Solving Problems: A Chemistry Handbook
... from the Sun. You are probably familiar with the damage that exposure to ultraviolet radiation can do to your skin in the form of sunburn. Ultraviolet radiation can also harm other animals and plants. In the 1980s, scientists documented that the ozone layer around Earth was becoming measurably thinn ...
... from the Sun. You are probably familiar with the damage that exposure to ultraviolet radiation can do to your skin in the form of sunburn. Ultraviolet radiation can also harm other animals and plants. In the 1980s, scientists documented that the ozone layer around Earth was becoming measurably thinn ...
Unit 10 complete 2016-2017
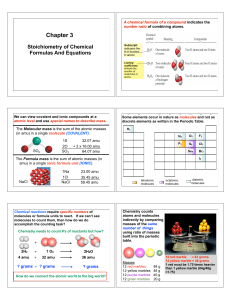
... Percent composition is the percent by mass that each element contributes to the entire molar mass of a compound. 1) Find the molar mass of the compound. 2) Take the mass that the element contributed to the molar mass and divide by the molar mass; multiply by 100 to get %. Ex) H2O molar mass= 18.02g/ ...
... Percent composition is the percent by mass that each element contributes to the entire molar mass of a compound. 1) Find the molar mass of the compound. 2) Take the mass that the element contributed to the molar mass and divide by the molar mass; multiply by 100 to get %. Ex) H2O molar mass= 18.02g/ ...
Honors Chemistry
... Percent composition is the percent by mass that each element contributes to the entire molar mass of a compound. 1) Find the molar mass of the compound. 2) Take the mass that the element contributed to the molar mass and divide by the molar mass; multiply by 100 to get %. Ex) H2O molar mass= 18.02g/ ...
... Percent composition is the percent by mass that each element contributes to the entire molar mass of a compound. 1) Find the molar mass of the compound. 2) Take the mass that the element contributed to the molar mass and divide by the molar mass; multiply by 100 to get %. Ex) H2O molar mass= 18.02g/ ...
Chapter 4 Classifying Reactions: Chemicals in Balance
... (c) Solid barium reacts with solid sulfur to produce solid barium sulfide. (d) Aqueous lead(II) nitrate and solid magnesium react to form aqueous magnesium nitrate and solid lead. What Is Required? You must write the skeleton equation for each of the reactions. What Is Given? The name and state of e ...
... (c) Solid barium reacts with solid sulfur to produce solid barium sulfide. (d) Aqueous lead(II) nitrate and solid magnesium react to form aqueous magnesium nitrate and solid lead. What Is Required? You must write the skeleton equation for each of the reactions. What Is Given? The name and state of e ...
Chapter 3 2013
... 3. The molecular formula will be a whole number multiple of the empirical formula determined BY THE MOLAR MASS ratio ...
... 3. The molecular formula will be a whole number multiple of the empirical formula determined BY THE MOLAR MASS ratio ...
OCR AS Level Chemistry B (Salters) H033
... CHEMISTRY B (SALTERS) H033 For first assessment in 2016 ...
... CHEMISTRY B (SALTERS) H033 For first assessment in 2016 ...
contact - DTU Kemi
... DTU Chemistry works hard to ensure the best possible platform to generate and develop ideas to the benefit of a sustainable development. In order to strengthen the research profile and our visibility further, the Department will be organized in two major sections in 2015. One section will cover Phys ...
... DTU Chemistry works hard to ensure the best possible platform to generate and develop ideas to the benefit of a sustainable development. In order to strengthen the research profile and our visibility further, the Department will be organized in two major sections in 2015. One section will cover Phys ...
B.Sc Chemistry - Calicut University
... evolution of the theory from the above viewpoint. After studying this module, the student realizes the failures of classical physics theories in explaining the microworld and he admits that a new theory is the need of the hour, which is quantum theory. Module II deals with the basic principles (the ...
