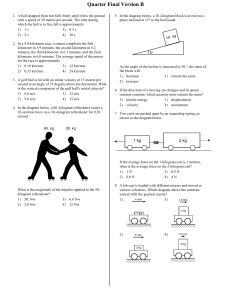
AHSGE Review
... The top of a wave is called the crest, and the bottom is the trough. The measurement from crest to crest or trough to trough is the wavelength. The distance the particles in a medium move when a wave passes is the amplitude. The length of time necessary for a wave to pass is the period, and th ...
... The top of a wave is called the crest, and the bottom is the trough. The measurement from crest to crest or trough to trough is the wavelength. The distance the particles in a medium move when a wave passes is the amplitude. The length of time necessary for a wave to pass is the period, and th ...
Mathcad - MerminBohmEPRBell
... therefore are not independent, no matter how far apart they may be. Together they are in a well-defined correlated state, but their individual properties are uncertain. When measurement determines the state of the particle at A, the correlated property of its distant partner at B becomes known insta ...
... therefore are not independent, no matter how far apart they may be. Together they are in a well-defined correlated state, but their individual properties are uncertain. When measurement determines the state of the particle at A, the correlated property of its distant partner at B becomes known insta ...
Chapter 4.2 Quantum Models
... The Heisenberg uncertainty principle states that it is impossible to determine simultaneously both the position and velocity of an electron or any other particle ...
... The Heisenberg uncertainty principle states that it is impossible to determine simultaneously both the position and velocity of an electron or any other particle ...
Atoms Matter Energy Notes
... Matter is anything that 1. _____________________ and 2. ___________________________ Mass – amount of ________________________ Volume- amount of _____________________________________________ The study of matter is _________________________________ Law of Conservation of Matter Matter cannot be ____ ...
... Matter is anything that 1. _____________________ and 2. ___________________________ Mass – amount of ________________________ Volume- amount of _____________________________________________ The study of matter is _________________________________ Law of Conservation of Matter Matter cannot be ____ ...
Energy Levels and Light Absorption
... • If photons of the right energy are incident on a material, they can cause the promotion of electrons – excited states – The photons are absorbed by the molecules – If the sample is thick enough, the particular wavelengths can be completely absorbed – If white light is used, the absorption of the p ...
... • If photons of the right energy are incident on a material, they can cause the promotion of electrons – excited states – The photons are absorbed by the molecules – If the sample is thick enough, the particular wavelengths can be completely absorbed – If white light is used, the absorption of the p ...
Matter–wave interference of particles selected from a molecular
... individual object is associated with a wave function that may eventually delocalize by far more than the body’s own extension. Numerous experiments have verified this concept at the microscopic scale but intuition wavers when it comes to delocalization experiments with complex objects. While quantum ...
... individual object is associated with a wave function that may eventually delocalize by far more than the body’s own extension. Numerous experiments have verified this concept at the microscopic scale but intuition wavers when it comes to delocalization experiments with complex objects. While quantum ...
Wksht Momentum and Collisions
... 5) A force of 6 N [west] acts on a body for 10 sec. a) What is the impulse acting on the body? b) What is the body's change in momentum ? c) The mass of the body is 3 kg, what is its change in velocity ? 6) A 0.40 kg ball travels toward the wall at a speed of 10 m/s. If it bounces back at 5 m/s, wha ...
... 5) A force of 6 N [west] acts on a body for 10 sec. a) What is the impulse acting on the body? b) What is the body's change in momentum ? c) The mass of the body is 3 kg, what is its change in velocity ? 6) A 0.40 kg ball travels toward the wall at a speed of 10 m/s. If it bounces back at 5 m/s, wha ...
File
... The acceleration and the net force vector are directed perpendicular to each other. False; the acceleration and net force are always directed in the same direction. In this case, F and a are directed inward; this happens to be perpendicular to the tangential velocity vector. If the net force act ...
... The acceleration and the net force vector are directed perpendicular to each other. False; the acceleration and net force are always directed in the same direction. In this case, F and a are directed inward; this happens to be perpendicular to the tangential velocity vector. If the net force act ...
2010 Pacing Pacing Guide - High School Science Help
... are used to reveal relationships and other important features of data. Predictions are made from trends based on the data. The shape of the curve fit to experimentally obtained data is used to determine the relationship of the plotted quantities. All experimental data do not follow a linear re ...
... are used to reveal relationships and other important features of data. Predictions are made from trends based on the data. The shape of the curve fit to experimentally obtained data is used to determine the relationship of the plotted quantities. All experimental data do not follow a linear re ...
NASC 1110
... Electrons are distributed from the lowest energy levels up in agreement with the exclusion principle. In metals, the highest energy band is partially full. In insulators, it is completely full. ...
... Electrons are distributed from the lowest energy levels up in agreement with the exclusion principle. In metals, the highest energy band is partially full. In insulators, it is completely full. ...