Specific Heat of Copper
... The heater supplies 850J of energy to the water every second (850W= 850J/s). So in 4minutes 20seconds(260s), energy transferred to the water = 850 x 260 = 221x103J. Answer: 1.75kg Example 6 The 850W heater was then placed into a hole in a piece of copper of mass 1.75kg. (A) Calculate the tempe ...
... The heater supplies 850J of energy to the water every second (850W= 850J/s). So in 4minutes 20seconds(260s), energy transferred to the water = 850 x 260 = 221x103J. Answer: 1.75kg Example 6 The 850W heater was then placed into a hole in a piece of copper of mass 1.75kg. (A) Calculate the tempe ...
Unit 09 - Midland ISD
... Endothermic process- heat absorbing process Exothermic process- heat releasing process Specific heat- Amount of heat it takes to raise 1g of a substance by 1°C ...
... Endothermic process- heat absorbing process Exothermic process- heat releasing process Specific heat- Amount of heat it takes to raise 1g of a substance by 1°C ...
Thermochemistry Lesson 2
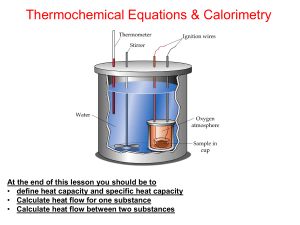
... At the end of this lesson you should be to • define calorimeter and calorimetry • Know five (5) ways to determine enthalpy change for a reaction • Do calculations for enthaphy of reaction using calorimetry ...
... At the end of this lesson you should be to • define calorimeter and calorimetry • Know five (5) ways to determine enthalpy change for a reaction • Do calculations for enthaphy of reaction using calorimetry ...
Energy Worksheet - MICDS Intranet Menu
... A 5.0 kilogram block of ice at -10.0oC is placed in a container of warm water. The entire block of ice is warmed to 0.0oC, and 4.0 kilograms of ice remains unmelted. At this point, how many joules were transferred from the warm water completely? q = mct = 5000gr x 2.09 J/gr oC x (-10oC - 0oC) = - 10 ...
... A 5.0 kilogram block of ice at -10.0oC is placed in a container of warm water. The entire block of ice is warmed to 0.0oC, and 4.0 kilograms of ice remains unmelted. At this point, how many joules were transferred from the warm water completely? q = mct = 5000gr x 2.09 J/gr oC x (-10oC - 0oC) = - 10 ...
Heat is energy transferring in a system and its surroundings.
... How much heat input is needed to raise the temperature of an empty 10 kg vat made of copper from 10°C to 60°C? ...
... How much heat input is needed to raise the temperature of an empty 10 kg vat made of copper from 10°C to 60°C? ...
Aalborg Universitet Measuring Device
... equipment on the test piece leaving some cracks or drill holes on the object. For all of the existing apparatus there is direct contact between the heat absorption sensor (sensor plate) and the surface of the test piece, thereby seriously affecting the result due to the transition insulation factor ...
... equipment on the test piece leaving some cracks or drill holes on the object. For all of the existing apparatus there is direct contact between the heat absorption sensor (sensor plate) and the surface of the test piece, thereby seriously affecting the result due to the transition insulation factor ...
energy sources i
... - The study of conjugate and coupling problems to obtain more full information on heat and mass transfer at the condition of material treatment; the optimization conditions for technology processes and the methods for their realization; - The obtaining the conditions for monitoring, controlling and ...
... - The study of conjugate and coupling problems to obtain more full information on heat and mass transfer at the condition of material treatment; the optimization conditions for technology processes and the methods for their realization; - The obtaining the conditions for monitoring, controlling and ...
q - webhosting.au.edu
... of the HCl and NaOH solutions was the same, 22.50°C, and the final temperature of the mixed solution was 25.86°C. Calculate the heat change for the neutralization reaction on a molar basis: ...
... of the HCl and NaOH solutions was the same, 22.50°C, and the final temperature of the mixed solution was 25.86°C. Calculate the heat change for the neutralization reaction on a molar basis: ...
Heat Transmission Coefficient Measurements in Buildings Utilizing a
... equipment on the test piece leaving some cracks or drill holes on the object. For all of the existing apparatus there is direct contact between the heat absorption sensor (sensor plate) and the surface of the test piece, thereby seriously affecting the result due to the transition insulation factor ...
