Energy - GZ @ Science Class Online
... Fossil fuels are a limited resource. Extraction and mining can be expensive and can damage the surrounding area. Carbon dioxide gas that is released upon burning the fuels are contributing to the warming of the climate. Human society has a dependence on fossil fuels for energy but needs to consider ...
... Fossil fuels are a limited resource. Extraction and mining can be expensive and can damage the surrounding area. Carbon dioxide gas that is released upon burning the fuels are contributing to the warming of the climate. Human society has a dependence on fossil fuels for energy but needs to consider ...
FLUIDS, states of matter and thermal energy notes
... the gas constant (R) is given by: k = R/N An advanced look at the relationship between pressure and the kinetic theory: The pressure exerted by an ideal gas on its container is due to the force exerted on the walls of the container by the collisions of the molecules with the walls of area A. The col ...
... the gas constant (R) is given by: k = R/N An advanced look at the relationship between pressure and the kinetic theory: The pressure exerted by an ideal gas on its container is due to the force exerted on the walls of the container by the collisions of the molecules with the walls of area A. The col ...
Acceleration - Solon City Schools
... states that all objects in the universe attract each other through gravitational force. Gravity is the force of attraction between any two objects. F = G (m1 m2/d2) ...
... states that all objects in the universe attract each other through gravitational force. Gravity is the force of attraction between any two objects. F = G (m1 m2/d2) ...
第四章理想气体的热力过程
... To find the Entropy change, start with the expression derived from the first law, replacing dU using the definition of specific heat at constant volume and using the definition of entropy ...
... To find the Entropy change, start with the expression derived from the first law, replacing dU using the definition of specific heat at constant volume and using the definition of entropy ...
1 What Is Energy?
... All matter is made of particles that are always in random motion. Because the particles are in motion, they have kinetic energy. Thermal energy is all of the kinetic energy due to random motion of the particles that make up an object. As you can see in Figure 5, particles move faster at higher tempe ...
... All matter is made of particles that are always in random motion. Because the particles are in motion, they have kinetic energy. Thermal energy is all of the kinetic energy due to random motion of the particles that make up an object. As you can see in Figure 5, particles move faster at higher tempe ...
\bf {The First Law of Thermodynamics for Closed Systems}\\
... Chapter 3 Lecture notes for Thermodynamics: An Engineering Approach, 3rd Ed by Cengel and Boles ...
... Chapter 3 Lecture notes for Thermodynamics: An Engineering Approach, 3rd Ed by Cengel and Boles ...
Assemblage: Exercises in Statistical Mechanics ====== [A] Ensemble Theory - classical gases
... (b) Evaluate the contribution of defects to the entropy and to the specific heat to first order in exp (−ω/2T ). A14. N atoms of mass m of an ideal classical gas are in a cylinder with insulating walls, closed at one end by a piston. The initial volume and temperature are V0 and T0 , respectively. ( ...
... (b) Evaluate the contribution of defects to the entropy and to the specific heat to first order in exp (−ω/2T ). A14. N atoms of mass m of an ideal classical gas are in a cylinder with insulating walls, closed at one end by a piston. The initial volume and temperature are V0 and T0 , respectively. ( ...
Fluids and Thermo Review
... the law which relates the pressure, volume, number of moles, and temperature of an ideal gas internal energy the sum of the potential and kinetic energy of the molecules of a substance kinetic theory of gases the description of matter as being made up of extremely small particles which are in consta ...
... the law which relates the pressure, volume, number of moles, and temperature of an ideal gas internal energy the sum of the potential and kinetic energy of the molecules of a substance kinetic theory of gases the description of matter as being made up of extremely small particles which are in consta ...
Lesson #5 – Electric Potential
... Conservative vs UnConservative Forces If the work done by a force upon a body depends only on the bodies starting and ending point and not upon the path taken then the force is called “conservative” otherwise it is called “non-conservative.” ...
... Conservative vs UnConservative Forces If the work done by a force upon a body depends only on the bodies starting and ending point and not upon the path taken then the force is called “conservative” otherwise it is called “non-conservative.” ...
J. Electrical Systems 7-2 (2011): 225-236 Magnetic bearings in kinetic energy
... The losses in an AMB are mainly due to resistive heating in the coils of the electromagnets and eddy-currents in the core and target material [12]. A large part of these losses are due to the bias current for linearization, which can not be removed completely, since this not only would destroy linea ...
... The losses in an AMB are mainly due to resistive heating in the coils of the electromagnets and eddy-currents in the core and target material [12]. A large part of these losses are due to the bias current for linearization, which can not be removed completely, since this not only would destroy linea ...
Monday, Oct. 28, 2002 - UTA HEP WWW Home Page
... Let’s consider a free ball inside a box under uniform circular motion. How does this motion look like in an inertial frame (or frame outside a box)? ...
... Let’s consider a free ball inside a box under uniform circular motion. How does this motion look like in an inertial frame (or frame outside a box)? ...
No Slide Title
... A 0.5 kg ball is dropped to the floor from a height of 2 m. If it bounces back to a height of 1.8 m, what is the magnitude of its change in momentum? Some energy is lost in the bounce. Just before it hits the ground, its velocity is: (use conservation of ME) mgh=1/2mv2 so v=(2gh)=(2*9.8*2)= 6.26 m ...
... A 0.5 kg ball is dropped to the floor from a height of 2 m. If it bounces back to a height of 1.8 m, what is the magnitude of its change in momentum? Some energy is lost in the bounce. Just before it hits the ground, its velocity is: (use conservation of ME) mgh=1/2mv2 so v=(2gh)=(2*9.8*2)= 6.26 m ...
work is done - Portal UniMAP
... internal energy is a constant. – First Law of Thermodynamics (restated): The total internal energy of an isolated system is constant. • It is impossible to completely isolate a reaction from its surroundings, but it is possible to measure the change in the internal energy of the system, ΔU, as energ ...
... internal energy is a constant. – First Law of Thermodynamics (restated): The total internal energy of an isolated system is constant. • It is impossible to completely isolate a reaction from its surroundings, but it is possible to measure the change in the internal energy of the system, ΔU, as energ ...
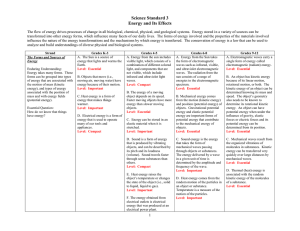
Standard 3: Energy and its Effects
... A. Electromagnetic waves carry a single form of energy called electromagnetic (radiant) energy. Level: Essential B. An object has kinetic energy because of its linear motion, rotational motion, or both. The kinetic energy of an object can be determined knowing its mass and speed. The object’s geomet ...
... A. Electromagnetic waves carry a single form of energy called electromagnetic (radiant) energy. Level: Essential B. An object has kinetic energy because of its linear motion, rotational motion, or both. The kinetic energy of an object can be determined knowing its mass and speed. The object’s geomet ...
Document
... the same potential, i.e. potential is constant everywhere inside a conductor Finally, since one of the points can be arbitrarily close to the surface of the conductor, the electric potential is constant everywhere inside a conductor and equal to its value at the surface! Note that the potential insi ...
... the same potential, i.e. potential is constant everywhere inside a conductor Finally, since one of the points can be arbitrarily close to the surface of the conductor, the electric potential is constant everywhere inside a conductor and equal to its value at the surface! Note that the potential insi ...










![Assemblage: Exercises in Statistical Mechanics ====== [A] Ensemble Theory - classical gases](http://s1.studyres.com/store/data/008930189_1-a7a37d9ca413714c6a603f524253db38-300x300.png)