Stoichiometry - HCC Learning Web
... • A molar mass is the mass of 1 mol of a substance (i.e., g/mol). • The molar mass of an element is the atomic weight for the element from the periodic table. If it is diatomic, it is twice that atomic weight. • The formula weight (in amu’s) will be the same number as the molar mass (in g/mol). © 20 ...
... • A molar mass is the mass of 1 mol of a substance (i.e., g/mol). • The molar mass of an element is the atomic weight for the element from the periodic table. If it is diatomic, it is twice that atomic weight. • The formula weight (in amu’s) will be the same number as the molar mass (in g/mol). © 20 ...
- Angelo State University
... Chemical Reactions and Chemical Equations • A chemical reaction occurs when atoms of different elements combine and create a new chemical compound, with properties which may be completely unlike those of its constituent elements. • A chemical reaction is written in a standard format called a chemica ...
... Chemical Reactions and Chemical Equations • A chemical reaction occurs when atoms of different elements combine and create a new chemical compound, with properties which may be completely unlike those of its constituent elements. • A chemical reaction is written in a standard format called a chemica ...
Stoichiometry: Calculations with Chemical Formulas and
... • A molar mass is the mass of 1 mol of a substance (i.e., g/mol). • The molar mass of an element is the atomic weight for the element from the periodic table. If it is diatomic, it is twice that atomic weight. • The formula weight (in amu’s) will be the same number as the molar mass (in g/mol). © 20 ...
... • A molar mass is the mass of 1 mol of a substance (i.e., g/mol). • The molar mass of an element is the atomic weight for the element from the periodic table. If it is diatomic, it is twice that atomic weight. • The formula weight (in amu’s) will be the same number as the molar mass (in g/mol). © 20 ...
4 Unit Packet - SRHSchem
... The empirical formulas we have calculated in the preceding section express the simplest atomic ratio between the elements in the compound. An empirical formula does not necessarily represent the actual numbers of atoms present in a molecule of a compound; it represents only the ratio between those n ...
... The empirical formulas we have calculated in the preceding section express the simplest atomic ratio between the elements in the compound. An empirical formula does not necessarily represent the actual numbers of atoms present in a molecule of a compound; it represents only the ratio between those n ...
Chemical equations and stoichiometry
... A mixture of unknown percentage composition of white BaCl2●2H2O crystals and white Na2SO4●10H2O crystals is provided. How could you determine the percent composition of the mixture? 1. How could you get a reaction to occur? 2. What reaction would occur? 3. What would you need to measure? ...
... A mixture of unknown percentage composition of white BaCl2●2H2O crystals and white Na2SO4●10H2O crystals is provided. How could you determine the percent composition of the mixture? 1. How could you get a reaction to occur? 2. What reaction would occur? 3. What would you need to measure? ...
molecular formula
... The atomic mass in grams of any element contains 1 mole of atoms. This is the same number of particles as there are in exactly 12 grams of carbon-12. 1 mole of atoms = 6.0221 x 1023 atoms 1 mole of molecules = 6.0221 x 1023 molecules 1 mole of ions = 6.0221 x 1023 ions 1 mole of formula units = 6.02 ...
... The atomic mass in grams of any element contains 1 mole of atoms. This is the same number of particles as there are in exactly 12 grams of carbon-12. 1 mole of atoms = 6.0221 x 1023 atoms 1 mole of molecules = 6.0221 x 1023 molecules 1 mole of ions = 6.0221 x 1023 ions 1 mole of formula units = 6.02 ...
Mole/Stoich PowerPoint Notes
... • What does a chemical equation ACTUALLY tell you? What does it all mean? • Ex: N2(g) + 3H2(g) ---> 2NH3(g) • This says that 1 molecule of N2 reacts with 3 molecules of H2 to give 2 molecules of NH3 gas. • Let’s say you have 1 mole of nitrogen gas and an unlimited supply of hydrogen gas – 1. How muc ...
... • What does a chemical equation ACTUALLY tell you? What does it all mean? • Ex: N2(g) + 3H2(g) ---> 2NH3(g) • This says that 1 molecule of N2 reacts with 3 molecules of H2 to give 2 molecules of NH3 gas. • Let’s say you have 1 mole of nitrogen gas and an unlimited supply of hydrogen gas – 1. How muc ...
Chemistry MCQs - Target Publications
... (C) Dalton’s (D) Thomson’s The sum of the masses of reactants and products is equal in any physical or chemical reaction. This is in accordance with (A) Law of multiple proportion (B) Law of definite composition (C) Law of conservation of mass (D) Law of reciprocal proportion Pure water can be obtai ...
... (C) Dalton’s (D) Thomson’s The sum of the masses of reactants and products is equal in any physical or chemical reaction. This is in accordance with (A) Law of multiple proportion (B) Law of definite composition (C) Law of conservation of mass (D) Law of reciprocal proportion Pure water can be obtai ...