... equipment on the test piece leaving some cracks or drill holes on the object. For all of the existing apparatus there is direct contact between the heat absorption sensor (sensor plate) and the surface of the test piece, thereby seriously affecting the result due to the transition insulation factor ...
Heat
... • Radiation: The energy emitted by matter in the form of electromagnetic waves (or photons) as a result of the changes in the electronic configurations of the atoms or molecules. • Unlike conduction and convection, the transfer of heat by radiation does not require the presence of an intervening med ...
... • Radiation: The energy emitted by matter in the form of electromagnetic waves (or photons) as a result of the changes in the electronic configurations of the atoms or molecules. • Unlike conduction and convection, the transfer of heat by radiation does not require the presence of an intervening med ...
Thermal Analysis of Heat Transfer Enhancement
... heat sink application [2, 3]. TiO2 in de-ionized water nanofluid has been studied by Naphon and Nakharintr [3] in term of heat transfer characteristic by varying three different channel heights. Sohel et al [2] on the other hand studied effect of different flow rates to thermal performance of Al 2O3 ...
... heat sink application [2, 3]. TiO2 in de-ionized water nanofluid has been studied by Naphon and Nakharintr [3] in term of heat transfer characteristic by varying three different channel heights. Sohel et al [2] on the other hand studied effect of different flow rates to thermal performance of Al 2O3 ...
Energy Efficiency Towards Building Envelope An Analysis
... still low. This is due to the correct building orientation that has been applied during the construction of this building. Correct orientation application is the main factor in the reduction of OTTV value without the need to comply with other factors. If we look at the value of the U-Value where the ...
... still low. This is due to the correct building orientation that has been applied during the construction of this building. Correct orientation application is the main factor in the reduction of OTTV value without the need to comply with other factors. If we look at the value of the U-Value where the ...
convective heat transfer coefficients: experimental
... for heated plates (Rebay et al., 2002) or roof mounted flat plate solar collectors (Sharples and Charlesworth, 1998). Studies about convection heat transfer in buildings are focused mainly on developing theoretical models (Sartori, 2006; Mirsadeghi et al, 2012), and a little on experimental measurem ...
... for heated plates (Rebay et al., 2002) or roof mounted flat plate solar collectors (Sharples and Charlesworth, 1998). Studies about convection heat transfer in buildings are focused mainly on developing theoretical models (Sartori, 2006; Mirsadeghi et al, 2012), and a little on experimental measurem ...
JIF 314 Thermodynamics
... by the amount |QH| from HTR, turning part of this heat into work |W|, and the balance of heat, |QL| =|QH| -|W|, is rejected into the LTR. After the rejection of |QL|, the heat engine’s state will resume to the initial state i. ...
... by the amount |QH| from HTR, turning part of this heat into work |W|, and the balance of heat, |QL| =|QH| -|W|, is rejected into the LTR. After the rejection of |QL|, the heat engine’s state will resume to the initial state i. ...
CHAPTER 3 - RIT
... 3-1C (a) If the lateral surfaces of the rod are insulated, the heat transfer surface area of the cylindrical rod is the bottom or the top surface area of the rod, As D 2 / 4 . (b) If the top and the bottom surfaces of the rod are insulated, the heat transfer area of the rod is the lateral surface ...
... 3-1C (a) If the lateral surfaces of the rod are insulated, the heat transfer surface area of the cylindrical rod is the bottom or the top surface area of the rod, As D 2 / 4 . (b) If the top and the bottom surfaces of the rod are insulated, the heat transfer area of the rod is the lateral surface ...
Energy efficiency in architecture
... Air cavities Air cavities within walls or an attic space in the roof ceiling combination reduce the solar heat gain factor, thereby reducing space-conditioning loads. The performance improves if the void is ventilated. Heat is transmitted through the air cavity by convection and radiation. A cavity ...
... Air cavities Air cavities within walls or an attic space in the roof ceiling combination reduce the solar heat gain factor, thereby reducing space-conditioning loads. The performance improves if the void is ventilated. Heat is transmitted through the air cavity by convection and radiation. A cavity ...
JIF 314 Thermodynamics
... by the amount |QH| from HTR, turning part of this heat into work |W|, and the balance of heat, |QL| =|QH| -|W|, is rejected into the LTR. After the rejection of |QL|, the heat engine’s state will resume to the initial state i. ...
