain only one aci ...
Photopolymer
A photopolymer is a polymer that changes its properties when exposed to light, often in the ultraviolet or visible region of the electromagnetic spectrum. These changes are often manifested structurally, for example hardening of the material occurs as a result of cross-linking when exposed to light. An example is shown below depicting a mixture of monomers, oligomers, and photoinitiators that conform into a hardened polymeric material through a process called curing,.A wide variety of technologically useful applications rely on photopolymers, for example some enamels and varnishes depend on photopolymer formulation for proper hardening upon exposure to light. In some instances, an enamel can cure in a fraction of a second when exposed to light, as opposed to thermally cured enamels which can require half an hour or longer. Curable materials are widely used for medical, printing, and photoresist technologies. Changes in structural and chemical properties can be induced internally by chromophores that the polymer subunit already possesses, or externally by addition of photosensitive molecules. Typically a photopolymer consists of a mixture of multifunctional monomers and oligomers in order to achieve the desired physical properties, and therefore a wide variety of monomers and oligomers have been developed that can polymerize in the presence of light either through internal or external initiation. Photopolymers undergo a process called curing, where oligomers are cross-linked upon exposure to light, forming what is known as a network polymer. The result of photo curing is the formation of a thermoset network of polymers. One of the advantages of photo-curing is that it can be done selectively using high energy light sources, for example lasers, however, most systems are not readily activated by light, and in this case a photoinitiator is required. Photoinitiators are compounds that upon radiation of light decompose into reactive species that activate polymerization of specific functional groups on the oligomers. An example of a mixture that undergoes cross-linking when exposed to light is shown below. The mixture consists of monomeric styrene and oligomeric acrylates.Most commonly, photopolymerized systems are typically cured through UV radiation, since ultraviolet light is more energetic; however, the development of dye-based photoinitiator systems have allowed for the use of visible light, having potential advantages of processes that are more simple and safe to handle. UV curing in industrial processes has greatly expanded over the past several decades. Many traditional thermally cured and solvent-based technologies can be replaced by photopolymerization technologies. The advantages of photopolymerization over thermally cured polymerization include high rates of polymerization and environmental benefits from elimination of volatile organic solvents.There are two general routes for photoinitiation: free radical and ionic. The general process involves doping a batch of neat polymer with small amounts of photoinitiator, followed by selective radiation of light, resulting a highly cross-linked product. Many of these reactions do not require solvent which eliminates termination path via reaction of initiators with solvent and impurities, in addition to decreasing the overall cost.