Chapter 3 Stoichiometry
... A chemical change involves a reorganization of the atoms in one or more substances. Chemical equation is a representation of a chemical reaction with the reactants on the left side of an arrow and the products on the right side C2H5OH + 3O2 2CO2 + 3H2O Reactants Products Atoms have been reorganize ...
... A chemical change involves a reorganization of the atoms in one or more substances. Chemical equation is a representation of a chemical reaction with the reactants on the left side of an arrow and the products on the right side C2H5OH + 3O2 2CO2 + 3H2O Reactants Products Atoms have been reorganize ...
Stoichiometry: Calculations with Chemical Formulas and Equations
... Limiting Reactants If 20.0 g of Fe2O3 are reacted with 8.00 g Al(s) in the following reaction, Which reactant is limiting? How much is left of the excess reagent ?. ...
... Limiting Reactants If 20.0 g of Fe2O3 are reacted with 8.00 g Al(s) in the following reaction, Which reactant is limiting? How much is left of the excess reagent ?. ...
Stoichiometry: Calculations with Chemical Formulas and Equations
... Limiting Reactants If 20.0 g of Fe2O3 are reacted with 8.00 g Al(s) in the following reaction, Which reactant is limiting? How much is left of the excess reagent ?. ...
... Limiting Reactants If 20.0 g of Fe2O3 are reacted with 8.00 g Al(s) in the following reaction, Which reactant is limiting? How much is left of the excess reagent ?. ...
The Mole - Cloudfront.net
... • Molecular Mass/Molecular Weight: If you have a single molecule, mass is measured in amu’s instead of grams. But, the molecular mass/weight is the same numerical value as 1 mole of molecules. Only the units are different. (This is the beauty of Avogadro’s Number!) ...
... • Molecular Mass/Molecular Weight: If you have a single molecule, mass is measured in amu’s instead of grams. But, the molecular mass/weight is the same numerical value as 1 mole of molecules. Only the units are different. (This is the beauty of Avogadro’s Number!) ...
Duplex-single strand denaturing transition in DNA oligomers
... glycol) (PEG) is one of the most effective and widely used. glycol) (PEG) is one of the most effective and widely used. This layer, due to its hydrophilicity, reduces the affinity of nanotubes toward protein binding. We have chosen the biotin - streptavidin binding to demonstrate the effectiveness o ...
... glycol) (PEG) is one of the most effective and widely used. glycol) (PEG) is one of the most effective and widely used. This layer, due to its hydrophilicity, reduces the affinity of nanotubes toward protein binding. We have chosen the biotin - streptavidin binding to demonstrate the effectiveness o ...
Synthesis of novel dendrimers having aspartate grafts and their
... to consider the aspartate based dendrimers could be used as a solubility enhancer just like the PAMAM dendrimers. One hand, as the same of PAMAM dendrimers, this kind of structures consisted of amido linkage and the carbon–carbon bond length was large enough, which created a similar internal microen ...
... to consider the aspartate based dendrimers could be used as a solubility enhancer just like the PAMAM dendrimers. One hand, as the same of PAMAM dendrimers, this kind of structures consisted of amido linkage and the carbon–carbon bond length was large enough, which created a similar internal microen ...
Thermodynamics of Micelle Formation
... concentration of SDS at the inflection point of the titration-curve, n=8592.13. Analysis of the tabulated data revealed a CMC of 7.8 mM SDS at the inflexion midpoint, and approximates the literature value for the at 25°C (8.2 mM [6]). The enthalpy change of demicellization is -4.52 kJ/mol To determi ...
... concentration of SDS at the inflection point of the titration-curve, n=8592.13. Analysis of the tabulated data revealed a CMC of 7.8 mM SDS at the inflexion midpoint, and approximates the literature value for the at 25°C (8.2 mM [6]). The enthalpy change of demicellization is -4.52 kJ/mol To determi ...
ME 533 Lecture 7 Pla..
... • The electric dipole radiation, corresponding to a transition between vibrational levels of the same electronic state, is permitted for molecules having permanent dipole moments pm. • In the framework of the model of the harmonic oscillator,- the selection rule requires v 1 ...
... • The electric dipole radiation, corresponding to a transition between vibrational levels of the same electronic state, is permitted for molecules having permanent dipole moments pm. • In the framework of the model of the harmonic oscillator,- the selection rule requires v 1 ...
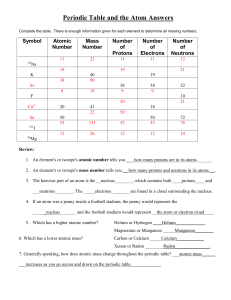
Periodic Table and the Atom Answers
... do this reaction with 125 grams of acetic acid and 275 grams of aluminum hydroxide. Two calculations are required. One determines the quantity of aluminum acetate that can be made with 125 grams of acetic acid and the other determines the quantity of aluminum acetate that can be made using 275 grams ...