... by the amount |QH| from HTR, turning part of this heat into work |W|, and the balance of heat, |QL| =|QH| -|W|, is rejected into the LTR. After the rejection of |QL|, the heat engine’s state will resume to the initial state i. ...
Common architectural implementations of thermal mass storage are
... can even be beneficial for external finishes to have low thermal mass, as well as low conductivity, to increase the effectiveness of insulation. Thermal lag from mass can greatly reduce the need for insulation in the building envelope, especially in climates with large temperature swings from day t ...
... can even be beneficial for external finishes to have low thermal mass, as well as low conductivity, to increase the effectiveness of insulation. Thermal lag from mass can greatly reduce the need for insulation in the building envelope, especially in climates with large temperature swings from day t ...
Estimation of Atomic Mass from Specific Heat Data
... elements. This approximation has been found to be valid for metals and solid elements with atomic masses over 40. Specific heat can be thought of as the amount of heat required to raise the temperature of one gram of a substance one degree Celsius. Specific heat constants have the units of J/g C. T ...
... elements. This approximation has been found to be valid for metals and solid elements with atomic masses over 40. Specific heat can be thought of as the amount of heat required to raise the temperature of one gram of a substance one degree Celsius. Specific heat constants have the units of J/g C. T ...
Characteristics of a One Dimensional Longitudinal Wave
... In the aerospace overhaul and repair industry, there is a continual drive to lower costs and turn-around time. Some parts are not cost effective to repair because it is more economical for the customer to buy new replacement parts. However, there are parts that are very expensive, therefore, very co ...
... In the aerospace overhaul and repair industry, there is a continual drive to lower costs and turn-around time. Some parts are not cost effective to repair because it is more economical for the customer to buy new replacement parts. However, there are parts that are very expensive, therefore, very co ...
Heat review sheet
... hand by touching a hot spoon in a bowl of hot soup. The hot water transfers its heat through molecules bumping into each other. This continues until the spoon and soup are the same temperature. ...
... hand by touching a hot spoon in a bowl of hot soup. The hot water transfers its heat through molecules bumping into each other. This continues until the spoon and soup are the same temperature. ...
LATENT HEAT STORAGE SYSTEMS
... solar energy is intermittent, unpredictable, and available only during the day. Hence, its application requires efficient thermal energy storage so that the surplus heat collected during sunshine hours may be stored for later use during the night. Similar problems rise in heat recovery systems, wher ...
... solar energy is intermittent, unpredictable, and available only during the day. Hence, its application requires efficient thermal energy storage so that the surplus heat collected during sunshine hours may be stored for later use during the night. Similar problems rise in heat recovery systems, wher ...
SPECIFIC HEAT CAPACITY
... as its temperature changes from 25°C to 20°C? 8. A piece of metal with a mass of 4.68 g absorbs 256 J of heat when its temperature increases by 182°C. What is the specific heat of the metal? 9. If 335 g water at 65.5°C loses 9750 J of heat, what is the final temperature? 10. The temperature of a sam ...
... as its temperature changes from 25°C to 20°C? 8. A piece of metal with a mass of 4.68 g absorbs 256 J of heat when its temperature increases by 182°C. What is the specific heat of the metal? 9. If 335 g water at 65.5°C loses 9750 J of heat, what is the final temperature? 10. The temperature of a sam ...
worksheet
... 9. The temperature of a cup hot chocolate is 110o F. A metal spoon is placed in the hot chocolate. Conduction will continue until the entire spoon reaches __________o F. ...
... 9. The temperature of a cup hot chocolate is 110o F. A metal spoon is placed in the hot chocolate. Conduction will continue until the entire spoon reaches __________o F. ...
Building insulation materials

Building insulation materials are the building materials which form the thermal envelope of a building or otherwise reduce heat transfer.Insulation may be categorized by its composition (natural or synthetic materials), form (batts, blankets, loose-fill, spray foam, and panels), structural contribution (insulating concrete forms, structured panels, and straw bales), functional mode (conductive, radiative, convective), resistance to heat transfer, environmental impacts, and more. Sometimes a thermally reflective surface called a radiant barrier is added to a material to reduce the transfer of heat through radiation as well as conduction. The choice of which material or combination of materials is used depends on a wide variety of factors. Some insulation materials have health risks, some so significant the materials are no longer allowed to be used but remain in use in some older buildings such as asbestos fibers and urea