... evolution of the theory from the above viewpoint. After studying this module, the student realizes the failures of classical physics theories in explaining the microworld and he admits that a new theory is the need of the hour, which is quantum theory. Module II deals with the basic principles (the ...
chemistry (che) - Wisconsin Lutheran College
... spectra. 3 hrs. lab. Prereq: CHE 342 or concurrent enrollment. CHE 350 Biochemistry 1. 3 cr. The first course in a comprehensive biochemistry sequence studying the chemistry of biological systems. Topics include structure and chemistry of amino acids, carbohydrates, lipids, proteins, and nucleic aci ...
... spectra. 3 hrs. lab. Prereq: CHE 342 or concurrent enrollment. CHE 350 Biochemistry 1. 3 cr. The first course in a comprehensive biochemistry sequence studying the chemistry of biological systems. Topics include structure and chemistry of amino acids, carbohydrates, lipids, proteins, and nucleic aci ...
- Academy Test Bank
... 6. The fully hydrated form of sodium sulfate is the decahydrate, Na2SO4 • 10H2O. When heated the hydrated salt loses water. How many water molecules are found per formula unit in a partially dehydrated sample of sodium sulfate with a formula mass of 160.1 amu (i.e. find n for Na2SO4 • nH2O)? A) 1 wa ...
... 6. The fully hydrated form of sodium sulfate is the decahydrate, Na2SO4 • 10H2O. When heated the hydrated salt loses water. How many water molecules are found per formula unit in a partially dehydrated sample of sodium sulfate with a formula mass of 160.1 amu (i.e. find n for Na2SO4 • nH2O)? A) 1 wa ...
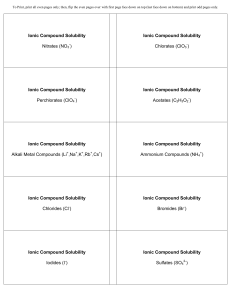
Ionic Compound Solubility Nitrates (NO3 ) Ionic Compound
... To Print, print all even pages only; then, flip the even pages over with first page face down on top (last face down on bottom) and print odd pages only. ...
... To Print, print all even pages only; then, flip the even pages over with first page face down on top (last face down on bottom) and print odd pages only. ...
Amines - ncert
... pair more available for sharing with the proton of the acid. Moreover, the substituted ammonium ion formed from the amine gets stabilised due to dispersal of the positive charge by the +I effect of the alkyl group. Hence, alkylamines are stronger bases than ammonia. Thus, the basic nature of aliphat ...
... pair more available for sharing with the proton of the acid. Moreover, the substituted ammonium ion formed from the amine gets stabilised due to dispersal of the positive charge by the +I effect of the alkyl group. Hence, alkylamines are stronger bases than ammonia. Thus, the basic nature of aliphat ...
Support Material
... Doping is the process of increasing the conductivity of intrinsic semiconductors by adding an appropriate amount of suitable impurity in Si or Ge. * n-type semiconductors : Silicon or Germinium (group-14) doped with electron rich impurity (group-15 element like P or As), Here conductivity is due to ...
... Doping is the process of increasing the conductivity of intrinsic semiconductors by adding an appropriate amount of suitable impurity in Si or Ge. * n-type semiconductors : Silicon or Germinium (group-14) doped with electron rich impurity (group-15 element like P or As), Here conductivity is due to ...
Chapter 6 Quantities in Chemical Reactions
... (http://lardbucket.org) in an effort to preserve the availability of this book. Normally, the author and publisher would be credited here. However, the publisher has asked for the customary Creative Commons attribution to the original publisher, authors, title, and book URI to be removed. Additional ...
... (http://lardbucket.org) in an effort to preserve the availability of this book. Normally, the author and publisher would be credited here. However, the publisher has asked for the customary Creative Commons attribution to the original publisher, authors, title, and book URI to be removed. Additional ...
Higher Chemistry Resources Guide - Glow Blogs
... video can also be found on the RSC website.) The molecular nature of sulfur can be discussed during an exploration of ...
... video can also be found on the RSC website.) The molecular nature of sulfur can be discussed during an exploration of ...
Regents Review Live
... other by 1.7 or more collide and bond (like a metal atom and a nonmetal atom), the one with the higher electronegativity will pull the valence electrons away from the atom with the lower electronegativity to form a (-) ion. The atom that was stripped of its valence electrons forms a (+) ion. If th ...
