SHM - ThisIsPhysics
... SHM 1) A body of mass 200 g is executing simple harmonic motion with an amplitude of 20 mm. The maximum force which acts upon it is 0.064 N. Calculate: (a) its maximum velocity (b) its period of oscillation ...
... SHM 1) A body of mass 200 g is executing simple harmonic motion with an amplitude of 20 mm. The maximum force which acts upon it is 0.064 N. Calculate: (a) its maximum velocity (b) its period of oscillation ...
Matter
... – Collection of the combination same type of atom elements and/or – Cannot be compounds or decomposed both. • Compound • USUALLY – 2 or more different heterogeneous atoms chemically bonded together. ...
... – Collection of the combination same type of atom elements and/or – Cannot be compounds or decomposed both. • Compound • USUALLY – 2 or more different heterogeneous atoms chemically bonded together. ...
Chapter 1
... The effect of this notion? I very much fear ’Twill make doubtful all things that were formerly clear. Till soon the cat doctors will say in reports, “We’ve just flipped a coin and we’ve learned he’s a corpse.” ...
... The effect of this notion? I very much fear ’Twill make doubtful all things that were formerly clear. Till soon the cat doctors will say in reports, “We’ve just flipped a coin and we’ve learned he’s a corpse.” ...
In 1913 Bohr proposed his quantized shell model of the atom to
... the atom to explain how electrons can have stable orbits around the nucleus. The motion of the electrons in the Rutherford model was unstable because, according to classical mechanics and electromagnetic theory, any charged particle moving on a curved path emits electromagnetic radiation; thus, the ...
... the atom to explain how electrons can have stable orbits around the nucleus. The motion of the electrons in the Rutherford model was unstable because, according to classical mechanics and electromagnetic theory, any charged particle moving on a curved path emits electromagnetic radiation; thus, the ...
Blackbody Radiation
... • Looking at intensity and Rayleigh-Jean’s law, we have intensity higher as frequency gets higher • In fact, it predicts that the intensity of light at high frequencies will get higher and higher and the total energy radiated will approach infinity – but this is impossible • Great difference between ...
... • Looking at intensity and Rayleigh-Jean’s law, we have intensity higher as frequency gets higher • In fact, it predicts that the intensity of light at high frequencies will get higher and higher and the total energy radiated will approach infinity – but this is impossible • Great difference between ...
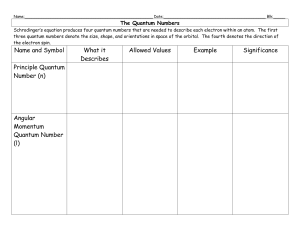
e-the-quantum-numberssv-2
... Name:______________________________________________________ Date:___________________________________________ Blk:_____ ...
... Name:______________________________________________________ Date:___________________________________________ Blk:_____ ...
Instructions - People Server at UNCW
... d. A person pulls a toboggan for a distance of 35 m along the j. A recording engineer works in a soundproofed room that is 40.0 snow with a rope directed at 60o above the snow. The tension in dB quieter than outside. If the intensity in the room is the rope is 100 N. How much work is done on the tob ...
... d. A person pulls a toboggan for a distance of 35 m along the j. A recording engineer works in a soundproofed room that is 40.0 snow with a rope directed at 60o above the snow. The tension in dB quieter than outside. If the intensity in the room is the rope is 100 N. How much work is done on the tob ...
Lecture powerpoint
... W > 0: The environment does work on the system and the system’s energy increases. W < 0: The system does work on the environment and the system’s energy decreases. ...
... W > 0: The environment does work on the system and the system’s energy increases. W < 0: The system does work on the environment and the system’s energy decreases. ...
Quiz 4
... 3. (7 points) Calculate the frequency (in Hz) of a photon emitted by a hydrogen atom when its electron drops from the n = 5 state to the n = 3 state. In this problem ni = 5 and nf = 3. ...
... 3. (7 points) Calculate the frequency (in Hz) of a photon emitted by a hydrogen atom when its electron drops from the n = 5 state to the n = 3 state. In this problem ni = 5 and nf = 3. ...
Lecture 9
... theory is known as Fermi liquid theory. He introduced the idea phenomenologically, and later Abrikosov and Kalatnikov gave a formal derivation using diagrammatic perturbation theory to all orders. Landau suggested describing the excited states of the interacting system as in one-to-one correspondenc ...
... theory is known as Fermi liquid theory. He introduced the idea phenomenologically, and later Abrikosov and Kalatnikov gave a formal derivation using diagrammatic perturbation theory to all orders. Landau suggested describing the excited states of the interacting system as in one-to-one correspondenc ...
PHYSICS 223 Exam-2
... A wave of frequency 120 Hz and wavelength =10 cm is set on the left wire a) The wavelength on the right side will be 40 cm. b) The velocity of the wave will be the same in both sides c) The wavelength of the wave is the same at both sides. d) The tension on the wire at the left side is twice the t ...
... A wave of frequency 120 Hz and wavelength =10 cm is set on the left wire a) The wavelength on the right side will be 40 cm. b) The velocity of the wave will be the same in both sides c) The wavelength of the wave is the same at both sides. d) The tension on the wire at the left side is twice the t ...
L 34 Modern Physics [1]
... Einstein received the 1921 Nobel Prize for explaining the photoelectric effect • A radical idea was needed to explain the photoelectric effect. • Light is an electromagnetic wave, but when it interacts with matter (the metal surface) it behaves like a particle • Light is a particle called a photon ...
... Einstein received the 1921 Nobel Prize for explaining the photoelectric effect • A radical idea was needed to explain the photoelectric effect. • Light is an electromagnetic wave, but when it interacts with matter (the metal surface) it behaves like a particle • Light is a particle called a photon ...

















![L 33 Modern Physics [1] Modern Physics](http://s1.studyres.com/store/data/003217156_1-265c5a519e2bca3f33717b4abd842898-300x300.png)

![L 34 Modern Physics [1]](http://s1.studyres.com/store/data/008622077_1-047a8df5b8f51427a7d951942e25e95f-300x300.png)