... do this reaction with 125 grams of acetic acid and 275 grams of aluminum hydroxide. Two calculations are required. One determines the quantity of aluminum acetate that can be made with 125 grams of acetic acid and the other determines the quantity of aluminum acetate that can be made using 275 grams ...
Chemical Equilibrium
... • The equilibrium constant (K) is the ratio of the mathematical product of the concentrations of substances formed at equilibrium to the mathematical product of the concentrations of reacting substances. Each concentration is raised to a power equal to the coefficient of that substance in the chemic ...
... • The equilibrium constant (K) is the ratio of the mathematical product of the concentrations of substances formed at equilibrium to the mathematical product of the concentrations of reacting substances. Each concentration is raised to a power equal to the coefficient of that substance in the chemic ...
Stoichiometry w RICE
... To convert between moles and mass, you need to use the atomic mass found on the periodic table. Calculate the mass of 0.625 moles of calcium. -According to the periodic table, the atomic mass of calcium is 40.078 amu, so the molar mass of calcium is 40.078 g/mol. ...
... To convert between moles and mass, you need to use the atomic mass found on the periodic table. Calculate the mass of 0.625 moles of calcium. -According to the periodic table, the atomic mass of calcium is 40.078 amu, so the molar mass of calcium is 40.078 g/mol. ...
Determination of the diffusion coefficient of sucrose in water and its
... k is the Boltzmann constant and T is the absolute temperature. There are several important assumptions implicit in equation (17). Two of these are that the solute is spherical, and considerably larger than the solvent molecules. Deviations from spherical geometry (such as oblate or prolate ellipsoid ...
... k is the Boltzmann constant and T is the absolute temperature. There are several important assumptions implicit in equation (17). Two of these are that the solute is spherical, and considerably larger than the solvent molecules. Deviations from spherical geometry (such as oblate or prolate ellipsoid ...
Examples
... The same as: 1) Gram Molecular Mass (for molecules) 2) Gram Formula Mass (ionic compounds) 3) Gram Atomic Mass (for elements) – molar mass is just a much broader term than these other specific masses ...
... The same as: 1) Gram Molecular Mass (for molecules) 2) Gram Formula Mass (ionic compounds) 3) Gram Atomic Mass (for elements) – molar mass is just a much broader term than these other specific masses ...
MOLES AND CALCULATIONS USING THE MOLE CONCEPT
... 1. A mole is the amount of any substance that contains as many elementary entities as there are atoms in exactly 1.00 g of hydrogen-1. 2. A mole is the amount ... in exactly 12.00 g of carbon-12. 3. 6.02 x 1023 of anything 4. It is important to state the entities involved: atoms, molecules, ions, el ...
... 1. A mole is the amount of any substance that contains as many elementary entities as there are atoms in exactly 1.00 g of hydrogen-1. 2. A mole is the amount ... in exactly 12.00 g of carbon-12. 3. 6.02 x 1023 of anything 4. It is important to state the entities involved: atoms, molecules, ions, el ...
1617 Ch3 Practice Test Key Student
... 3. Nitrogen gas, N2, can be prepared by reacting copper (II) oxide with ammonia gas, NH 3. Along with production of N2, the reaction also produces solid copper, Cu, and gaseous water. A) Write a complete balanced equation for the reaction described. The correct formula for copper (II) oxide must be ...
... 3. Nitrogen gas, N2, can be prepared by reacting copper (II) oxide with ammonia gas, NH 3. Along with production of N2, the reaction also produces solid copper, Cu, and gaseous water. A) Write a complete balanced equation for the reaction described. The correct formula for copper (II) oxide must be ...
The Mole - Firelands Local Schools
... If these do not round nicely to whole #s (see example 2 below), multiply all the values by the smallest # that will make all values whole #s. These numbers will become the subscripts on each element ...
... If these do not round nicely to whole #s (see example 2 below), multiply all the values by the smallest # that will make all values whole #s. These numbers will become the subscripts on each element ...
Host–guest chemistry

In supramolecular chemistry, host–guest chemistry describes complexes that are composed of two or more molecules or ions that are held together in unique structural relationships by forces other than those of full covalent bonds. Host–guest chemistry encompasses the idea of molecular recognition and interactions through noncovalent bonding. Noncovalent bonding is critical in maintaining the 3D structure of large molecules, such as proteins and is involved in many biological processes in which large molecules bind specifically but transiently to one another. There are four commonly mentioned types of non-covalent interactions: hydrogen bonds, ionic bonds, van der Waals forces, and hydrophobic interactions.