... other by 1.7 or more collide and bond (like a metal atom and a nonmetal atom), the one with the higher electronegativity will pull the valence electrons away from the atom with the lower electronegativity to form a (-) ion. The atom that was stripped of its valence electrons forms a (+) ion. If th ...
AS/A level
... Further evidence for this model comes from successive ionisation energies. Explain how these provide evidence for aspects of the model described. Sketch the expected pattern of successive ionisation energies for an atom of aluminium and use it to illustrate your answer. ...
... Further evidence for this model comes from successive ionisation energies. Explain how these provide evidence for aspects of the model described. Sketch the expected pattern of successive ionisation energies for an atom of aluminium and use it to illustrate your answer. ...
chemistry - Ethiopian Ministry of Education
... 1.1.2 Major Fields of Chemistry The universe is just like a very big chemical laboratory, rearranging atoms and subatomic particles to produce elements and compounds. While planets are made up of rocks which are nothing but arrangement of compounds, an atmosphere is a mixture of compounds separated ...
... 1.1.2 Major Fields of Chemistry The universe is just like a very big chemical laboratory, rearranging atoms and subatomic particles to produce elements and compounds. While planets are made up of rocks which are nothing but arrangement of compounds, an atmosphere is a mixture of compounds separated ...
Physical Sciences Grade 10 Term 2
... tube and fills the test tube up to the ¾ mark with water. The contents of the test tube are then shaken vigorously to dissolve the chemicals, use a rubber stopper to close the test tube before shaking it. If possible measure the mass of all the test tubes with their contents and record this mass. To ...
... tube and fills the test tube up to the ¾ mark with water. The contents of the test tube are then shaken vigorously to dissolve the chemicals, use a rubber stopper to close the test tube before shaking it. If possible measure the mass of all the test tubes with their contents and record this mass. To ...
5. Coenzyme HAD+ is derived
... 3 Independent work of students under the guidance of a teacher: Working in small groups, the solution situational tasks, consultations, discussions, presentations, essays, performance tests, discuss the results of individual and group assignments. 4 Self-study students work with literature, electron ...
... 3 Independent work of students under the guidance of a teacher: Working in small groups, the solution situational tasks, consultations, discussions, presentations, essays, performance tests, discuss the results of individual and group assignments. 4 Self-study students work with literature, electron ...
molar mass
... Limiting Reactant (p. 128) eg. 2.00 g of NaI reacts with 2.00 g of Pb(NO3)2. Determine the LR and calculate the amount of PbI2 produced. write a balanced equation find n for each reactant (Step #2) find moles produced by each reactant ...
... Limiting Reactant (p. 128) eg. 2.00 g of NaI reacts with 2.00 g of Pb(NO3)2. Determine the LR and calculate the amount of PbI2 produced. write a balanced equation find n for each reactant (Step #2) find moles produced by each reactant ...
Chemistry – A Molecular Sciences Appendices
... A compound is a pure substance that is made up of more than one element. Compounds can be ionic (CAMS Chapter 4) or covalent as described in (CAMS Chapter 5). Covalent compounds are said to be molecular because they exist as discrete molecules, but ionic compounds exist as extended three-dimensional ...
... A compound is a pure substance that is made up of more than one element. Compounds can be ionic (CAMS Chapter 4) or covalent as described in (CAMS Chapter 5). Covalent compounds are said to be molecular because they exist as discrete molecules, but ionic compounds exist as extended three-dimensional ...
AS Chemistry 1
... The formula of a hydrogen carbonate ion is HCO3There must be two hydrogen carbonate ions, each with charge 1î, to balance the two + charges on calcium. The formula with one Ca2+ and two HCO3î is written Ca(HCO3)2 ...
... The formula of a hydrogen carbonate ion is HCO3There must be two hydrogen carbonate ions, each with charge 1î, to balance the two + charges on calcium. The formula with one Ca2+ and two HCO3î is written Ca(HCO3)2 ...